- Title
-
Intralineage directional notch signaling regulates self-renewal and differentiation of asymmetrically dividing radial glia
- Authors
- Dong, Z., Yang, N., Yeo, S.Y., Chitnis, A., and Guo, S.
- Source
- Full text @ Neuron
|
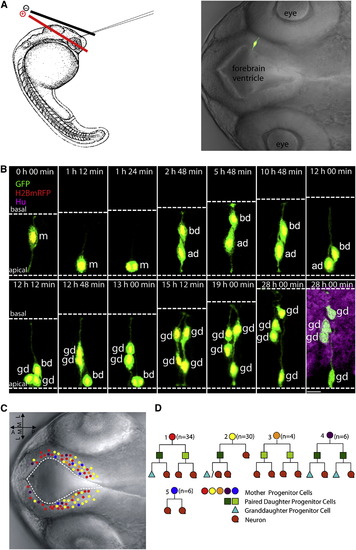
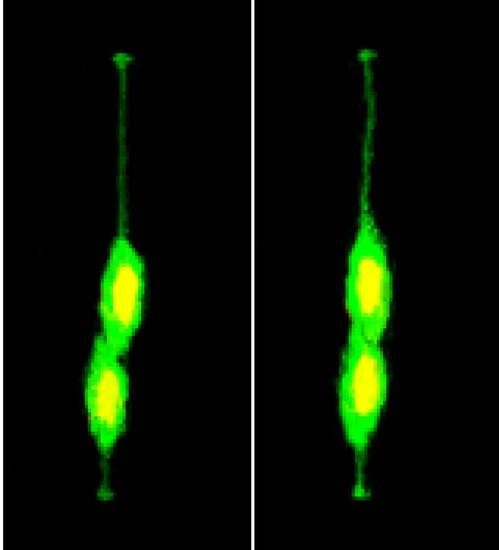
In Vivo Time-Lapse Imaging of Radial Glial Progenitor Cells in the Developing Zebrafish Forebrain Delineates Self-Renewal and Differentiation Divisions.(A) Left view is a schematic of electroporation. Right view is a representative image of a labeled individual radial glia cell in 28 hpf zebrafish forebrain.(B) Representative montage of selected images from time-lapse in vivo imaging of a single fluorescently labeled mother cell. The daughter cell on the left undergoes a differentiation division (generating two neurons), whereas the daughter cell on the right undergoes a self-renewal division (generating one progenitor and one neuron). Dashed white lines indicate the apical (bottom) and basal (top) surfaces. Time is shown on the top of each panel. Zero (0) hr 00 min equals the onset of time-lapse in vivo imaging (28 hpf). m, mother cell; ad, apical daughter cell, which maintains a more apical position; bd, basal daughter cell, which migrates to and maintains a more basal position; gd, granddaughter cells. Scale bar, 10 μm.(C) Nomarski images of zebrafish forebrain depicting the location of the mother cells (A-P, anterior posterior; M-L, medial lateral). Colors represent different cell fate lineages as shown in (D).(D) Different clone types observed by time-lapse in vivo imaging.See also Figure S1 and Movie S1. |
|
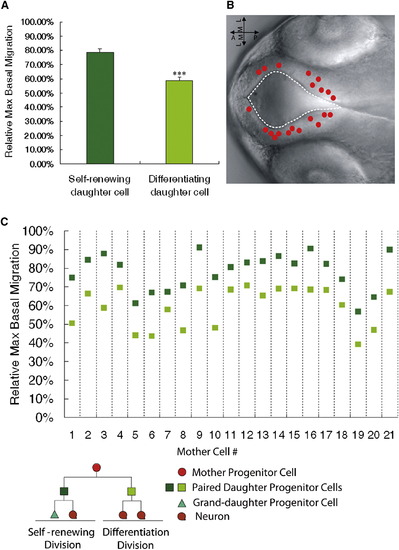
In Vivo Time-Lapse Imaging Coupled with Clonal Analyses Reveals that the Self-Renewing Daughter Cell Migrates More Basally than the Differentiating Sibling(A) Quantification of the relative maximum basal migration of the self-renewing and differentiating siblings in paired daughter cells. ***p < 0.001, t test. Data are shown as the mean ± SEM.(B) Nomarski image of zebrafish forebrain depicting the location of the mother cells giving rise to the 21 paired daughter cells (red circles) that show different cell fates.(C) Relative maximum basal migration of the self-renewing daughter cell (dark green) and the differentiating daughter cell (light green).See also Figure S2. |
|
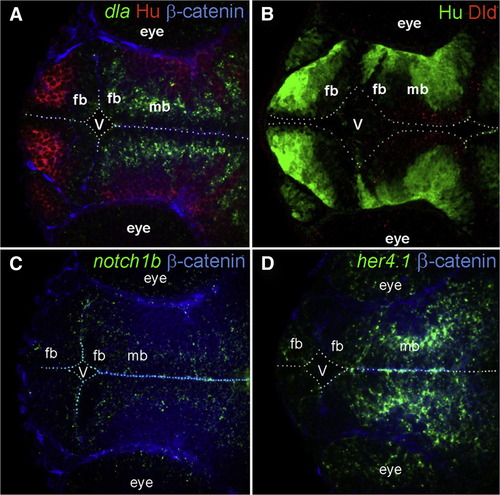
