- Title
-
Phenothiourea Sensitizes Zebrafish Cranial Neural Crest and Extraocular Muscle Development to Changes in Retinoic Acid and IGF Signaling
- Authors
- Bohnsack, B.L., Gallina, D., and Kahana, A.
- Source
- Full text @ PLoS One
|
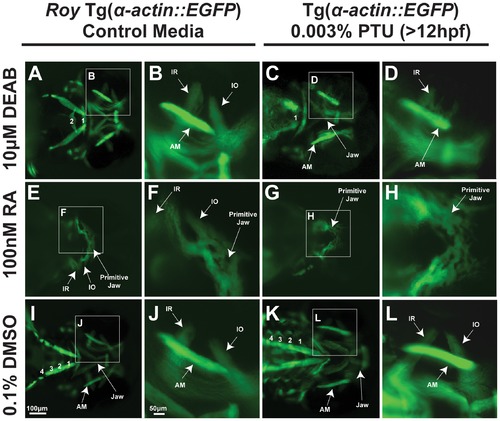
PTU modulates retinoic acid regulation of craniofacial muscles. 72 hpf Tg(α-actin::EGFP) embryos (ventral view) were treated with 10 μM DEAB (A–D) at 24 hpf, 100 nM retinoic acid (RA) (E–F) at 28 hpf, , or 0.1% dimethylsulfoxide (DMSO; I–L) in the roy background (in the absence of PTU; A, B, E, F, I, J) or in the presence of 0.003% PTU which was added to the media at 12 hpf (C, D, G, H, K, L). 10 μM DEAB in the presence of PTU caused thickening of the inferior rectus (C, D) and absence of the inferior oblique (IO) compared to treatment with DEAB in the roy background (absence of PTU; A, B). In addition, only 1 pharyngeal arch (PA) was present in embryos treated with DEAB and PTU (C). Embryos treated with DEAB alone had 2 pharyngeal arches (A) while the control embryos had 4 pharyngeal arches (I). Exogenous retinoic acid inhibited craniofacial muscle development which was worse in the absence of PTU (G. H compared to E, F). AM, anterior mandibulae. |
|
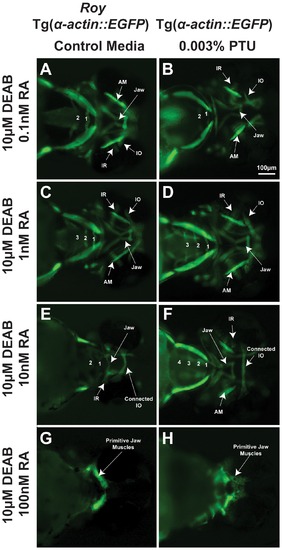
PTU alters sensitivity of craniofacial tissues to retinoic acid. 72 hpf Tg(α-actin::EGFP) embryos (ventral view) were treated with 10 μM DEAB at 24 hpf and increasing concentrations of retinoic acid (RA; 0.1 nM, 1 nM, 10 nM, 100 nM) at 28 hpf in the roy background (in the absence of PTU; A, C, E, G) or presence of 0.003% PTU added at 12 hpf (B, D, F, H). In the roy background and in the absence of PTU, 1 nM retinoic acid improved the DEAB-induced effects on pharyngeal arch formation (C), but at higher concentrations (10 nM (E) and 100 nM (G)), retinoic acid suppressed pharyngeal arch development. DEAB-induced extraocular muscle disorganization in the presence of PTU (Figure 1A, B) was rescued by 0.1 nM retinoic acid (B). The DEAB effects on pharyngeal arch development in the presence of PTU was improved by 1 nM (D) and rescued by 10 nM retinoic acid (F). Teratogenic effects of retinoic acid on jaw musculature and pharyngeal arches were lessened in the presence of PTU (F, H compared to E, G). IR, inferior rectus; IO, inferior oblique; AM, anterior mandibulae. |
|
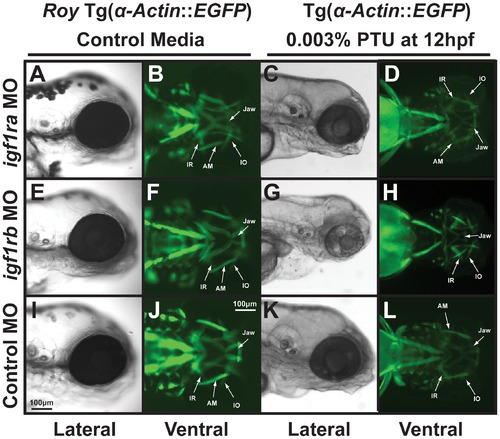
