- Title
-
Multiple roles for Med12 in vertebrate endoderm development
- Authors
- Shin, C.H., Chung, W.S., Hong, S.K., Ober, E.A., Verkade, H., Field, H.A., Huisken, J., and Stainier, D.Y.
- Source
- Full text @ Dev. Biol.
|
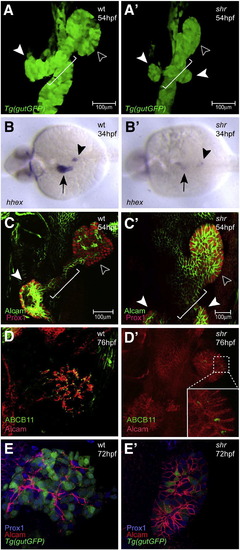
shr is required for liver and ventral pancreas development. Confocal projections of wild-type and shrs435 mutant embryos in the Tg(gutGFP)s854 line at 54 hpf (A, A′), hhex expression in wild-type and shrs435 mutant embryos at 34 hpf (B, B'), and confocal projections of Alcam (green) and Prox1 (red) expression at 54 hpf (C, C'), of Alcam (red) and ABCB11 (green) expression at 76 hpf (D, D'), and of Alcam (red) and Prox1 (blue) expression in the Tg(gutGFP)s854 line at 72 hpf (E, E′). (A, A′) The size of the liver (black arrowhead) and pancreas (white arrowhead) is often reduced in shrs435 mutants compared to wild-type. By 54 hpf, the formation of the hepatopancreatic duct (white bracket) is initiated in wild-type embryos, whereas shrs435 mutants exhibit an ambiguous duct morphology (white bracket) and two ventral pancreatic buds instead of one (white arrowheads). (B, B′) hhex is clearly expressed in the wild-type liver (black arrow) and pancreatic islet (black arrowhead) while its expression can be detected in only a few cells in the liver and pancreas regions of shrs435 mutants. (C, C′) Prox1 is expressed in the liver (black arrowhead) and pancreas (white arrowhead) in wild-type and shrs435 mutant embryos. At 54 hpf, Alcam has started to show restricted localization in wild-type liver, while it remains distributed along the entire surface of liver cells in shrs435 mutants. To better visualize hepatic Alcam expression in the mutants, a 1.2x magnified image is shown in panel C′. (D, D′) ABCB11 expression is almost absent in shrs435 mutants. To better visualize hepatic ABCB11 expression in the mutants, a magnified image is shown in an inset. (E, E') By 72 hpf, Alcam localizes on or near the apical side of hepatocytes in wild-type, while it remains distributed along the entire surface of liver cells in shrs435 mutants. All images except B, B′ are ventral views, anterior to the top; B, B′ are dorsal views, anterior to the left. EXPRESSION / LABELING:
PHENOTYPE:
|
|
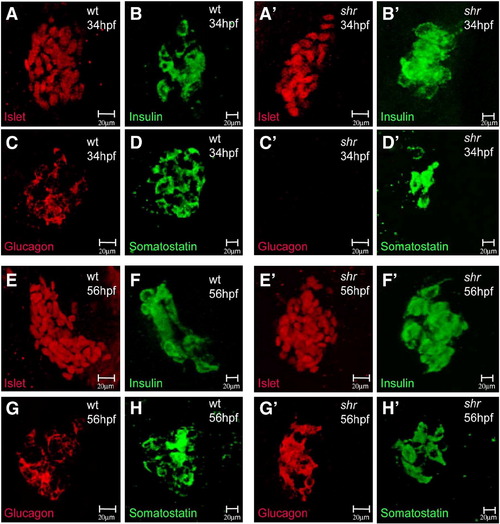
shr is required for dorsal pancreas development. Confocal projections of wild-type and shrs435 mutant embryos at 34 (A–D′) and 56 (E–H′) hpf stained for Islet (red, A, A′, E, E′), Insulin (green, B, B′, F, F′), Glucagon (red, C, C′, G, G′), and Somatostatin (green, D, D′, H, H′) expression. The pancreatic expression of Islet and Insulin appears unaffected in shrs435 mutants at 34 and 56 hpf (A′, B′, E′, F′). The expression of Somatostatin appears reduced (D′) and that of Glucagon almost completely absent (C′) in shrs435 mutants at 34 hpf, but restored to wild-type levels by 56 hpf (G′, H′). All images are ventral views, anterior to the top. |