Expression of Notch Signaling Components in the Developing Zebrafish Brain FISH shows the expression of dla (green; A), notch 1b (green; C), and her 4.1 (green; D) in 36 hpf embryos. Fluorescent immunohistochemistry shows the expression of Dld protein (red; B) in 36 hpf embryos. Hu (red in A, green in B) labels postmitotic neurons, and β-catenin (blue) depicts the outline of the embryo. fb, forebrain; V, ventricle; mb, midbrain. |
|
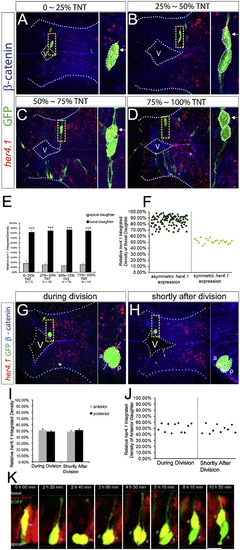
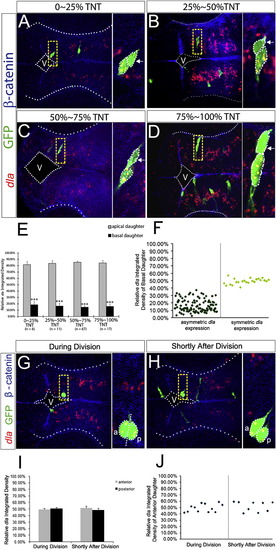
The Basal Daughter Cell Expresses a Higher Level of her4.1(A–D) FISH of her4.1 (red) coupled with immunohistochemistry of GFP (green) and β-catenin (blue) in forebrain paired daughter cells. Images are assembled according to the distance of the basal daughter cell to the ventricular surface. TNT, total neuroepithelium thickness. Enlargement of the yellow-boxed area is shown on the right of each panel. V, ventricle.(E and F) Quantification of the FISH signal of her4.1 in paired daughter cells shown in a bar graph (E) or scatterplot (F). ***p < 0.001 versus apical, t test.(G and H) her4.1 expression in forebrain progenitor cells during division (G) or shortly after division (H). V, ventricle; a, anterior; p, posterior.(I and J) Quantification for (G) and (H) in bar graph (I) or scatterplot (J).(K) Representative montage of selected images from time-lapse in vivo imaging of a single EGFP-labeled mother cell in the hindbrain of her4:dRFP transgenic embryo. m, mother cell; d, daughter cells. Scale bar, 10 μm. Error bars in (E) and (I) represent SEM.See also Figure S3 and Movie S2. |
|
The Apical Daughter Cell Expresses a Higher Level of dla (A–D) FISH of dla (red) coupled with immunohistochemistry of GFP (green) and β-catenin (blue) in forebrain paired daughter cells. TNT, total neuroepithelium thickness; V, ventricle. (E and F) Quantification for (A)–(D) in bar graph (E) and scatterplot (F). ***p < 0.001 versus apical, t test. (G and H) dla expression during (G) or shortly after division (H). V, ventricle; a, anterior; p, posterior. (I and J) Quantification for (G) and (H) in bar graph (I) and scatterplot (J). Error bars in (E) and (I) represent SEM. See also Figure S4. |
|
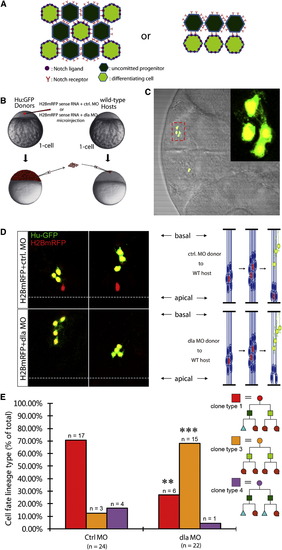
