- Title
-
The transcription factor six1a plays an essential role in the craniofacial myogenesis of zebrafish
- Authors
- Lin, C.Y., Chen, W.T., Lee, H.C., Yang, P.H., Yang, H.J., and Tsai, H.J.
- Source
- Full text @ Dev. Biol.
|
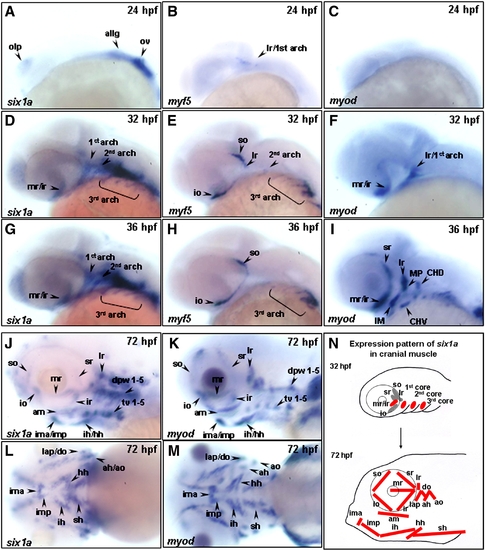
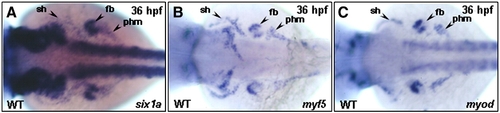
The temporal expressions of six1a, myf5 and myod during cranial muscle development of zebrafish. The temporal expressions of six1a (A, D, G, J, L), myf5 (B, E, H) and myod (C, F, I, K, M) transcripts of zebrafish were analyzed by whole-mount in situ hybridization in embryos from lateral view (A–K) and ventral view (L–M). The transcript of six1a in the olp, ov, and allg at 24 hpf (A); in the mr, ir, 1st, 2nd, and 3rd arches at 32 and 36 hpf (D, G); and in all the cranial muscles at 72 hpf (J, L). The myf5 transcript was detected in the craniofacial region at 24 hpf (B, arrow); in the so, io, 2nd and 3rd arches at 32 hpf (E); and in the so, io, and 3rd arches at 36 hpf (H). Although the myod was not expressed in the craniofacial muscles at 24 hpf (C), it was detected in the mr/ir, lr, and 1st arch at 32 hpf (F); in the mr, ir, sr, lr, MP, IM, CHD, and CHV at 36 hpf (I); and in all the cranial muscles at 72 hpf (K, M). The schematic diagram illustrates the expression of six1a in the cranial muscles during 32–72 hpf (N). ah, adductor hyoideus; allg, anterior lateral line and vestibular ganglia; am, adductor mandibulae; ao, adductor operculi; do, dilator operculi; dpw1–5, dorsal pharyngeal wall 1–5; hh, hyohyoideus; ih, interhyoideus; ima, intermandibularis anterior; imp, intermandibularis posterior; io, inferior oblique; ir, inferior rectus; lap, levator arcus palatini; lr, lateral rectus; mr, medial rectus; olp, olfactory placode; ov, otic vesicle; sh, sternohyoideus; so, superior oblique; sr, superior rectus and tv 1–5, transversus ventralis 1–5. CHD: the constrictor hyoideus dorsalis, which differentiates to ah and ao; CHV: the constrictor hyoideus ventralis, which differentiates to ih and hh; IM: the intermandibularis, which differentiates to ima and imp; MP: the masticatory plate, which differentiates to CD and am. CD: the constrictor dorsalis, which differentiates to lap and do. EXPRESSION / LABELING:
|
|
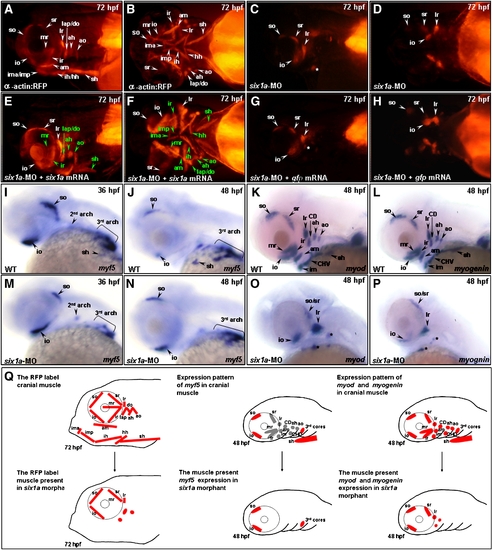