|
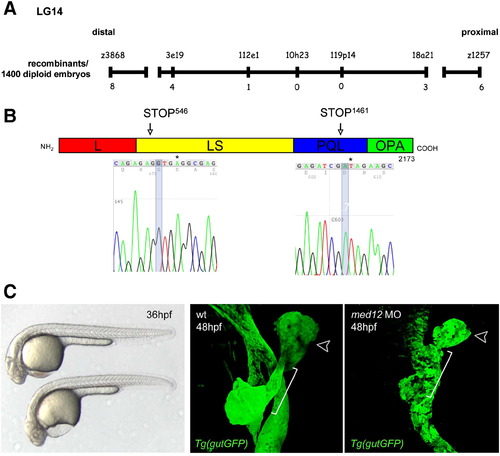
shr encodes Med12. (A) Genetic map of the shr region on chromosome 14. Numbers below the SSLP markers indicate the number of recombination events in 1400 diploid embryos tested. (B) Sequencing of two shr mutant alleles reveals the molecular lesions (leading to the premature truncation of Med12). L, leucine-rich domain; LS, leucine-serine-rich domain; PQL, proline-glutamine-leucine-rich domain; OPA, opposite-paired domain. Numbering corresponds to the amino acid number. (C) Knock-down of med12 by the injection of 2 ng of a splice-site MO into the Tg(gutGFP)s854 line recapitulates the shr mutant phenotypes at 36 (bright field; lateral view) and 48 hpf (confocal projections of Tg(gutGFP)s854 line (green), black arrowhead points to the liver and white bracket indicates the hepatopancreatic duct; ventral views, anterior to the top). |
|
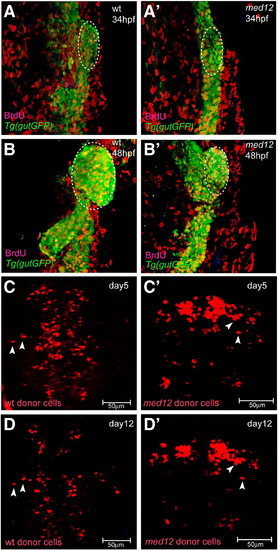
Med12 does not appear to be essential for embryonic cell proliferation or viability. (A–B′) Confocal projections of wild-type and med12s435 mutant embryos in the Tg(gutGFP)s854 line (green) at 34 (A, A′) and 48 (B, B′) hpf in conjunction with BrdU (red) antibody staining. Yellow cells are endodermal cells (green) with BrdU (red) incorporation and white dashed lines surround the area used to count BrdU-positive cells. The overall level of endodermal BrdU incorporation in wild-type and med12s435mutant embryos appears similar (Table 1). (C–D′) Confocal projections of wild-type host embryos transplanted with wild-type (C, D) or med12s435 mutant (C′, D′) donor cells, visualized at days 5 (C, C′) and 12 (D, D′). Most wild-type and med12s435mutant donor cells survive until at least day 12 (D, D′). White arrowheads point to individual rhodamine dextran-labeled donor cells. The same embryo is shown in panels C and D, and another in panels C′ and D′. Images A–B′ are ventral views, anterior to the top; C–D′ are dorsal views, anterior to the top. |
|
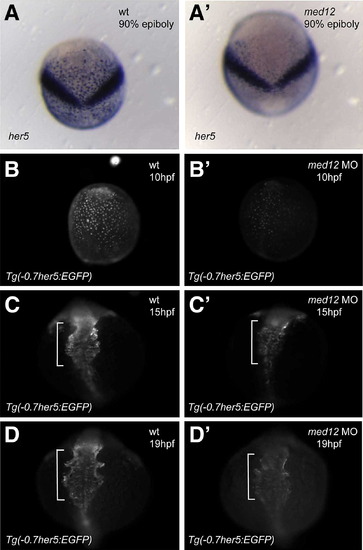
