- Title
-
Extraocular ectoderm triggers dorsal retinal fate during optic vesicle evagination in zebrafish
- Authors
- Kruse-Bend, R., Rosenthal, J., Quist, T.S., Veien, E.S., Fuhrmann, S., Dorsky, R.I., and Chien, C.B.
- Source
- Full text @ Dev. Biol.
|
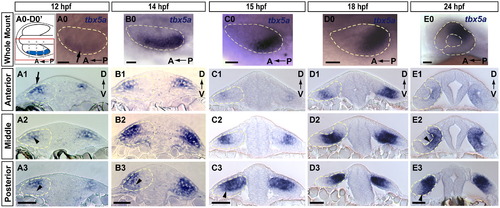
Expression domain of prospective dorsal marker tbx5a is broad and dynamic. Expression of tbx5a in the developing retina at (A) 12, (B) 14, (C) 15, (D) 18 and (E) 24 hpf (blue). (0 whole mount; 1–3, transverse sections). (A0–D0 dorsal views; E0, lateral view). (A0–D0′) Schematic diagram of orientation for A0–D0 whole-mount dorsal view images. Each image shows a single optic vesicle (red box in schematic), with the midline oriented toward the top. Dashed gray lines show approximate location of anterior, middle, and posterior sections in 1–3. (A) tbx5a mRNA is expressed in dorsolateral optic vesicle (arrow in A0, arrowhead in A2) at 12 hpf, and is absent from the ventral leaflet (arrowhead in A3). Extraocular ectoderm is directly overlying this portion of the optic vesicle (arrow in A1). (B) At 14 hpf expression extends to the ventral leaflet in posterior sections (arrowhead in B3). (C and D) tbx5a domain moves posteriorly from 15–18 hpf and resides primarily in the dorsal (prospective retinal) leaflet (arrowhead in C3). (E) tbx5a expression still covers much of the eye at 24 hpf, including prospective dorsal pole, central retinal domain medial to the lens (arrowhead in E2), and extending to the ventral domain (arrowhead in E3). Scale bars=50 μM. Dashed yellow lines outline optic vesicles in all images, also lens and choroid fissure in E0. |
|
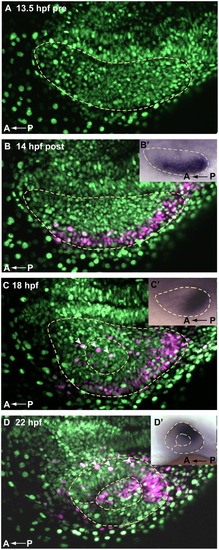
Cells of the lateral optic vesicle give rise to prospective dorsal and central retina. A Kaede fate map of 14 hpf lateral retina shows that cell movements generally recapitulate movements of tbx5a expression, indicating that dorsal fate initiation could be a discrete early event. (A–D) Dorsal view, maximum intensity projections of single optic vesicles, oriented as shown in the schematic (Fig. 1 A0–D0′) with the midline toward the top. Dashed yellow lines outline optic vesicles (A–D′), lenses (C–D′), and choroid fissure (D and D′). (A) Pre-activation, ubiquitous NLS green Kaede, 13.5 hpf. (B) 14 hpf, immediately after photoactivation, cells of lateral optic vesicle express photoactivated Kaede (magenta). (B′) 14 hpf whole mount tbx5a expression, dorsal view (C) 18 hpf, photoactivated Kaede cells from lateral optic vesicle have moved posteriorly. (C′) 18 hpf whole mount tbx5a expression, dorsal view (D) 22 hpf, photoactivated magenta cells reside primarily in the dorsal and central retinal domains, as well as in the lens. (D′) 24 hpf whole mount tbx5a expression, lateral view. Movements of photocleaved Kaede 14 hpf lateral retinal cells are generally consistent with the pattern of movement of tbx5a—compare magenta domains in A–D to A′–D′ insets and to Fig. 1 sections. White arrowheads in C and D mark extraocular ectodermal cells overlying the retina and lens. |
