- Title
-
Exon- and contraction-dependent functions of titin in sarcomere assembly
- Authors
- Shih, Y.H., Dvornikov, A.V., Zhu, P., Ma, X., Kim, M.J., Ding, Y., Xu, X.
- Source
- Full text @ Development

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |
|
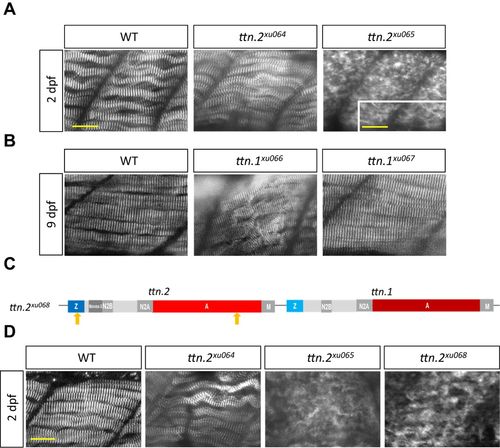
Exon-dependent functions of titin in sarcomere assembly in somites. (A) ttn.2xu065, but not ttn.2xu064, exhibits disrupted Z-disk formation in somites at 2 dpf. Shown are whole-mount immunostaining for α-Actinin. The inset is a magnified image showing residual striated structure in ttn.2xu065. (B) ttn.1xu066, but not ttn.1xu067, exhibits sarcomere disarray in somites at 9 dpf. (C) Schematic representation of the ttn.2 cis double mutant ttn.2xu068. Arrows indicate locations of mutations. (D) Immunostaining for α-Actinin in somites at 2 dpf indicates that the Z-disk structure in ttn.2xu065 and ttn.2xu068 mutants is disrupted to the same extent. Scale bars: 20 μm and 8 μm (inset in A). |
|
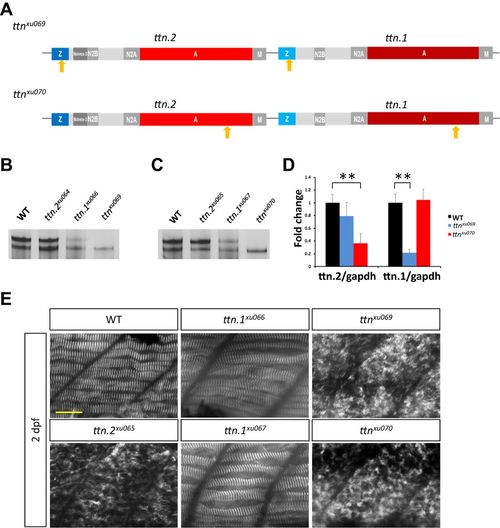
Mutants ttnxu69 and ttnxu070 overcome protein compensation and exhibit more severe phenotypes. (A) Schematic representation of two double mutants, ttnxu069 and ttnxu070. Arrows indicate locations of mutations. (B) Protein extracts from 2 dpf larvae of ttnxu69 mutants analyzed by 2% SDS-agarose gel and silver staining. (C) Protein analysis of ttnxu070. (D) ttn.2 and ttn.1 mRNA expression levels in ttnxu070 and ttnxu69 mutants, as revealed by qPCR using primers targeting ttn.2 and ttn.1, respectively. Means±s.d., N=5. **P<0.01. (E) Immunostaining of 2 dpf embryos for α-Actinin. Scale bar: 20 μm. |
|
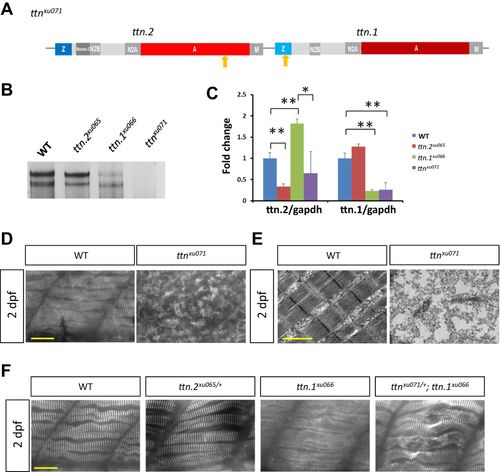
Generation of a ttn.2/1 near-null mutant by targeting two highly used exons. (A) Schematic representation of the ttnxu071 mutant. Arrows indicate locations of mutations. (B) Analysis of protein extracts from 2 dpf WT larvae and ttn.2xu065, ttn.1xu066 and ttnxu071 mutants. (C) qPCR with primers targeting ttn.2 and ttn.1, respectively. Means±s.d., N=5. *P<0.05, ** P<0.01. (D) The Z-disk structure marked by α-Actinin was disrupted in the ttnxu071 mutant. (E) Electron micrographs of somites of 2 dpf WT and ttnxu071 mutant larvae. (F) Z-disk structure was normal in ttn.2xu065/+ and ttn.1xu066 at 2 dpf, but sarcomere disarray was noted in ttnxu071/+ and ttn.1xu066. Scale bars: 20 μm (D,F) and 1 µm (E). |