Med12 regulates her5 expression in endodermal cells during gastrulation and somitogenesis. (A, A′) In situ hybridization at 90% epiboly shows specific reduction of her5 expression in endodermal cells in med12s432 mutants compared to wild-type siblings. The V shaped stripe represents the domain of her5 expression in the midbrain–hindbrain boundary. (B–D′) Injection of med12 MO into the Tg(-0.7her5:EGFP)ne2067 line caused decreased GFP expression. Note that the Tg(-0.7her5:EGFP)ne2067 line shows GFP expression in the pharyngeal endoderm as well as the organ-forming region during late somitogenesis stages (C, D). White bracket in C–D′ indicates pharyngeal region. Exposure time for imaging B, B′ was 300 ms; C, C′ was 250 ms; D, D′ was 250 ms. Dorsal views, anterior to the top. EXPRESSION / LABELING:
|
|
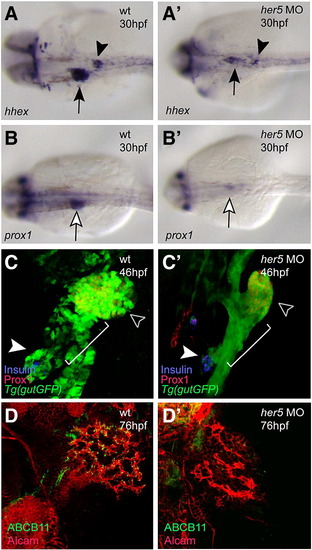
Her5 is required for the development of the endodermal organs. Wild-type and her5 MO-injected animals at 30 (A–B′), 46 (C, C′), and 76 (D, D′) hpf were analyzed for hhex (A, A′) and prox1 (B, B′) expression, confocal projections in the Tg(gutGFP)s854 line in conjunction with Prox1 (red) and Insulin (blue) expression (C, C′), and Alcam (red) and ABCB11 (green) expression (D, D′). (A, A′) hhex expression in the liver (black arrow) and pancreatic islet (black arrowhead) regions could be detected in only a few cells in her5 MO-injected embryos. (B, B′) prox1 expression in the liver region (white arrow) could only be detected at low levels in her5 MO-injected embryos. (C, C′) At 46 hpf, the formation of the hepatopancreatic duct (white bracket) is initiated in wild-type embryos, whereas in her5 MO-injected embryos, this process was perturbed, resulting in an ambiguous ductal morphology. Note the lack of engulfment of the Insulin-positive endocrine cells (white arrowhead) by the ventral pancreatic bud derived tissue in MO-injected embryos. Black arrowhead points to the liver. (D, D′) ABCB11 expression was almost absent in her5 MO-injected larvae. n = 20 in each set of MO-injections; the percentage of MO-injected embryos/larvae exhibiting the phenotype shown in panel C′ was 60% (n = 12/20), and the one in panel D′ was 60% (n = 12/20). Images A–B′ are dorsal views, anterior to the left; C–D′ are ventral views, anterior to the top. EXPRESSION / LABELING:
PHENOTYPE:
|
|
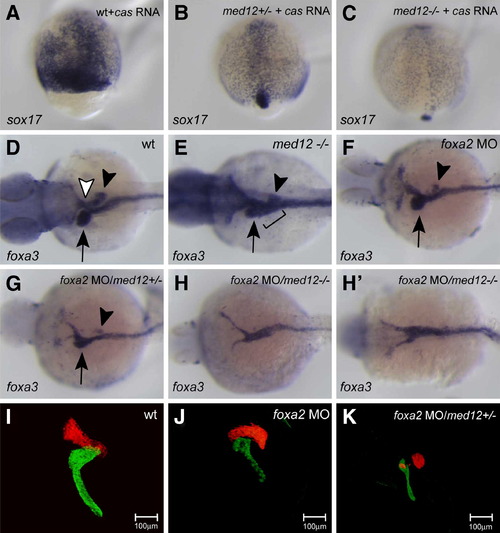
Med12 functions with Cas/Sox32 and Foxa2 to regulate endodermal organ development. (A–C) Embryos from med12s432 heterozygote incrosses injected with cas mRNA were harvested at 90% epiboly and analyzed for sox17 expression. med12s432 homozygous mutant embryos overexpressing cas mRNA showed decreased induction of sox17 expression (C) compared to med12 heterozygous (B) and wild-type (A) embryos. (D–H′) Embryos from wild-type crosses and med12s432 heterozygote incrosses injected with foxa2 MO were harvested at 48 hpf and analyzed for foxa3 expression. Wild-type foxa3 expression reveals