PTU alters IGF regulation of craniofacial muscle development. 72 hpf Tg(α-actin::EGFP) embryos that were injected with morpholinos against igf1ra (A–D), igf1rb (E–H), or control (globin; I–L) and were raised in control media (A, B, E, F, I, J) or with 0.003% PTU at 12 hpf (C, D, G, H, K, L). Morpholino knockdown of igf1ra or igf1rb in the roy background (in the absence of PTU; A, B, E, F) did not disrupt craniofacial development as compared to control (I, J). Morpholino knockdown of igf1ra in the presence of 0.003% PTU caused developmental delay only in the presence of 0.003% PTU (C, D compared to K, L). Knockdown of igf1rb inhibited jaw formation and caused mild thickening of extraocular muscles in the presence of 0.003% PTU (G, H), but not in control media (E, F). IR, inferior rectus; IO, inferior oblique; AM, anterior mandibulae. |
|
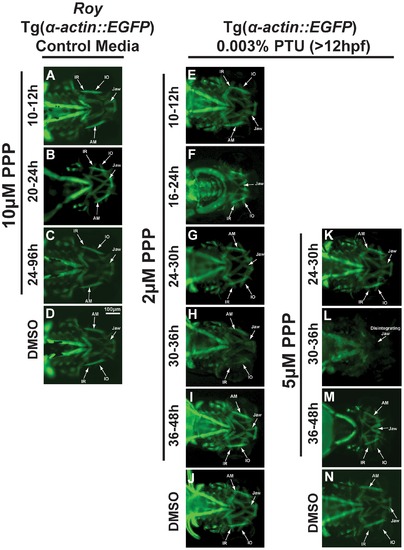
Craniofacial development is sensitive to IGF signaling and PTU during different time frames. 72 hpf Tg(α-actin::EGFP) embryos were treated with the tyrosine kinase receptor inhibitor, picropodophyllin (PPP; 2–10 μM) at different times in the roy background (in the absence of PTU; A–D) or in the presence of 0.003% PTU added at 12 hpf (E–K). Treatment with PPP between 10–12 hours had minimal effect on craniofacial development (A, E) regardless of treatment with 0.003% PTU. In the roy background and in the absence of PTU, treatment with 10 μM PPP between 20 and 24 hpf (B), or 24 and 96 hpf (C) did not affect craniofacial development compared to 0.1% DMSO control (D). Treatment with 2 μM PPP between 16 and 24 hpf in the presence of 0.003% PTU caused craniofacial maldevelopment and thickening of extraocular muscles (F) compared to 0.1% DMSO control (J). Treatment with 2 μM or 5 μM PPP between 24 and 30 hpf caused mild delay of jaw development in the presence of PTU (G). Treatment with 2 μM PPP between 30 and 36 (H) or 36 and 48 (I) hpf did not affect craniofacial development. Treatment with 5 μM PPP between 30 and 36 hpf in the presence of PTU caused poor adherence of the jaw (G) and dissolution of muscle structure after tissue manipulation (L) compared to 0.1% DMSO control (N). Treatment with 5 μM PPP from 36 to 48 hpf caused abnormal jaw and pharyngeal arch formation in the presence of 0.003% PTU (M). Treatment with PPP in the absence of PTU did not affect craniofacial development (M–T). IR, inferior rectus; IO, inferior oblique; AM, anterior mandibulae. |
|
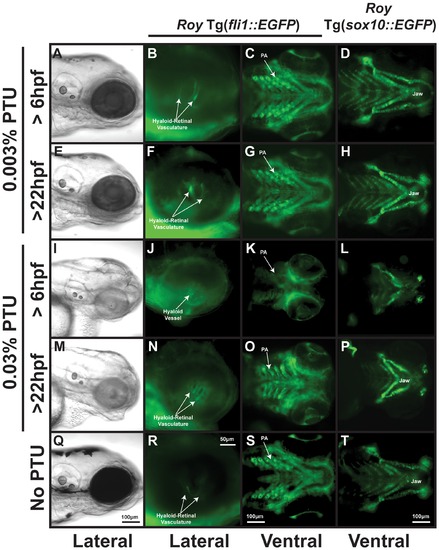