|
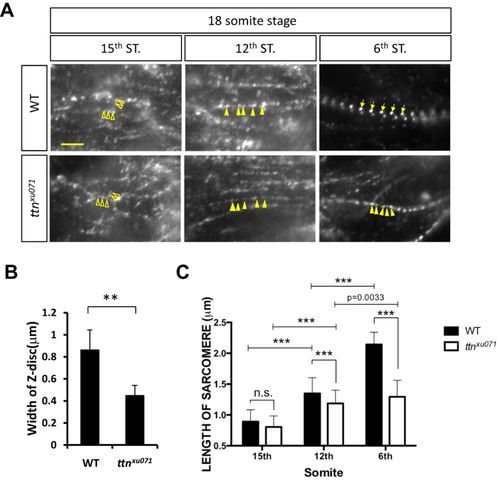
Phenotypes of de novo sarcomere assembly in the ttn.2/1-null mutant. (A) WT or ttnxu071 mutant embryos at the 18-somite stage were stained for α-Actinin. The images were taken at the 6th, 12th and 15th somites (ST). Scale bar: 5 μm. Z-bodies were scattered as irregular dots (open arrowheads) at the 15th somites and became periodic and aligned longitudinally (solid arrowheads) at the 12th somite. In the 6th somite, Z-bodies underwent lateral growth to form striated Z-disks (arrows) in WT embryos but not in the ttnxu071 mutant (solid arrowheads). (B) The width of the Z-disc of WT and ttnxu071 mutants at the 6th somite was quantified. Means±s.d. N=90 for each group from six embryos. **P<0.01. (C) The length of sarcomeres, marked by the distance between two adjacent Z-bodies or Z-disks, in 6th, 12th and 15th somites of WT and ttnxu071 mutant embryos. The length of sarcomeres did not increase at the 6th somite in the ttnxu071 mutant compared with WT, where sarcomere length increased to 2.2 µm. Means±s.d. ***P<0.00001. N=80 for each group in different somites from six embryos. n.s., not significant (P>0.05). EXPRESSION / LABELING:
PHENOTYPE:
|
|
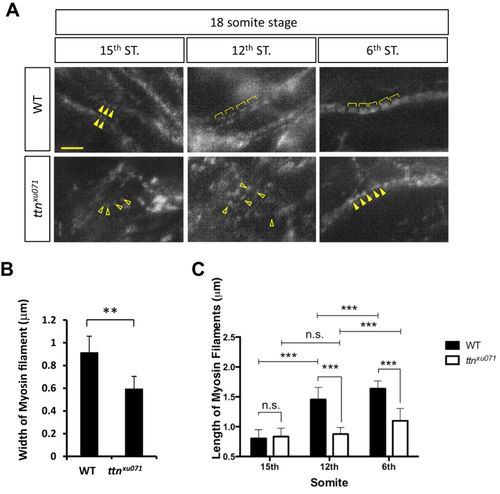
Phenotypes of thick filament assembly in the ttn.2/1-null mutant. (A) Immunostaining was performed in WT or ttnxu071 mutant embryos at the 18-somite stage with an F59 antibody against the myosin heavy chain. Scale bar: 5 μm. Myosin filaments were aligned longitudinally (solid arrowheads) at the 15th somite in WT embryos but remain scattered in ttnxu071 (open arrowheads). Expansion of thick filaments is seen in WT embryos at the 12th and 6th somites (brackets). However, the thick filaments in the ttnxu071 mutant remain scattered at the 12th somite (open arrowheads) and become aligned at the 6th somite (solid arrowheads). (B) The width of the thick (myosin) filaments of WT and ttnxu071 mutants at the 6th somite was quantified. Means±s.d., N=84 for each group from six embryos. **P<0.01. (C) The length of myosin filaments in the 6th, 12th and 15th somites of WT and ttnxu071 mutant embryos. The length of thick filaments does not increase in the ttnxu071 mutant, whereas thick filament length increases to 1.6 µm in WT. Means±s.d., ***P<0.00001. N=80 for each group in different somites from six embryos. n.s., not significant (P>0.05). EXPRESSION / LABELING:
PHENOTYPE:
|
|
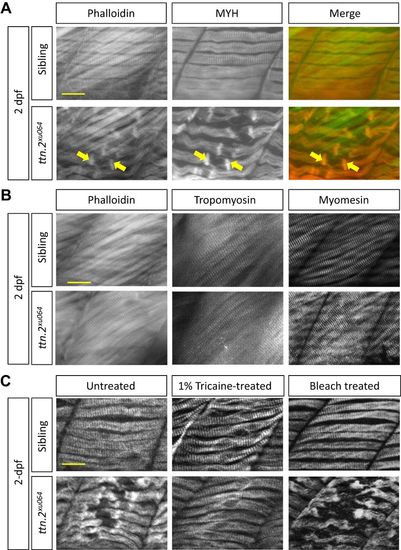
Contraction-dependent myofibril damage to the slow muscle of ttn.2xu064. (A) Slow muscle defects in ttn.2xu064 embryos. Immunostaining for Myosin and Phalloidin staining to label Actin filaments in 2 dpf embryos. Arrows indicate the two rod-like structures enclosing the damaged myofibril region. (B) Fast muscle is intact in ttn.2xu064. Immunostaining with antibodies against Tropomyosin and Myomesin or staining with Phalloidin in 2 dpf embryos. (C) ttn.2xu064 mutants and siblings treated with 1% tricaine for 24 h or 0.07% bleach at 1 dpf for 5 min were fixed and immunostained for Myosin at 2 dpf. Scale bars: 5 μm. |
|
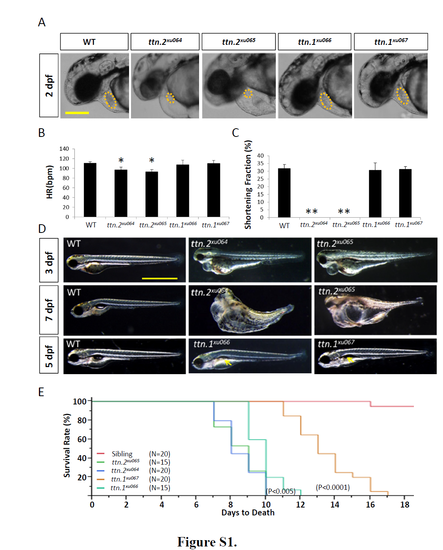
Phenotypes of ttn.2 and ttn.1 mutants. A, Cardiac edema with decreased ventricle size (dashed circles) was noted in both ttn.2xu064 and ttn.2xu065, but not in ttn.1xu066 and ttn.1xu067. Shown are bright-field images at 2 dpf. Scale bar, 0.05 mm. B and C, In ttn.2xu064 and ttn.2xu065, the heart rate was slightly decreased (B), and the ventricular shortening fraction was reduced to zero (C). * and ** indicates p <0.05 and p <0.01, respectively . Means ±S.D. N=5. D, Bright-field images demonstrated that the cardiac edema was still present in both ttn.2xu064 and ttn.2xu065 at 3 dpf, but muscle disarray was more obvious in ttn.2xu065. Both ttn.2 mutants had whole-body edema at 7 dpf. ttn.1xu066 and ttn.1xu067 showed deflation of the swim bladder at 5 dpf (arrows). Scale bar, 1 mm. E, ttn.2xu064 and ttn.2xu065 mutants started to die at 7 dpf and could survive up to 10 dpf; ttn.1xu066 could survive until 12 dpf; and ttn.1xu067 started to die at 11 dpf and could live up to 17 dpf. PHENOTYPE:
|
|
The Assembly of Titin Into Premyofibrils. Titin isoforms were assembled into premyofibrils and became striated at the 12th somite of an 18- somite embryo, as indicated by immunostaining using MIR, an anti-Titin antibody. Loss of Titin in somites in the ttnxu071 mutant has been seen. Scale bar, 20 µm. 18 somite stage |