Six1a is required for the development of mr, ir, sh and all pharyngeal muscles. Embryos derived from the transgenic line Tg(α-actin:RFP) (A–H), all of whose skeletal muscles appear as red fluorescent protein (RFP), were injected with 8 ng of six1a-morpholino oligonucleotide (MO) to specifically inhibit six1a mRNA translation. RFP signal was detected only in the so, io, sr, lr, and remnant dorsal branchial arch muscle (white star) primordia in the six1a-MO-injected embryos (A vs. C and B vs. D). When embryos were injected with 8 ng of six1-MO together with 150 pg of six1a mRNA, results showed that the defective muscle primordia induced by six1a-MO were rescued and appeared as RFP-labeled muscles (E, F; the rescued muscles are marked in green typeface). In contrast, the rescue experiment failed when embryos were injected with 8 ng of six1a-MO with 200 pg of gfp mRNA (G, H), suggesting that the defects of six1a morphants were specific. The expressions of myf5 (I, J, M, N), myod (K, O), and myogenin (L, P) were also observed at the stages indicated. When wild-type embryos were injected with six1a-MO, myf5 was expressed normally in the six1a morphants, both at 36- (I vs. M) and at 48-hpf (J vs. N), except sh. On the other hand, the expressions of myod (K vs. O) and myogenin (L vs. P) were decreased in the extraocular io, so, sr and lr in the six1 morphants at 48 hpf. Weak signals of myod and myogenin were also noticed in the remnant dorsal branchial muscles (black stars) of six1a morphants. The schematic diagram illustrates the cranial muscle defects in six1a morphants and compares the expressions of myf5, myod and myogenin between wild-type (upper row, Q) and six1a morphants (lower row, Q). Lateral view: A, C, E, G and I–P; and ventral view: B, D, F and H. ah, adductor hyoideus; am, adductor mandibulae; ao, adductor operculi; do, dilator operculi; hh, hyohyoideus; ih, interhyoideus; ima, intermandibularis anterior; imp, intermandibularis posterior; io, inferior oblique; ir, inferior rectus; lap, levator arcus palatini; lr, lateral rectus; mr, medial rectus; sh, sternohyoideus; so, superior oblique and sr, superior rectus. EXPRESSION / LABELING:
PHENOTYPE:
|
|
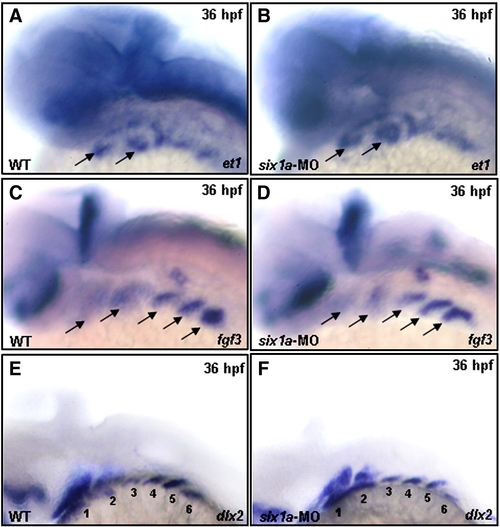
Loss of Six1a function does not impede the normal development of mesoderm core and pharyngeal pouch. The expression patterns of et1, fgf3 and dlx2 were examined in the wild-type and in the six1a-MO-injected embryos at 36 hpf. Results showed that the transcripts of et1, fgf3 and dlx2 exhibited similarly in ventral mesoderm cores (A vs. B, arrows), pharyngeal pouches (C vs. D, arrows), and neural crest cells (E vs. F, six arches), respectively, between wild-type embryos and six1a morphants. EXPRESSION / LABELING:
|
|
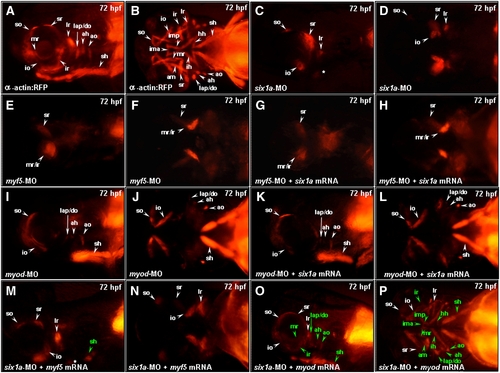
