- Title
-
Foxd3 mediates zebrafish myf5 expression during early somitogenesis
- Authors
- Lee, H.C., Huang, H.Y., Lin, C.Y., Chen, Y.H., and Tsai, H.J.
- Source
- Full text @ Dev. Biol.
|
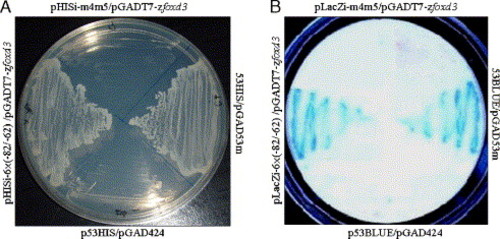
The yeast one-hybrid system was used to identify Foxd3 bound to the myf5 -82/-62 cassette. (A) The yeast one-hybrid assay of clones transfected with the plasmids indicated the following: pHISi-6× (-82/-62), which contains six repeats of -82/-62; pGADT7-zfoxd3, which contains foxd3 with the GAL4 activation domain; pHISi-m4m5, which contains the mutated -82/-62 cassette; p53HIS/pGAD53m, the positive control; p53HIS/pGAD424, the negative control. Yeasts that harbored plasmids containing wild-type -82/-62 grew under growth-inhibiting conditions (histidine and leucine were absent, 60 mM of 3-amino-1,2,4-triazole was present) when Foxd3 fused to the activation domain was expressed. Yeasts containing plasmids with mutated -82/-62 did not grow. (B) The colony-lift filter method was used to perform β-galactosidase assays. Yeasts were transformed, as indicated, with each of the following plasmids: pLacZi-6× (-82/-62), the bait plasmid that contains six repeats of -82/-62 and carries the lacZ reporter gene; pLacZi-m4m5, which contains four repeats of the mutated -82/-62 sequence; p53BLUE/pGAD53m, the positive control; p53BLUE/pGAD424, the negative control. Positive β-galactosidase activity, shown in blue, was detected only when the foxd3 activation domain fusion was expressed in yeasts containing wild-type -82/-62. No activity was detected in yeasts with mutated -82/-62. |
|
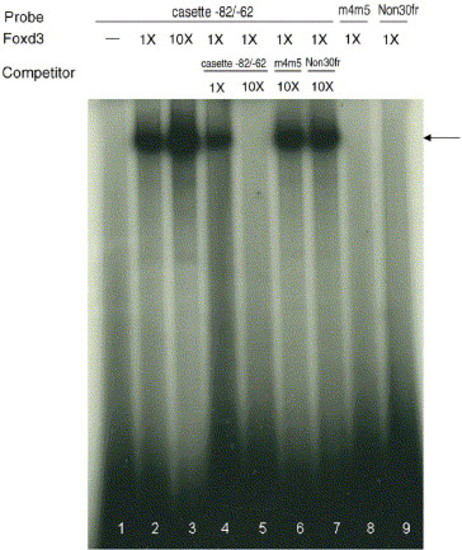
Binding between purified recombinant Foxd3 and 32P-radiolabeled oligonucleotide probes was studied with the electrophoretic mobility shift assay. The arrow indicates the shifted band formed by double-stranded oligonucleotide and recombinant Foxd3. Three probes were used: cassette -82/-62, mutated -82/-62 (m4m5, in which the -70/-62 sequence was mutated), and a nonspecific DNA sequence (Non-30fr). Radiolabeled cassette -82/-62, without added nuclear extracts, was the negative control (lane 1). The shifted bands were abolished completely when excess amount of unlabeled -82/-62 oligonucleotide was added (lanes 4 and 5). Recombinant Foxd3 did not bind to m4m5 (lane 8) or Non30fr (lane 9). In addition, m4m5 and Non30fr competitors did not compete for binding with Foxd3 and cassette -82/-62 (lanes 6 and 7). |