Clonal Knockdown of dla Reveals Intralineage Notch Signaling in Daughter Cells of Asymmetric Division(A) A schematic depicting two different modes of Notch signaling. Left view shows lateral inhibition. Right view illustrates intralineage.(B) Overview of the transplantation strategy.(C) Representative image of a single four-cell clone. Inset is the enlargement of the area highlighted by the red dashed box.(D) Representative images of two single clones in control morpholino group (top) and dla morpholino group (bottom). In control, two single four-cell clones contain one progenitor and three neuronal granddaughter cells. In the dla morpholino group, two single four-cell clones contain four neuronal granddaughter cells.(E) Quantification for (D).**p < 0.01, ***p < 0.001 versus Ctrl MO, z test.See also Figure S5. |
|
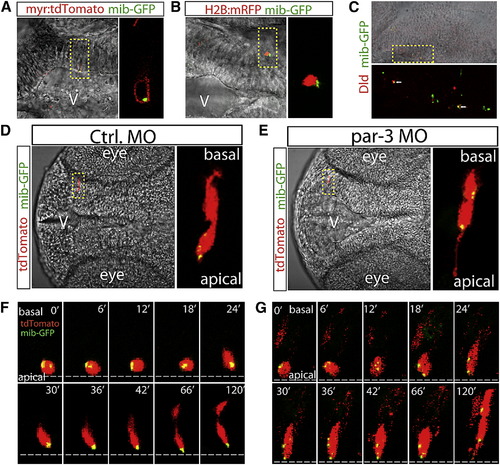
The Fate Determinant Mib Is Unequally Segregated to the Apical Daughter in Par-3-Dependent Manner(A) Mib-GFP is detected in the cytosol within the membrane-targeted myr:tdTomato.(B) Mib-GFP is detected in close proximity to the nucleus reporter H2B:mRFP.(C) Mib-GFP colocalizes with Dld.(D) Mib-GFP was unequally segregated into the apical daughter.(E) The unequal segregation of mib-GFP was eliminated by knockdown of par-3.(F) Selected frames of time-lapse live imaging show unequal segregation of Mib-GFP to the apical daughter cell during division.(G) Selected frames of time-lapse live imaging show equal distribution of Mib-GFP in both daughter cells during division in the par-3 morphant.See also Figure S6 and Movies S3 and S4. |
|
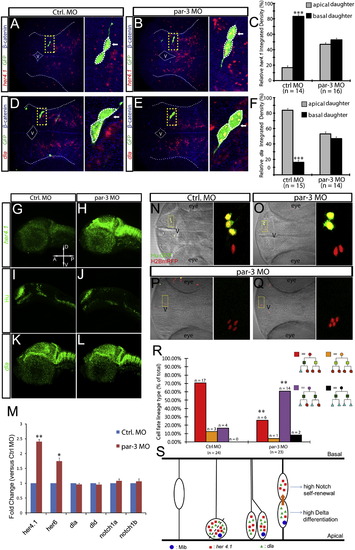
Par-3 Is Essential for Restricting Notch Activity to the Basal Daughter Thereby Limiting Self-Renewal(A–F) her4.1 (A and B) or dla (D and E) expression in control (A and D) and par-3 (B and E) morphants. (C) and (F) are quantifications. ***p < 0.001 versus apical, t test.(G–L) Expression of her4.1 (G and H), Hu (I and J), and dla (K and L) in control (G, I, and K) or par-3 morphants (H, J, and L).(M) Quantitative reverse-transcription PCR shows relative fold change of her4.1, her6, dla, dld, notch1a, and notch1b in control versus par-3 morphants. *p < 0.05, **p < 0.01, versus Ctrl MO.(N–R) Transplantation similar to that described in Figure 5B was carried out. Representative image of four-cell clones derived from control (N) or par-3 morphants (O–Q) in otherwise wild-type brains. In control the single clone is composed of one progenitor (red) and three neurons (green), whereas par-3-deficient clones contained fewer neurons (green) and more progenitors (red). (R) Quantification for (N)–(Q). **p < 0.01, versus Ctrl MO, z test.(S) A model for regulated self-renewal and differentiation in asymmetrically dividing radial glia progenitors. See Discussion.Error bars in (C), (F), and (M) represent SEM.See also Figure S7. |