|
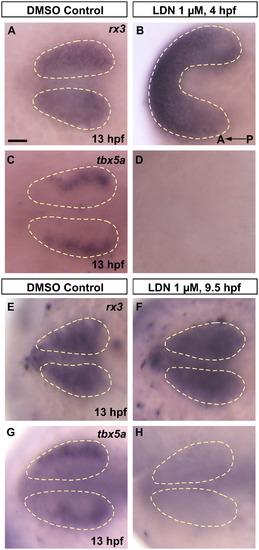
bmp signals necessary for dorsal fate initiation are acting between 9.5 hpf and 13 hpf. Pharmacological inhibition of bmp signaling using the small molecule inhibitor LDN 193189 prevents initiation of dorsal fate in the retina. (A and B) rx3 expression following 4–13 hpf continuous treatment with 1% DMSO in E2/GN (control) or 1 μM LDN 193189 in 1% DMSO E2/GN shows that treatment with LDN 193189 leads to severely dorsalized embryos, yet these embryos still form an eye field. (C and D) Contrary to DMSO controls, eye fields in LDN treated embryos fail to initiate expression of the prospective dorsal marker tbx5a. (E and F) Embryos treated from 9.5–13 hpf with 1% DMSO in E2/GN (control) or 1 μM LDN 193189 in 1% DMSO E2/GN both show normal rx3 expression, indicating normal eye field formation. (G and H) However, contrary to controls, optic vesicles in treated embryos fail to initiate expression of the prospective dorsal marker tbx5a, suggesting that dorsal fate is never initiated if bmp signaling is inhibited after 9.5 hpf. All images are dorsal views, anterior to the left. Scale bar=50 μM. Dashed yellow lines outline optic vesicle domains. |
|
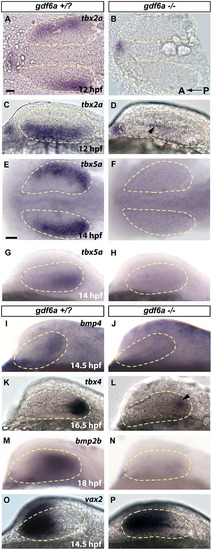
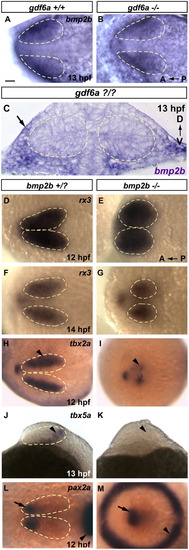
gdf6a is necessary for establishing dorsal retinal fate. In gdf6a mutants, dorsal retinal patterning genes either fail to express, or have greatly downregulated expression. Ventral genes are turned on normally, but expand shortly after to fill almost the entire retinal field. Expression of dorsally or ventrally expressed genes is shown at stages shortly after they are first detectable in wild type. (A–D) Robust 12 hpf tbx2a in gdf6a siblings is only faintly detectable in gdf6a mutants (arrowhead in D). (E–H) Expression of tbx5a begins at 12 hpf (14 hpf shown in figure) in sibling embryos, but is never seen in gdf6a mutants (F and H). (I–N) Similarly, bmp4 and bmp2b are completely lost in gdf6a mutants, while tbx4 expression is initiated at very low levels (arrowhead in L). (O and P) Expression of vax2 is only slightly expanded at 14.5 hpf, and continues to expand to fill most of the retina by 24 hpf (See supplementary Fig. 3R) Anterior to the left; A, B, E and F are dorsal views; C, D, G–R are lateral views. Scale bars=50 μM. Dashed yellow lines outline optic vesicles. |