|
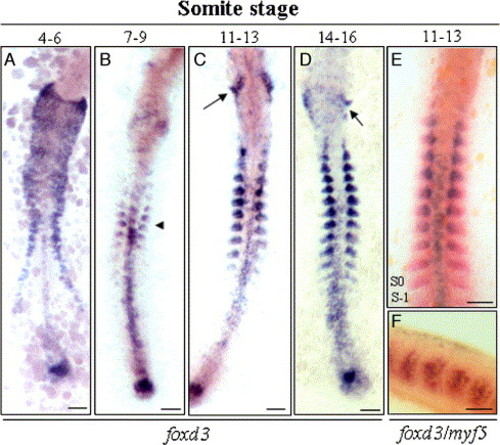
Temporal and spatial expression of foxd3 in zebrafish embryos at different somite stages. (A) At the 4- to 6-somite stage, foxd3 was transcribed in the floor plate, presumptive neural crest cells, and tail bud. (B) At the 7- to 9-somite stage, foxd3 transcripts were first detected in the somites (arrowhead) and the signals were weak in migrating neural crest cells. In addition to the somitic mesoderm, foxd3 mRNA also was found in the tail bud and posterior premigratory crest. (C, D) At the 11- to 16-somite stage, foxd3 transcription increased incrementally after each pair of somites was formed. foxd3 transcripts in the lateral head were down-regulated, but foxd3 expression was strong in the somites and cranial ganglia posterior to the otic vesicle (arrow). (E, F) In the 11- to 13-somite stage of wild-type embryos, double in situ hybridization using red-labeled myf5 and blue-labeled foxd3 probes was used to show that expressions of these genes were colocalized in the posterior part of the somites. Scale bars: 100 μm. EXPRESSION / LABELING:
|
|
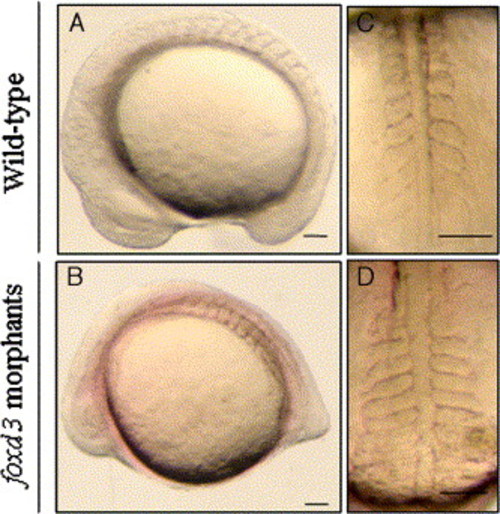
The effect of inhibiting Foxd3 protein translation on somitogenesis in zebrafish embryos. Lateral and dorsal views of wild-type (upper panels) and foxd3-MO1-injected (lower panels) 12- to 14-somite stage embryos. Embryos that were injected with 10 ng of foxd3-MO1 displayed an abnormal phenotype, including an abnormal tail bud, a reduced head (A vs. B), and wider somites with an irregular somite boundary (C vs. D). Scale bars: 100 μm. PHENOTYPE:
|
|
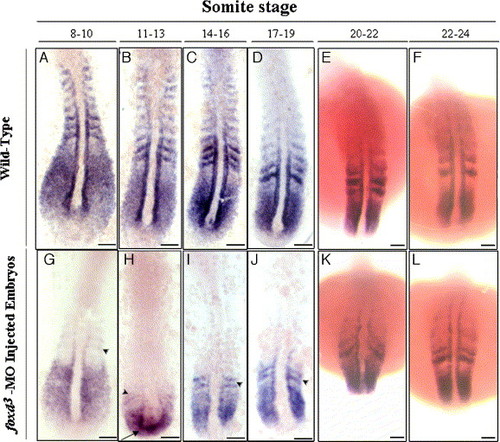
Whole-mount in situ hybridizations showing gene expression in wild-type (A–F) and the foxd3-MO1-injected embryos (G–L) at different somite stages. In embryos with 8 to 19 somites, myf5 expression in the somites and adaxial cells of foxd3-MO1-injected (10 ng) embryos was much lower than in wild-type embryos. Weak myf5 signals appeared in somites 0 and -1 (arrowheads) and in presomitic mesoderm. Ectopic expression of myf5 was observed in the tail bud (arrow in H). By the 20- to 24-somite stage, myf5 expression patterns in foxd3-MO1-injected and wild-type embryos were similar (E, F vs. K, L). Scale bars: 100 μm. EXPRESSION / LABELING:
|
|
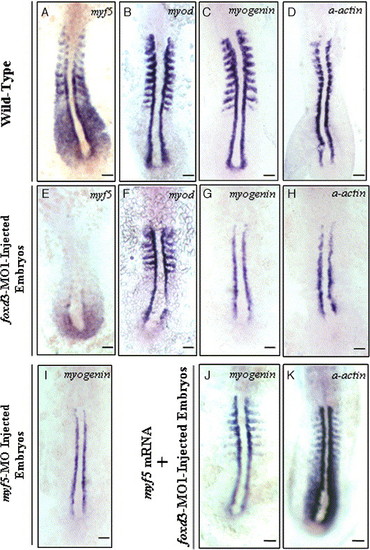
Effect of inhibiting Foxd3 protein synthesis on myf5, myod, myogenin, and α-actin expression in embryos with 10–12 somites. In foxd3-MO1-injected (10 ng) embryos, myf5 expression in the somites, adaxial cells, and presomitic mesoderm was reduced dramatically (A vs. E) but myod expression was unchanged (B vs. F). In foxd3-MO1-injected embryos, the expression of myogenin (C vs. G) and α-actin (D vs. H) was abolished in the somites, except in adaxial cells. Embryos injected with 4 ng of myf5-MO exhibited reduced myogenin expression in the somites, a finding similar to the defective phenotype of foxd3 morphants (I vs. G). Co-injection of 600 ng/μl myf5-capped mRNA rescued expression of myogenin (J) and α-actin (K) in foxd3-MO1-injected embryos. Scale bars: 100 μm. EXPRESSION / LABELING:
|
|
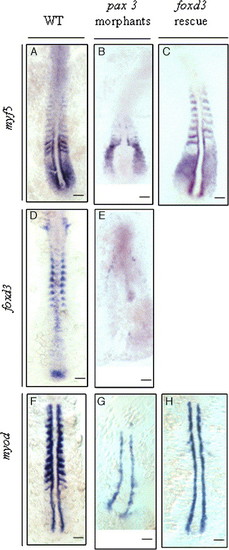
The effect of inhibiting Pax3 protein synthesis on the expression of foxd3, myf5, and myod in the zebrafish embryos at 5 to 8 somites. Probes were used to detect myf5 (A, B, C), foxd3 (D, E), and myod (F, G, H) in the wild-type embryos (A, D, F), the pax3-MO1-injected (6 ng) embryos (B, E, G), and the embryos co-injected with 6 ng of pax3-MO1 and 25 pg of foxd3 mRNA (C, H). (B) In pax3-MO1-injected embryos, myf5 expression was restricted in the presomitic mesoderm (PSM) and weak in somites 0 and -1 and in the adaxial cells on the sides of somites 0 and -1. (E) The shape of the neural plate became abnormal in the pax3-MO1 morphants, and foxd3 expression was weak in their neural fold and tail bud. (G) Meanwhile, myod expression was down-regulated in the somites, but it was expressed in adaxial cells. In the rescue experiment, co-injection of foxd3 mRNA and pax3-MO1 restored myf5 expression in the somites (C), whereas the myod expression was not rescued (H). Scale bars: 100 μm. EXPRESSION / LABELING:
|

Unillustrated author statements EXPRESSION / LABELING:
|
Reprinted from Developmental Biology, 290(2), Lee, H.C., Huang, H.Y., Lin, C.Y., Chen, Y.H., and Tsai, H.J., Foxd3 mediates zebrafish myf5 expression during early somitogenesis, 359-372, Copyright (2006) with permission from Elsevier. Full text @ Dev. Biol.