|
Representative montage of sequential frames from in vivo time-lapse imaging of a single fluorescently-labeled mother cell in the developing zebrafish forebrain, related to Figure 1. From these images, the following can be observed: 1) 0 – 48 min, the mother cell undergoes apically directed INM to reach the apical surface. 2) 1h - 1h 24min: The mother cell divides, and its division axis is largely perpendicular to the apical basal axis. 3) 1h 36 min: The two daughter cells assume a differential positioning along the apical basal neural axis. 4) 1h 48min - 9h: The two daughter cells undergo basally directed INM while maintaining their relative apical basal positions. 5) 9h 12min – 10h 48min: The two daughter cells undergo apically directed INM while maintaining their relative apical basal positions. 6) 11h – 11h 36min: The apical daughter divides with its division axis largely perpendicular to the apical basal axis and generates two granddaughter cells that migrate basally. 7) 11h 48 min – 12h: the basal daughter divides with its division axis largely perpendicular to the apical basal axis and generates two granddaughter cells. ad: apical daughter cell which maintains a more apical position, bd: basal daughter cell which migrates to and maintains a more basal position. |
|
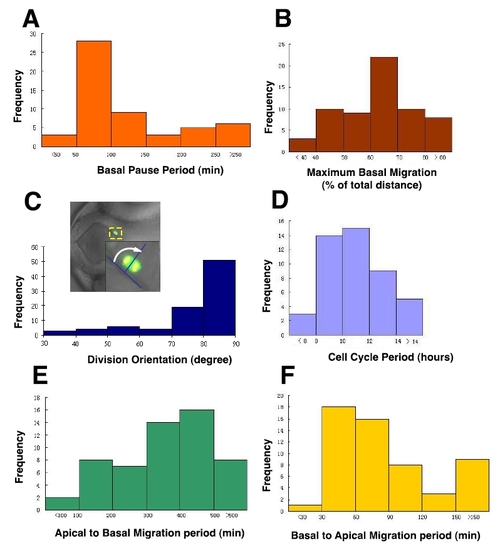
Heterogeneity of radial glia progenitor cell behavior observed through in vivo time-lapse imaging in the developing zebrafish forebrain from 28 hpf to 60 hpf, related to Figure 2. (A-F) Graphs show the frequency distributions of basal pause period (A), maximum basal migration (B), division orientation (C), cell cycle period (D), apical to basal migration period (E), and basal to apical migration period (F). Please refer to the Experimental Procedures for the measurement of these parameters. |
|
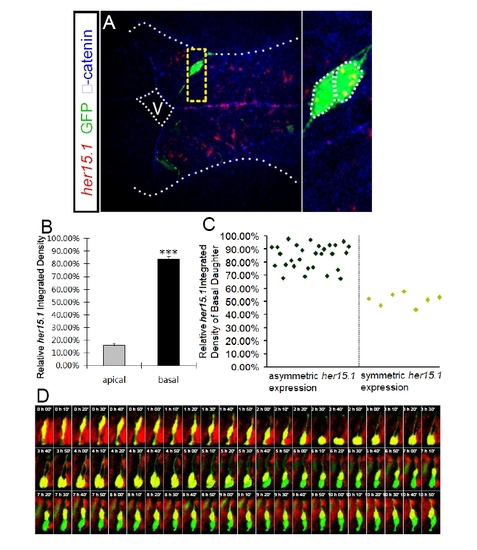
FISH coupled with clonal analyses and live imaging with her4:dRFP transgenic embryo reveal that the basal daughter cell exhibits higher Notch activity, related to Figure 4. (A) FISH of her15.1 (red) coupled with immunohistochemistry of GFP (green) and βcatenin (blue) in forebrain paired daughter cells. The profiles of each daughter cell and the apical and basal surfaces are highlighted by dashed