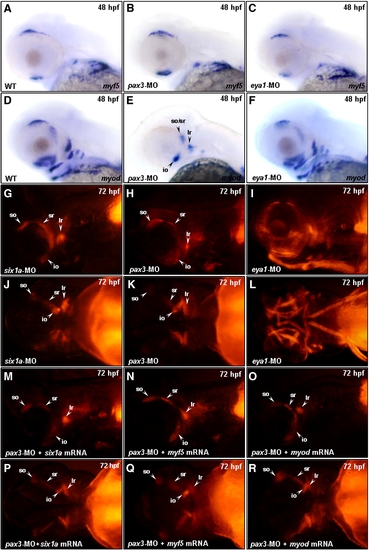
Similar to Six1a, Pax3 is required for the development of mr, ir and all arch muscles. Embryos derived from the wild-type strain (A–F) and from the transgenic line Tg(α-actin:RFP) (G–R) were examined. Whole-mount in situ hybridization was used to detect the expressions of myf5 and myod at 48 hpf in the wild-type embryos, which served as a control group (A, D). Compared to the control group, embryos injected with 3 ng of pax3-MO exhibited a normal expression of myf5 in head muscle primordia at 48 hpf (B), but these embryos expressed myod only in so/sr, lr, io, and some remnant branchial muscles (E). Embryos injected with 10 ng of eya1-MO expressed myf5 and myod normally in head muscle primordia at 48 hpf (C, F). We also noticed that the RFP expression of embryos injected with six1a-MO was similar to that of control group at 72 hpf (G vs. J). The RFP signal appeared in the so, io, sr, lr and some remnant branchial muscles in pax3-MO-injected embryos, which was similar to that of the six1a morphants at 72 hpf (G vs. H and J vs. K). The RFP signal appeared in all cranial muscles of the eya1-MO-injected embryos at 72 hpf (I and L). By co-injection of 10 ng of pax3-MO with 150 pg of six1a mRNA (M, P), 100 pg of myf5 mRNA (N, Q) or 50 pg of myod mRNA (O, R), we found that the defective expressions of six1a, myf5 and myod could not be rescued in the pax3 morphants.
|

