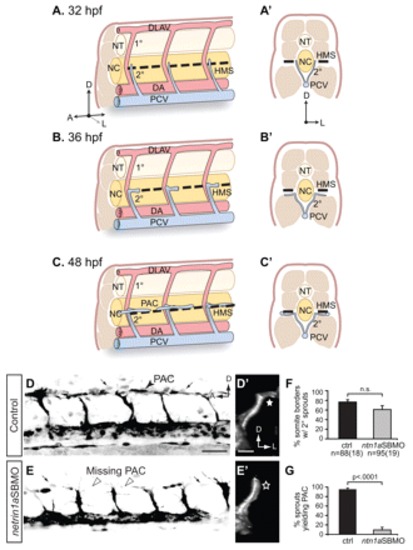
netrin 1a is required for turning of secondary sprouts at the horizontal myoseptum. (A-C2) Lateral (A-C) and corresponding transverse views (A2-C2) of parachordal chain (PAC) formation, showing dorsal aorta (DA, red), posterior cardinal vein (PCV, blue), horizontal myoseptum (HMS, dashed line), primary intersomitic vessels (1°, red) and secondary sprouts (2°, blue). DA and primary sprouts omitted for clarity. (A,A2) Secondary sprouts emerge from the PCV at <32 hpf and grow dorsally to the HMS. (B,B2) Dorsal growth stops at the HMS and sprouts turn and grow laterally. (C2) Lateral growth stops at the lateral-most aspect of the muscle, and the sprout then turns anteriorly and posteriorly to form the PAC. (D-E2) Removing netrin 1a function in 48 hpf plcg1y10; fli1a:egfpy1 zebrafish embryos. (D,E) Lateral views, reversed contrast. (D2,E2) Transverse view volume renderings of single hemisegments from D,E. (D,D2) In uninjected controls, secondary sprouts grow from the PCV and turn at the HMS (star, D2) to form the PAC (arrowhead, D). The fli1a:egfpy1 transgene also labels some dimly fluorescent non-endothelial cells near the HMS (small arrows, D,E). (E,E2) In netrin 1a morphants, secondary sprouts usually fail to turn laterally (star, E2) and form the PAC (arrowheads, E). (F,G) Secondary sprouts were quantified in five hemisegments per embryo in uninjected controls and netrin 1a morphants. (F) The fraction of intersomitic boundaries bearing secondary sprouts is not significantly changed in morphants. Uninjected controls, 75±5%; netrin 1a morphants, 61±8%. (G) Fewer secondary sprouts turn to form the PAC in morphants. Uninjected controls, 95±3%; netrin 1a morphants, 9±5%. All values are mean±s.e.m.; error bars show s.e.m.; P-value determined by Mann-Whitney U test; n.s., not significant; n, number of hemisegments (number of embryos). A, anterior; D, dorsal; L, lateral; DLAV, dorsal longitudinal anastomotic vessel; NT, neural tube; NC, notochord; 1°, primary sprouts; 2°, secondary sprouts. Scale bars: 50 μm.
|