Injection of myod mRNA enables embryos to rescue the defective muscle derived from cranial mesoderm in the six1a morphants. Embryos derived from Tg (α-actin:RFP) (A, B as non-treated) were injected with 8 ng of six1a-MO (C, D) and either 4 ng of myf5-MO (E, F) or myod-MO (I, J) to serve as control groups. Embryos injected with 4 ng of myf5-MO and 150 pg of six1a mRNA (G, H), 4 ng of myod-MO and 150 pg of six1a mRNA (K, L), 8 ng of six1a-MO and 100 pg of myf5 mRNA (M, N), and 8 ng of six1a-MO and 50 pg of myod mRNA (O, P) were used to examine the appearance of RFP-labeled muscles. Results showed that only sr and mr/ir muscles exhibited in the myf5-MO-injected embryos and in the myf5-MO-six1a-mRNA-injected embryos (E vs. G and F vs. H). In contrast, only so, io, lap/do, ah, ao, and sh muscles exhibited in the myod-MO-injected embryos and in the myod-MO-six1a-mRNA-injected embryos (I vs. K and J vs. L). Similar to six1a morphants, embryos co-injected with six1a-MO and myf5 mRNA exhibited the so, io, sr, lr and remnant dorsal branchial arch muscles. However, injection of myf5 mRNA enabled embryos to rescue only sh primordia muscle among defects induced by six1a-MO (M, N), while injection of myod mRNA enabled embryos to rescue all the defective muscle primordia induced by six1a-MO (O, P). The rescued muscles are marked in green typeface. Lateral view: A, C, E, G, I, K, M and O; Ventral view: B, D, F, H, J, L, N and P. ah, adductor hyoideus; am, adductor mandibulae; ao, adductor operculi; do, dilator operculi; hh, hyohyoideus; ih, interhyoideus; ima, intermandibularis anterior; imp, intermandibularis posterior; io, inferior oblique; ir, inferior rectus; lap, levator arcus palatini; lr, lateral rectus; mr, medial rectus; sh, sternohyoideus; so, superior oblique and sr, superior rectus. EXPRESSION / LABELING:
|
|
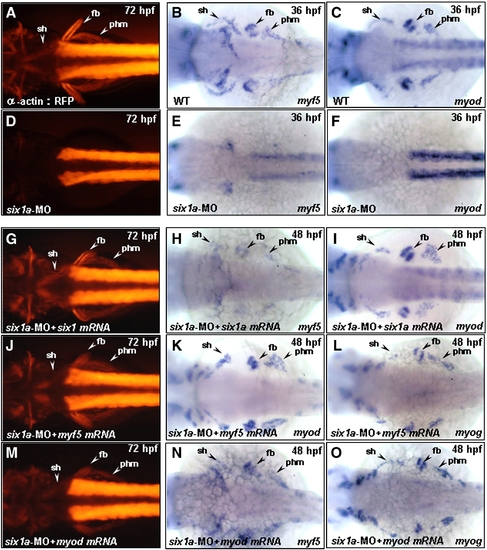
Injection of six1a-, myf5-and myod mRNA enables embryos to rescue trunk migratory head muscle sternohyoideus (sh) defect in six1a morphants. Dorsal views of embryos derived either from the transgenic line Tg(α-actin:RFP) (A, D, G, J, M) or from the wild-type (B, C, E, F, H, I, K, L). The RFP expression in the embryos derived from the transgenic line at 72 hpf (A) and the detection of myf5 and myod by whole-mount in situ hybridization at 36 hpf (B, C) served as control groups. Injection of embryos with either 8 ng of six1a-MO alone (D–F) or co-injection with 8 ng of six1a-MO and 150 pg of six1a mRNA (G–I), 100 pg of myf5 mRNA (J–L) or 50 pg of myod mRNA (M–O) were examined. RFP, myf5 and myod were not detected in sh, fb, or phm primordia in the six1a morphants (D–F); however, co-injection of six1a mRNA enabled embryos to rescue the defective expressions of RFP, myf5 and myod in sh, fb and phm primordia induced by six1a-MO at 48 hpf (H, I) and at 72 hpf (G). Meanwhile, injection of myf5 mRNA enabled embryos to rescue the