PTU at high concentrations inhibits neural crest development. 96 hpf roy Tg(fli1::EGFP) and roy Tg(sox10::EGFP) embryos were treated with 0.003% (A–H) or 0.03% (I–P) PTU at 6 hpf or 22 hpf. Treatment with 0.003% PTU at 6 (A–D) or 22 (E–H) hpf inhibited pigmentation (A, E) but did not affect development of the retinal-hyaloid vasculature (B, F), pharyngeal arches (PA; C, G), and jaw cartilage (D, H) as compared to untreated embryos (Q–T). At a concentration of 0.03%, PTU at 6 hpf inhibited neural crest development (I–L) including pharyngeal arch development (K), jaw formation (L), and the remodeling of the hyaloid vessel into the hyaloids retinal plexus (J). Treatment with 0.03% PTU at 22 hpf caused pharyngeal arch (O) and jaw malformation (M, P), but did not affect the hyaloid-retinal vasculature (N). |
|
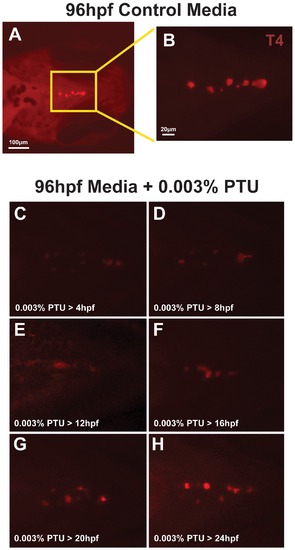
0.003% PTU Inhibits T4 in thyroid follicles. Wholemount immunostaining for T4 in 96 hpf embryos (ventral view) raised in control media (A, B) or in media containing 0.003% PTU. PTU was added at 4, 8, 12, 16, 20, or 24 hpf and carried through until the embryos were harvested at 96 hpf. Untreated 96 hpf embryos showed numerous thyroid follicles that contained T4 (A, B). Addition of 0.003% PTU between 4 and 12 hpf suppressed T4 in follicles at 96 hpf (C, E). Addition of 0.003% PTU between 16 and 20 hpf progressively increases T4 staining and treatment at 24 hpf does not affect T4. |
|
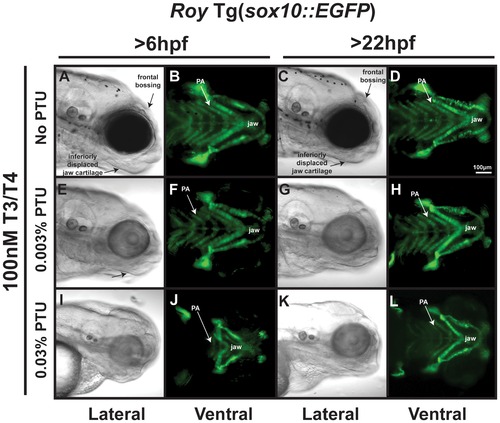
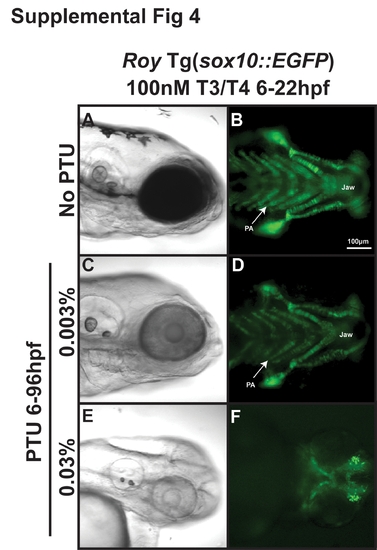
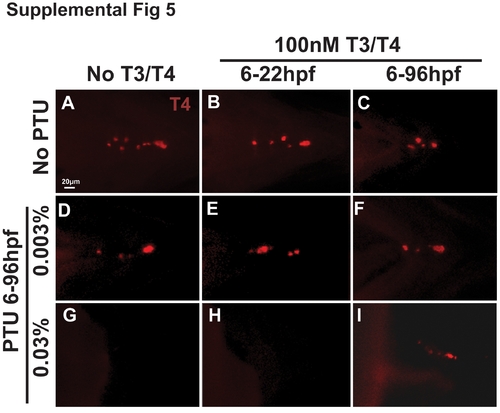
Thyroid hormone partially mediates PTU effect on neural crest. 96 hpf roy Tg(sox10::EGFP) embryos were treated with 100 nM T3/100 nM T4 and 0.003% (E–H) or 0.03% (I–L) PTU at 6 hpf or 22 hpf. High concentrations of T3 and T4 in the absence of PTU caused thickening of jaw cartilage and frontal bossing (A–D)) when added at 6 or 22 hpf. Treatment with 0.003% PTU lessened the effects of exogenous T3 and T4 (E–H compared to I–L). 100 nM T3/100 nM T4 improved, but did not restore the neural crest defects induced by treatment with 0.03% PTU at 6 (I, J compared to Figure 5I, L) or 22 hpf (K, L compared to Figure 5M, P). PA, pharyngeal arches. |
|
PTU lessens teratogenic effect of retinoic acid on neural crest. 72 hpf Tg(sox10::EGFP) embryos (ventral view) were treated with 100 nM retinoic acid (RA; A,B) at 28 hpf or 0.1% dimethylsulfoxide (DMSO; I–L) in the roy background (in the absence of PTU, A, C) or presence of 0.003% PTU (B, D) which was added to the media at 12 hpf. Exogenous retinoic acid inhibited neural crest-derived pharyngeal arch (PA) formation, which was worse in the absence of PTU (B, D compared to A, C). |
|
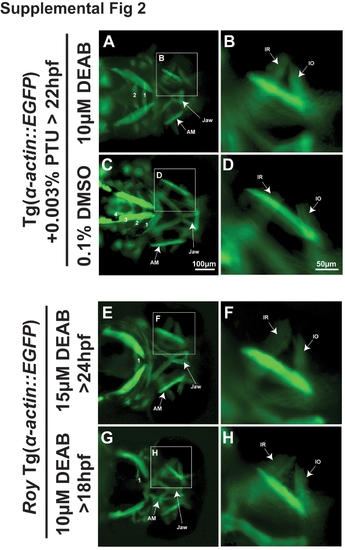