|
bmp2b is necessary for dorsal retina initiation. Despite severe morphological defects, bmp2b mutants develop rx3 expressing eye fields. They fail, however to initiate expression of several dorsal markers. (A–C) bmp2b is expressed in the extraocular ectoderm in 13 hpf gdf6a siblings (A) and this expression domain is unchanged in gdf6a mutants (B). (D–G) rx3 expression in siblings and bmp2b mutants at 12 and 14 hpf. (H and I) The earliest dorsal fate marker, tbx2a, is expressed in siblings beginning at 11 hpf (12 hpf shown, arrowhead). tbx2a is greatly downregulated in bmp2b mutants (H, arrowhead). (J and K) tbx5a expression is initiated at 12 hpf in siblings (13 hpf shown, arrowhead in I), but never turns on in bmp2b mutants (arrowhead in K where staining is absent). (L and M) In 12 hpf siblings, pax2a marks the prospective ventral retinal domain (arrow, L), which is also present in bmp2b mutants at this time point (arrow, M), despite severe dorsalization, which leads to a circular midbrain–hindbrain boundary (arrowheads; compare M with L). A and B, D–I, L and M are dorsal views, anterior to the left. J and K are lateral views, anterior to the left; C is a transverse section. Scale bars=50 μM. Dashed yellow lines outline optic vesicles where boundaries are visible. |
|
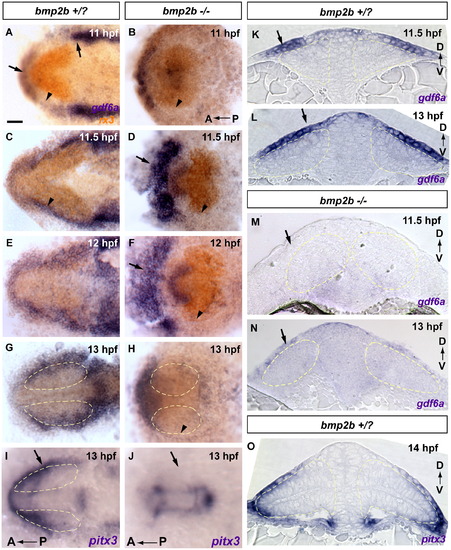
Dorsal initiation signal arises from non-neural ectoderm at 11–12 hpf. Extraocular gdf6a is expressed in the correct time and place to initiate dorsal fate within the retina, and expression is absent in bmp2b mutants, lacking dorsal fate. These results suggest that lateral head ectoderm is the tissue responsible for initiating dorsal fate within the retina. (A–H) gdf6a expression in blue, rx3 expression in orange. (A, C and K) gdf6a in +/? siblings is expressed in the non-neural head ectoderm, and is initially strongest anterior and posterior to the optic vesicle (arrows in A, arrowhead in A marks weaker expression lateral to the optic vesicle). At 11.5 hpf gdf6a expression has intensified covering the lateral portion of the optic vesicle (C, arrowhead; K transverse section, arrow). (E, G and L) gdf6a later expands to cover the majority of the dorsal leaflet of the optic vesicle (G; L transverse section, arrow). (K and L) Extraocular gdf6a at 11.5–13 hpf is directly adjacent to optic vesicle tbx5a, initiated at 12 hpf (Compare K and L to Fig. 1A0–A3). (B, D, F, H, M and N) In bmp2b mutants the lateral extraocular domain of gdf6a is not detectable or greatly downregulated (arrowheads in B, D, F and H, arrows in M and N). Expression of gdf6a anterior to the optic vesicle