structures such as the liver (black arrow), pancreas (black arrowhead), and swim bladder (white arrowhead) (D). med12s432 mutants show an ambiguous ductal morphology (black bracket) and defective development of endodermal organs (E). foxa2 MO-injected embryos showed slight perturbation of organ development (F), while med12s432 heterozygous embryos injected with foxa2 MO showed a more severe malformation of organs (G), and med12s432 homozygous mutant embryos injected with foxa2 MO showed an almost complete absence of accessory organs, exhibiting a solid endodermal rod (H, H′). (I–K) Confocal projections of larvae from med12s432 heterozygote outcrosses in the Tg (fabp1a:dsRed, elastase:GFP; insulin:dsRed) heterozygote line injected with foxa2 MO (J and K) and analyzed at 96 hpf. While foxa2 MO-injected larvae showed slight perturbation of liver and exocrine pancreas (J), med12s432 heterozygous larvae with foxa2 MO injection showed a more severe malformation of these organs (K). Images A–C are dorsal views, anterior to the top; D–H′ are dorsal views, anterior to the left; I–K are ventral views, anterior to the top. |
|
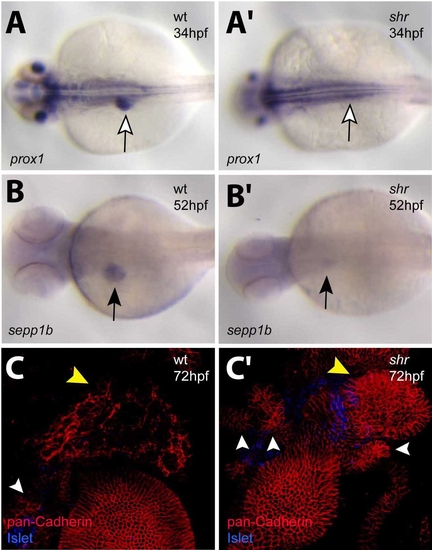
shr is required for liver development. Wild-type and shrs435 mutant animals at 34 (A, A′), 52 (B, B′), and 72 (C, C′) hpf were analyzed for hhex (A, A′) and sepp1b (B, B′) expression and confocal projections of pan-Cadherin (red) and Islet (blue) expression (C, C′). (A, A′) prox1 expression is evident in the liver (white arrow) of wild-type embryos but barely detectable in the liver region of shrs435 mutants. (B, B′) selenoprotein P, plasma, 1b (sepp1b) expression is detected at low levels in shrs435 mutants (black arrow). (C, C′) shrs435 mutants exhibit defective differentiation of hepatocytes as assessed by Cadherin immunostaining. Note the multiple, unfused ventral pancreatic buds in mutants (white arrowheads). Yellow arrow points to the liver. A–B′ are dorsal views, anterior to the left; C, C′ are ventral views, anterior to the top. EXPRESSION / LABELING:
PHENOTYPE:
|
|
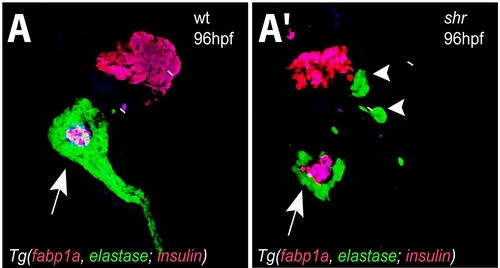
shr is required for ventral pancreas development. Confocal projections of wild-type and shrs435 mutant larvae in the Tg(fabp1a:dsRed, elastase:GFP; insulin:dsRed) line at 96 hpf. (A, A′) shrs435 mutant larvae show multiple Tg(elastase: GFP)-positive ventral pancreatic buds (white arrowheads), and defective elongation of the ventral pancreatic tail (white arrow). Despite defective hepatic differentiation, a population of liver cells expresses DsRed in the mutant larvae. All images are ventral views, anterior to the top. |
|