defective expressions of RFP, myod and myogenin in sh, fb and phm primordia induced by six1a-MO at 48 hpf (K, L) and at 72 hpf (J). Injection of myod mRNA enabled embryos to rescue the defective expressions of RFP, myf5 and myogenin in sh, fb and phm primordia induced by six1a-MO at 48 hpf (N, O) and at 72 hpf (M). fb, fin bud; phm, posterior hypoaxial muscle; and sh, sternohyoideus. EXPRESSION / LABELING:
|
|
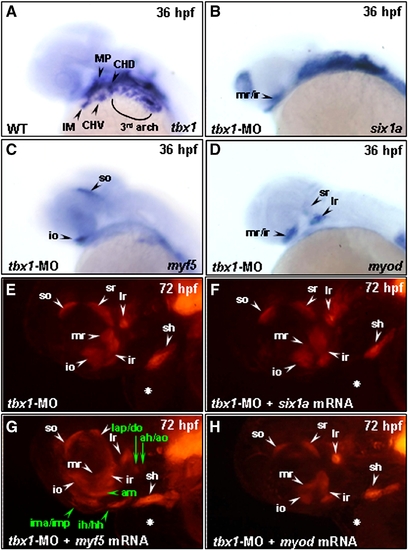
The function of Six1a in branchial muscle development requires Tbx1 and Myf5 to play a specification role on arch muscle cell fate. Embryos derived from the wild-type strain (A–D) and from the transgenic line Tg(α-actin:RFP) (E–H) were examined at lateral view. Whole-mount in situ hybridization was used to detect the tbx1 expression in arch muscle and cranial mesoderm in wild-type embryos at 36 hpf (A). Compared to the above control embryos, the expressions of six1a (B), myf5 (C) and myod (D) in the 10 ng group of tbx1-MO-injected embryos were lost in arch muscles, but retained in extraocular muscles. We also observed that all the pharyngeal arch muscles were lost, but that 6 extraocular muscles developed normally in the 10 ng group of tbx1-MO-injected embryos derived from Tg(α-actin:RFP) at 72 hpf (E). With co-injection of 10 ng of tbx1-MO and either 150 pg of six1a mRNA (F), 100 pg of myf5 mRNA (G) or 50 pg of myod mRNA (H) in embryos, we found that only myf5 mRNA enabled embryos to rescue the RFP expression in lap, do, ah, ao, am, ima/imp and ih/hh (marked in green typeface of G). The heart defect induced by injection of tbx1-MO is labeled with a white star. ah, adductor hyoideus; am, adductor mandibulae; ao, adductor operculi; do, dilator operculi; hh, hyohyoideus; ih, interhyoideus; ima, intermandibularis anterior; imp, intermandibularis posterior; io, inferior oblique; ir, inferior rectus; lap, levator arcus palatini; lr, lateral rectus; mr, medial rectus; sh, sternohyoideus; so, superior oblique and sr, superior rectus. Embryos were all lateral views. EXPRESSION / LABELING:
PHENOTYPE:
|
|
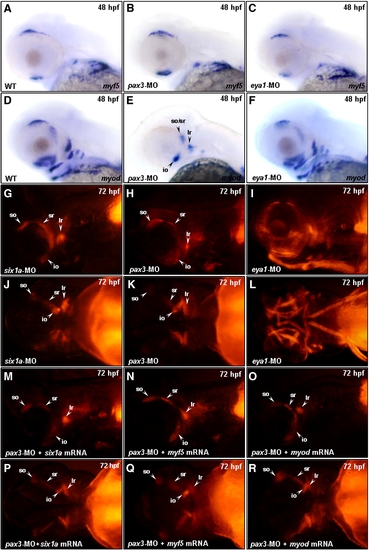
Similar to Six1a, Pax3 is required for the development of mr, ir and all arch muscles. Embryos derived from the wild-type strain (A–F) and from the transgenic line Tg(α-actin:RFP) (G–R) were examined. Whole-mount in situ hybridization was used to detect the expressions of myf5 and myod at 48 hpf in the wild-type embryos, which served as a control group (A, D). Compared to the control group, embryos injected with 3 ng of pax3-MO exhibited a normal expression of myf5 in head muscle primordia at 48 hpf (B), but these embryos expressed myod only in