- Title
-
Modulation of Agrin and RhoA Pathways Ameliorates Movement Defects and Synapse Morphology in MYO9A-Depleted Zebrafish
- Authors
- O'Connor, E., Cairns, G., Spendiff, S., Burns, D., Hettwer, S., Mäder, A., Müller, J., Horvath, R., Slater, C., Roos, A., Lochmüller, H.
- Source
- Full text @ Cells
|
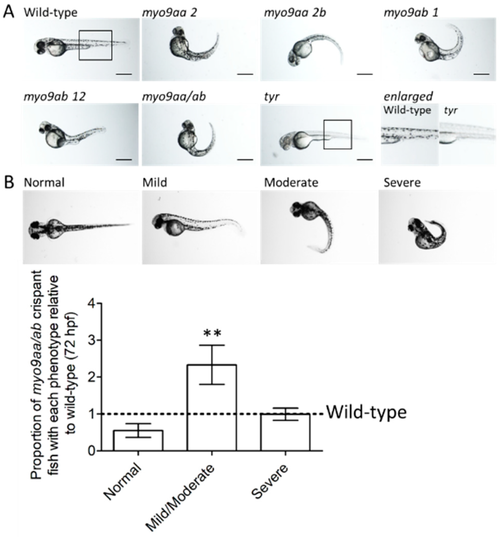
Phenotypes of myo9aa/ab crispant zebrafish. (A) Representative images of 48 hpf zebrafish injected with sgRNAs against myo9aa and myo9ab (numbers following the gene refer to exon targeted). Injection of a combination of all four sgRNAs was performed (myo9aa/ab), along with a control against the tyrosinase gene (tyr) in which no pigment can be observed, confirming knockdown success. Scale bar = 500 µm. (B) Images showing phenotypic classification of injected zebrafish at 72 hours post fertilisation (hpf), ranging from normal to severe. Graph shows proportion of myo9aa/ab crispant fish injected with all four sgRNAs (n = 7, including 160 fish in total) as compared to wild-types (black line, n = 7, including 138 fish in total). Error bars represent mean ± S.E.M, ** p < 0.01, paired t-test. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |
|
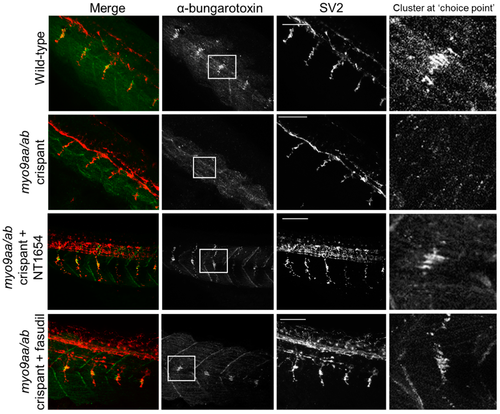
Neuromuscular junction (NMJ) morphology of myo9aa/abcrispant zebrafish at 24 hpf. Representative images of NMJs in wildtype, myo9aa/ab crispant, and NT1654 (0.15 ng) or fasudil-treated (10 µm) crispant zebrafish at 24 hpf. Acetylcholine receptors stained with αBTx (green), and motor neurons detected with an antibody against SV2 (red). White boxes demark areas enlarged in the right-hand panel, showing presence of αBTx-positive choice point clusters. Scale bar = 50 µm. |
|
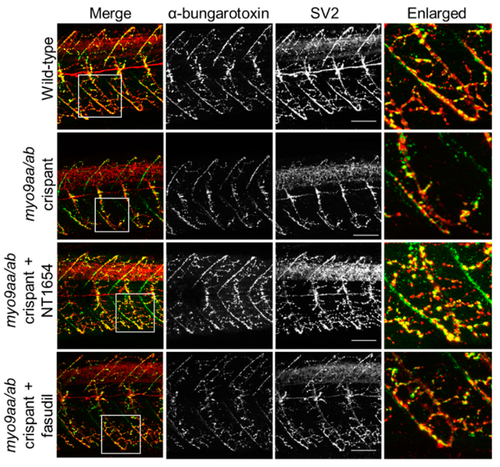
NMJ morphology of myo9aa/ab crispant zebrafish at 48 hpf. Representative images of NMJs in wild-type, myo9aa/ab crispant, and NT1654 (0.15 ng) or fasudil-treated (10 µm) crispant zebrafish at 48 hpf. Acetylcholine receptors stained with αBTx (green), and motor neurons detected with an antibody against SV2 (red). White boxes demark areas enlarged in the right-hand panel. Scale bar = 50 µm. PHENOTYPE:
|
|
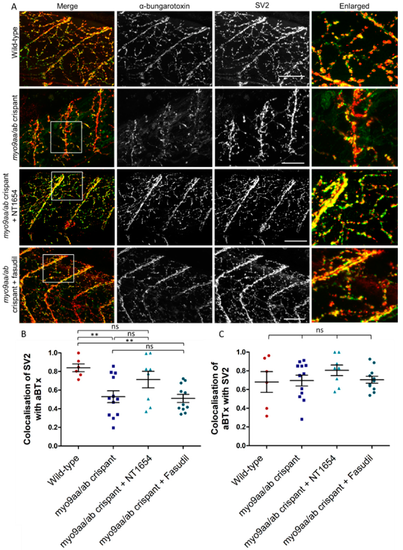
NMJ morphology of myo9aa/ab crispant zebrafish at 120 hpf. (A) Representative images of NMJs in wild-type, myo9aa/ab crispant, and NT1654 (0.15 ng) or fasudil-treated (10 µm) crispant zebrafish at 120 hpf. Acetylcholine receptors stained with αBTx (green), and motor neurons detected with an antibody against SV2 (red). White boxes demark areas enlarged in the right-hand panel. Scale bar = 50 µm. (B) Colocalisation of SV2-positive signal with αBTx and (C) colocalisation of αBTx with SV2-positive signal using Mander’s correlation coefficient (0 = no colocalisation, 1 = full colocalisation). Wildtype (n = 6 fish), myo9aa/ab crispant (n = 12 fish), crispant treated with NT1654 (0.15 ng, n = 8 fish), and crispant treated with fasudil (10 µm, n = 11 fish) were subject to analysis. Error bars = mean ± S.E.M. ** p ≤ 0.01 and ns = not significant, One-way ANOVA. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|