white lines. Enlargement of the yellow-boxed area is shown on the right of each panel. V: ventricle. (B) Quantification the FISH signal of her15.1 in paired daughter cells. The relative integrated density is the ratio of the integrated density in each daughter cell to the sum of the integrated densities of both daughter cells*** p < 0.001 vs apical, t-test. (C) Scatter plot shows the relative her15.1 integrated density of the basal daughter cell of each analyzed pair of daughter cells. 29 pairs of daughter cells show asymmetric her15.1 expression, while 7 pairs of daughter cells show symmetric her15.1 expression. (D) Representative montage of sequential frames from in vivo time-lapse imaging of a single fluorescently-labeled mother cell in the developing zebrafish hindbrain of her4:dRFP transgenic embryo. The dRFP signal reveals dynamic Notch activity in the mother cell and the paired daughter cells. The following can be observed: 1) 0 – 2h 20min, the mother cell undergoes apically directed INM to reach the apical surface. The mother cell has high Notch activity, as shown by the strong dRFP signal. 2) 2h 30min - 2h 50min: The mother cell divides to generate two paired daughter cells. Notch activity is not biased in the paired daughter cells at this stage, since dRFP signal evenly distributes in the paired daughter cells. 3) 3h 00min – 4h 50min, the paired daughter cells embark on basally directed INM while maintaining their relative apical basal positions. They show similar Notch activity. 4) 5h 00min – 10h 50min: The biased Notch activity in the paired daughter cells is observed as a down-regulation of dRFP signal in the apical daughter. There also appears to be a time-dependent decrease of the overall dRFP signal in the imaged tissues, which may reflect a global down-regulation of Notch activity as development progresses, although signal bleaching due to imaging cannot be ruled out. |
|
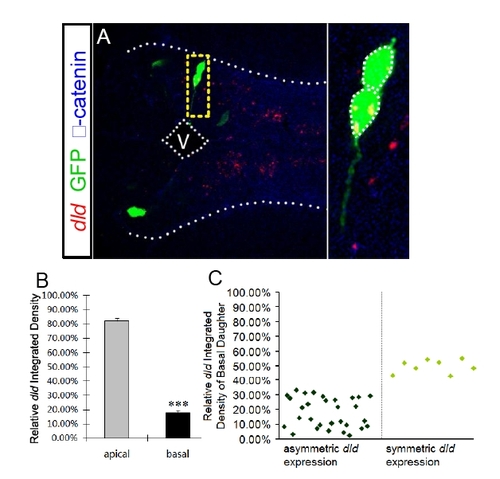
FISH coupled with clonal analyses reveals that the apical daughter cell expresses a higher level of dld, related to Figure 5. (A) FISH of dld (red) coupled with immunohistochemistry of GFP (green) and βcatenin (blue) in forebrain paired daughter cells. The profiles of each daughter cell and the apical and basal surfaces are highlighted by dashed white lines. Enlargement of the yellow-boxed area is shown on the right of each panel. V: ventricle. (B) Quantification the FISH signal of dld in paired daughter cells. *** p < 0.001 vs apical, t-test. (C) Scatter plot shows the relative dld integrated density of the basal daughter cell of each analyzed pair of daughter cells. 30 pairs of daughter cells show asymmetric dld expression, while 8 pairs of daughter cells show symmetric dld expression. |