is upregulated in mutants (arrows in D and F). (I, J and O) The extraocular lateral domain of the non-neural ectoderm and lens placode marker pitx3 is also absent in bmp2b mutants (arrows in I and J), indicating that bmp2b mutants do not form the non-neural ectodermal tissue necessary for gdf6a expression and subsequent initiation of dorsal fate. (A–J) whole mount dorsal view, anterior to the left. (K–O) transverse sections. Scale bars=50 μM. Dashed yellow lines outline optic vesicles. |
|
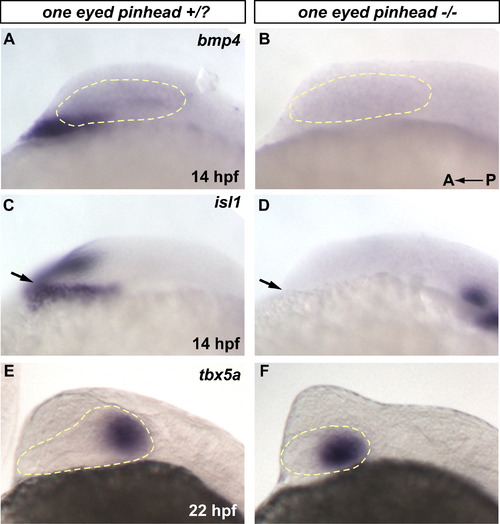
The prechordal plate, which expresses bmp4, is not necessary for dorsal retina initiation. (A-B) One eyed pinhead (oep m134) mutants do not initiate expression of bmp4 in the prechordal plate region (C-D) oep mutants do not form prechordal plate tissues as assayed by expression of the prechordal plate marker isl1 at 14 hpf (arrows in C and D). (E-F) However, dorsal retina develops normally in these mutants as assayed by expression of tbx5a at 22 hpf. |
|
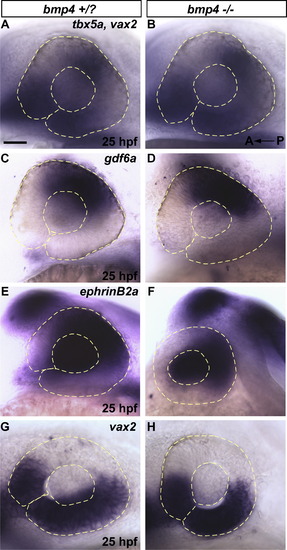
>bmp4 is not necessary for initiation or maintenance of dorsal retinal fate. bmp4 is not necessary for initiation or maintenance of dorsal retinal fate. (A-F) The dorsal-specifying genes tbx5a, gdf6a, and ephrinB2a are expressed normally in bmp4st72 mutants at 25 hpf, as is the ventral specifying gene vax2. (G-H) The ventral patterning gene vax2 is also normally expressed in bmp4 mutants at 25 hpf. mRNA expression of polarized dorsal and ventral genes (tbx2a, tbx5a, gdf6a, and vax2) is also initiated normally in bmp4st72 mutants at 12-15 hpf (data not shown). Scale Bar = 50 μm. |
|
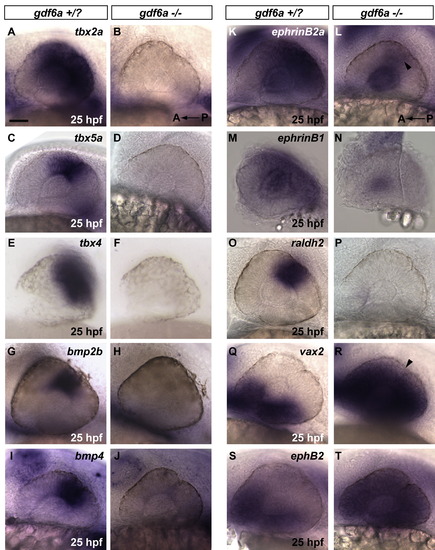
Gdf6a is necessary for maintenance of dorsal retinal fate. In gdf6a mutants, dorsal retinal patterning genes are absent or greatly downregulated at 25 hpf. Ventral genes are expanded to fill the majority of the retinal field. (A-P) Polarized dorsal 25 hpf expression of tbx2a, tbx5a, tbx4, bmp2b, bmp4,ephrinB2a, ephrinB1 and raldh2 in gdf6a siblings is absent in gdf6a mutants or faintly detectable in the case of ephrinB2a (arrowhead in L). (Q-T) Expression of vax2 and ephB2 ventral markers is robustly expanded at 25 hpf in gdf6a mutants to fill most of the retinal field (arrowhead in R marks small portion without vax2 expression). (A-T) Anterior to the left; lateral views. Scale Bar = 50 μm. |
Reprinted from Developmental Biology, 371(1), Kruse-Bend, R., Rosenthal, J., Quist, T.S., Veien, E.S., Fuhrmann, S., Dorsky, R.I., and Chien, C.B., Extraocular ectoderm triggers dorsal retinal fate during optic vesicle evagination in zebrafish, 57-65, Copyright (2012) with permission from Elsevier. Full text @ Dev. Biol.