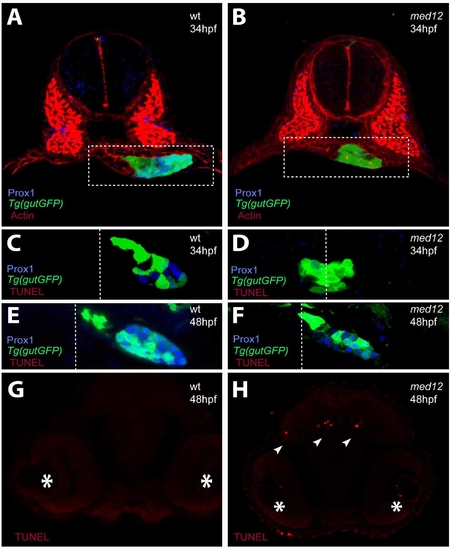
Analysis of apoptosis in med12s435 mutant embryos. (A, A′) Confocal projections of Prox1 (blue) and Actin (red) expression in wild-type and med12s435 mutants in the Tg(gutGFP)s854 line (green) at 34 hpf. White dashed lines surround the endodermal region. (C–F) Confocal projections of wild-type and med12s435 mutants in the Tg(gutGFP)s854 line (green) at 34 (C, D) and 48 (E, F) hpf in conjunction with anti-Prox1 (blue) antibody and TUNEL (red) staining. White dashed lines indicate the midline. In med12s435 mutants, Prox1 expression is mostly missing at 34 hpf but present at 48 hpf. Little if any TUNEL staining was observed in the endodermal tissues at these stages. (G–H) Confocal projections of TUNEL staining in the brain region of wild-type and med12s435 mutants. Asterisks indicate the eyes and arrowheads point to TUNEL-positive cells. Transverse sections, dorsal to the top. |
|
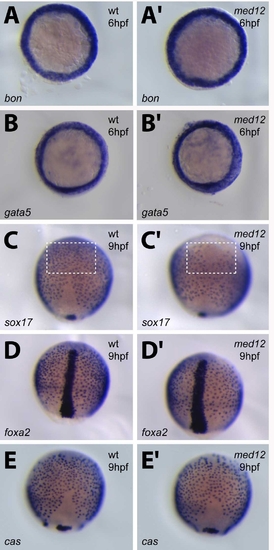
Med12 regulates anterior sox17 expression in endodermal cells during gastrulation. Wild-type and med12s432 mutants were analyzed at 60% (A–B′) and 90% (C–E′) epiboly for bon (A, A′), gata5 (B, B′), sox17 (C, C′), foxa2 (D, D′), and cas (E, E′) expression. White dashed lines in C and C′ surround a subset of sox17-positive cells in the anterior region. Note that, in med12s432 mutants, many of these cells are missing, whereas other early endodermal markers show comparable expression in wild-type and mutants. After in situ hybridization, the embryos were genotyped to unambiguously identify the mutants. A–B′ are animal pole views; C–E′ are dorsal views, anterior to the top. EXPRESSION / LABELING:
PHENOTYPE:
|
|
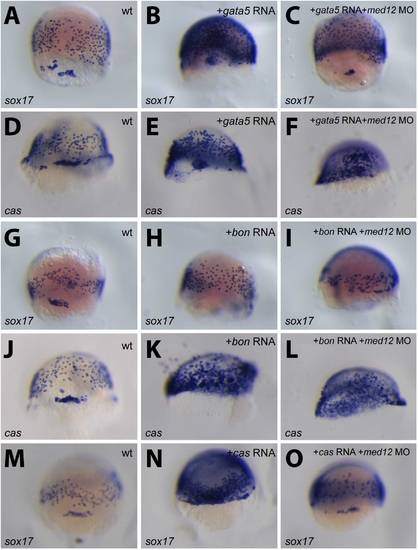
Med12 appears to regulate sox17 expression. Wild-type, and bon, cas/sox32, or gata5 mRNA-injected embryos with or without med12 MO were analyzed between 70 and 80% epiboly for sox17 (A, B, C, G, H, I, M, N, O) or cas/sox32 (D, E, F, J, K, L) expression. The ability of Gata5, Bon or Cas to up-regulate sox17 expression (B, H, N) appears to be affected by the injection of med12 MO (C, I, O). However, cas expression in these overexpression assays (E, K) appears not to be affected by med12 MO injections (F, L). All images are dorsal views, anterior to the top. EXPRESSION / LABELING:
|

Unillustrated author statements PHENOTYPE:
|
Reprinted from Developmental Biology, 317(2), Shin, C.H., Chung, W.S., Hong, S.K., Ober, E.A., Verkade, H., Field, H.A., Huisken, J., and Stainier, D.Y., Multiple roles for Med12 in vertebrate endoderm development, 467-479, Copyright (2008) with permission from Elsevier. Full text @ Dev. Biol.