|
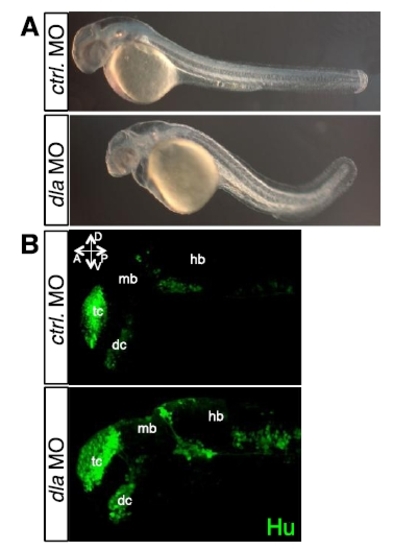
Validation of the dla morpholino antisense oligonucleotides, related to Figure 6. (A) Effect of the dla morpholino on general morphology of 36 hpf embryos. Control morpholino (upper) and dla morpholino (lower) are injected at the 1-cell stage. (B) Effect of the dla morpholino on neurogenesis in 24 hpf embryos. Immunohistochemistry of Hu indicates that Hu positive cells are significantly increased in dla morpholino injected embryos (lower) compared with control morpholino-injected embryos (upper). tc: telencephalon, dc: diencephalon, mb: midbrain, hb: hindbrain. |
|
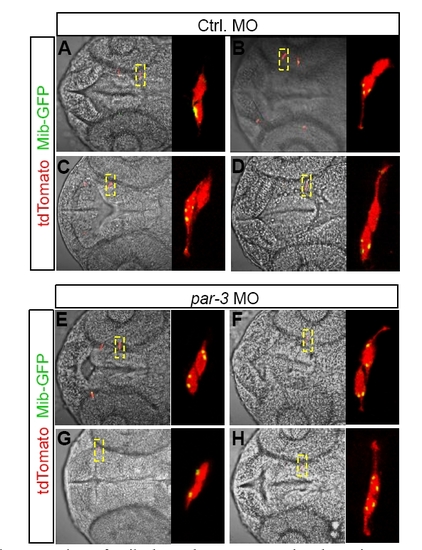
Unequal segregation of Mib depends on Par-3, related to Figure 7. Four representative images showing the localization of Mib-GFP in paired daughter cells in Control (A-D) and par-3 (E-H) morphants. |
|
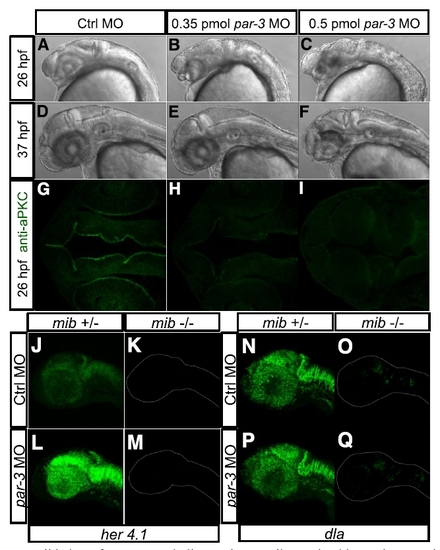
Validation of par-3 morpholino antisense oligonucleotides and expression of her4.1 and dla in mib/par-3 double deficient embryos, related to Figure 8. Injection of 0.35 pmoles par-3 morpholino results in mild defect in brain morphology at 26 hpf (B), while 0.5 pmoles par-3 morpholino results in severe defect in brain morphology at 26 hpf (C), compared with injection of control morpholino (A). At 37 hpf, the morphologic defects of both 0.35 pmoles (E) and 0.5 pmoles par-3 morpholino (F) recover to certain extent, compared with injection of control morpholino (D). (A) and (D), (B) and (E), (C) and (F) represent the same embryo at different developmental stages respectively. Immunostaining of aPKC shows that injection of both 0.35 pmoles (H) and 0.5 pmoles par-3 morpholino (I) can lead to loss of apico-basal cell polarity, as compared with injection of control morpholino (G). (J and K) FISH of her4.1 in control morpholino-injected embryo of mib +/- (J) or mib -/- (K). (L and M) FISH of her4.1 in par-3 morpholino-injected embryo of mib +/- (L) or mib -/- (M). (N and O) FISH of dla in control morpholino-injected embryo of mib +/- (N) or mib -/- (O). (P and Q) FISH of dla in par-3 morpholino-injected embryo of mib +/- (P) or mib -/- (Q). |