Effect of PTU is time sensitive. 72 hpf Tg(α-actin::EGFP) embryos treated with PTU at 22 hpf and 10 μM DEAB (A, B) at 24 hpf showed shortening of the jaw and only 2 pharyngeal arches, but minimal effect on extraocular muscle development compared to 0.1% DMSO (C, D). In the roy background and in the absence of PTU, treatment with 15 μM DEAB at 24 hpf (E, F) or 10 μM DEAB at 18 hpf (G, H) also disrupted jaw and pharyngeal arch formation, but did not cause thickening or loss of extraocular muscles. |
|
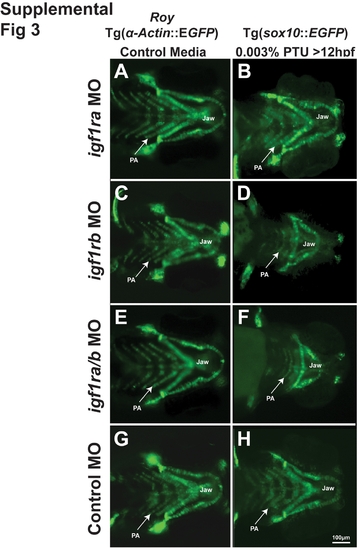
PTU alters IGF regulation of neural crest development. 72 hpf Tg(sox10::EGFP) embryos (ventral view) that were injected with morpholinos against igf1ra (A, B), igf1rb (C, D), igf1ra/igf1rb (E, F) or with a control (globin; G, H) and were raised in control media (roy background, A C, E, G) or media supplemented with 0.003% PTU at 12 hpf (B, D, F, H). Morpholino knockdown of igf1ra caused mild developmental delay only in the presence of 0.003% PTU (B compared to A, G, H). Knockdown of igf1rb or both igf1ra and igf1rb inhibited jaw and pharyngeal arch (PA) formation in the presence of 0.003% PTU (D, F), but not in control media (C, E). |
|
T3 and T4 are required after 22 hpf for rescue of PTU. 96 hpf roy Tg(α-actin::EGFP) were treated with 100 nM T3 and 100 nM T4 from 6 to 22 hpf in the absence (A, B) or presence of 0.003% (C, D) or 0.03% (E, F) PTU from 6 to 96 hpf. Exposure to exogenous T3 and T4 only between 6 and 22 hpf in the absence of PTU (A, B) or in the presence of 0.003% PTU (C, D) did not cause frontal bossing or displacement of jaw cartilage as was seen with exposure to T3 and T4 from 6 to 96 hpf (Figure 7A-D). Furthermore, exogenous T3 and T4 from 6 to 22 hpf did not rescue the craniofacial defects induced by 0.03% PTU (E, F). |
|
T3 and T4 partially restore T4 expression in PTU-treated embryos. Wholemount immunostaining for T4 in 96 hpf embryos (ventral view) demonstrated that in the absence of PTU exogenous treatment with 100 nM T3 and 100 nM T4 between 6 and 96 hpf mildly decreased T4 expression in thyroid follicles (C) compared to embryos treated with T3 and T4 from 6 to 22 hpf (B) and untreated controls (A). Treatment with 0.003% (D) and 0.03% PTU (G) progressively decreased T4 expression compared to embryos raised in the absence of PTU (A). Treatment with exogenous T3 and T4 from 6 to 22 hpf (E) or from 6 to 96 hpf (F) did not improve the effect of 0.003% PTU on T4 expression (D) while exogenous T3 and T4 from 6 to 96 hpf (I), but not from 6 to 22 hpf (H) slightly improved T4 expression in embryos treated with 0.03% PTU (G). |