so/sr, lr, io, and some remnant branchial muscles (E). Embryos injected with 10 ng of eya1-MO expressed myf5 and myod normally in head muscle primordia at 48 hpf (C, F). We also noticed that the RFP expression of embryos injected with six1a-MO was similar to that of control group at 72 hpf (G vs. J). The RFP signal appeared in the so, io, sr, lr and some remnant branchial muscles in pax3-MO-injected embryos, which was similar to that of the six1a morphants at 72 hpf (G vs. H and J vs. K). The RFP signal appeared in all cranial muscles of the eya1-MO-injected embryos at 72 hpf (I and L). By co-injection of 10 ng of pax3-MO with 150 pg of six1a mRNA (M, P), 100 pg of myf5 mRNA (N, Q) or 50 pg of myod mRNA (O, R), we found that the defective expressions of six1a, myf5 and myod could not be rescued in the pax3 morphants. EXPRESSION / LABELING:
PHENOTYPE:
|
|
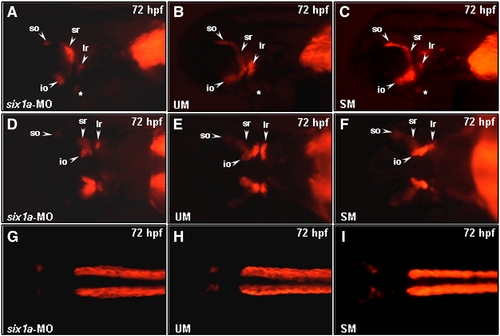
Comparison of the muscle defects induced by injection of six1a-MO, UM-MO and SM-MO into embryos. Embryos derived from the transgenic line Tg (α-actin:RFP) (A–I) were injected with either 8 ng of six1a-MO (A, D, G), 8 ng of UM-MO (B, E, H) or 8 ng of SM-MO (C, F, I) to specifically inhibit the translation of six1a mRNA. RFP signal was detected only in the so, io, sr, lr, and remnant dorsal branchial arch muscle (white star) primordia in the six1a-, UM-and SM-MO-injected embryos at 72 hpf (A–F), whereas RFP was not detected in the trunk migratory muscle primordia in the six1a-, UM-and SM-MO-injected embryos at 72 hpf (G–I). Lateral view: A–C; ventral view: D–E; dorsal view: G–I. io, inferior oblique; ir, inferior rectus; lr, lateral rectus; so, superior oblique and sr, superior rectus. |
|
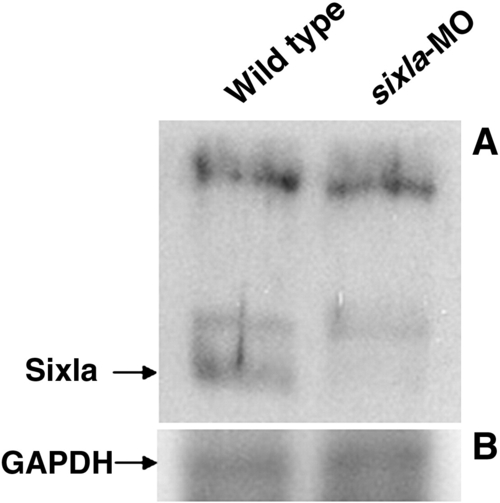
Western blot analysis to detect the decrease of Six1a protein level in six1a morphants. Total protein lysates were extracted separately from the wild-type embryos (left lane) and from the six1a-MO-injected embryos (right lane) at 48 hpf. After the total proteins were separated on a 12% SDS-PAGE, polyclonal antibodies of anti-Six1a (panel A) and anti-Glyceraldehyde 3-phosphate dehydrogenase (GAPDH; panel B) were carried out for Western blot analysis at 1:1000 dilution. The intensity of positive band for antibody against GAPDH served as a protein-loading control. |
|
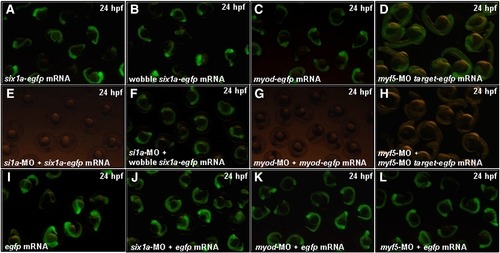
The confirmation of the specific activities of six1a-MO, myod-MO and myf5-MO in zebrafish embryos. Co-injection of six1a-MO (8 ng), myod-MO (4 ng) or myod-MO (4 ng) with its counterpart mRNA (100 pg) was employed into one-celled stage of zebrafish embryos. The Six1a-GFP fusion protein was detected at 24 hpf in embryos injected with a six1a-egfp mRNA (A). The Six1a-GFP fusion protein was not detected at 24 hpf in embryos co-injected with six1a-egfp mRNA and six1a-MO (E). The wobble Six1a-GFP fusion protein was detected at 24 hpf in embryos injected with a wobble six1a-egfp mRNA (B). The Six1a-GFP fusion protein was detected at 24 hpf in embryos co-injected with a wobble six1a-egfp mRNA and six1a-MO (F). The Myod-GFP fusion protein was detected at 24 hpf in embryos injected with myod-egfp mRNA (C). The Myod-GFP fusion protein was not detected in embryos co-injected with myod-egfp mRNA and myod-MO (G). Te GFP was detected in embryos injected with myf5-MO-targt-egfp mRNA at 24 hpf (D). The GFP signal was absent in embryos co-injected with myf5-MO-targt-egfp mRNA and myf5-MO (H). Embryos injected with egfp mRNA alone (I), co-injected with egfp mRNA and six1a-MO (J), egfp mRNA and myod-MO (K) and egfp mRNA and myf5-MO (L) served as control groups. |
|
The expression patterns of six1a, myf5 and myod in the migratory muscles of the trunk. The transcripts of six1a (A), myf5 (B), and myod (C) were all detected in the sternohyoideus (sh) primordia, fin bud (fb) and posterior hypaxial muscle (phm) that were derived from the anterior somites of zebrafish trunk at 36 hpf. |
|
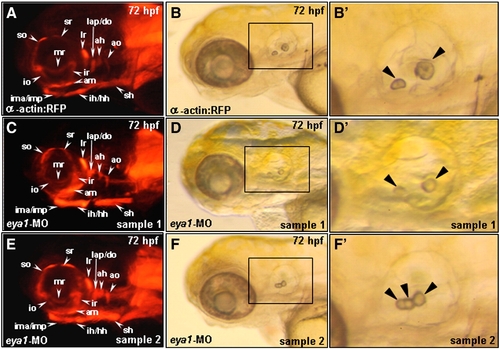
The defective phenotype of the eya1-MO-injected embryos at 72 hpf. Embryos derived from the from the transgenic line Tg(α-actin:RFP) (A-F) were examined. Compared to the control group (A, B, B′), embryos injected with 10 ng of eya1-MO exhibited a normal RFP expression in head muscle primordia at 72 hpf (C, E), an abnormal morphology of otic vesicle (D, F) and a reduced size of otolith (arrowhead in D′, F′). Panels B′, D′ and F′ are magnified from the indicated areas of B, D and F, respectively. |
|
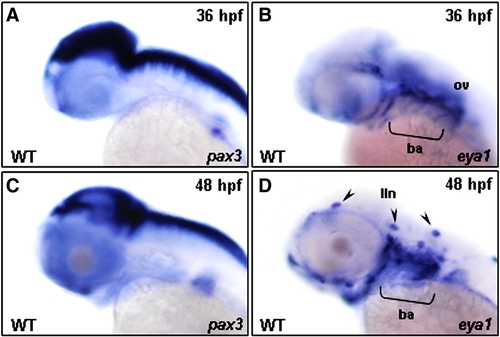
The expression patterns of pax3 and eya1 in the head region of zebrafish embryos. The temporal expressions of pax3 (A, C) and eya1 (B, D) transcripts in the zebrafish embryos were analyzed by whole-mount in situ hybridization from lateral view. The pax3 was neither expressed in the head muscle primordia at 36 (A) nor at 48 hpf (C), whereas the eya1 transcript was detected in the branchial arches (ba) and otic vesicle (ov) region at 36 hpf (B) and in lateral line neuromasts (lln) at 48 hpf (D). |
Reprinted from Developmental Biology, 331(2), Lin, C.Y., Chen, W.T., Lee, H.C., Yang, P.H., Yang, H.J., and Tsai, H.J., The transcription factor six1a plays an essential role in the craniofacial myogenesis of zebrafish, 152-166, Copyright (2009) with permission from Elsevier. Full text @ Dev. Biol.