- Title
-
Hedgehog signalling is required for maintenance of myf5 and myoD expression and timely terminal differentiation in zebrafish adaxial myogenesis
- Authors
- Coutelle, O., Blagden, C.S., Hampson, R., Halai, C., Rigby, P.W.J., and Hughes, S.M.
- Source
- Full text @ Dev. Biol.
|
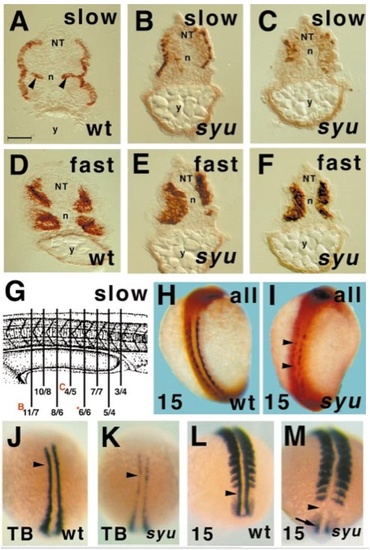
Sonic you zebrafish mutants have deficits in slow muscle. Transverse sections show that somitic muscle of sibling wild type or heterozygote (wt, A, D) and homozygous mutant sonic you (syu, B, C, E, F, I) 24-hpf zebrafish embryos contain superficial slow myosin-immunoreactive muscle fibres (A–C, I) and medial fast myosin-immunoreactive muscle fibres (D–F). Note that syu embryos entirely lack the medial slow muscle pioneer fibres observed in the nonmutant siblings (compare B, C with arrowheads in A) and show a progressive loss of lateral slow muscle fibres from rostral regions (not shown), through rostral yolk extension regions (A, B, somite ~15) to caudal yolk extension (C, somite ~18). Quantitation of numbers of slow muscle fibres in serial transverse sections through the yolk extension confirmed that the number of fibres was reduced throughout the region from around 20 on each side of the animal in wild-type embryos to fewer than half that number in syu embryos (G; sketch adapted from Kimmel et al., 1995). Values are numbers of slow MyHC-expressing cells on the left/right side of each transverse section. Red letters indicate sections shown in corresponding panels of this figure; asterisk, fig. 5D. Retarded slow muscle formation is already observed in wholemounts of 15 somite embryos (H, I, dorsal view, anterior to top). Adaxial slow muscle fibres, here visualised by general sarcomeric MyHC immunohistochemistry, lie next to the notochord, but have yet to migrate through the somite to their final superficial position. In syu embryos, MyHC is undetectable at some rostrocaudal positions (arrowheads, I) and is reduced elsewhere. Wholemount in situ mRNA hybridisation for myoD shows that this early muscle marker is reduced in adaxial cell precursors (arrowheads, J–M) of slow muscle fibres in some individuals from syu heterozygote crosses (K, M) compared to the wild-type pattern (J, L) at both tailbud (J, K) and 15 somite (L, M) stages; dorsal views, anterior to top. Loss of myoD appeared fully penetrant at 15 somite, but of reduced penetrance at tailbud stages (see text). Note the residual myoD transcript in the preadaxial cells of the 15 somite syu embryo tailbud (arrow, M). NT, neural tube; n, notochord; y, yolk extension. Bar, 50 μm in A–F, 175 μm in H, I, and 135 μm in J–M. |
|
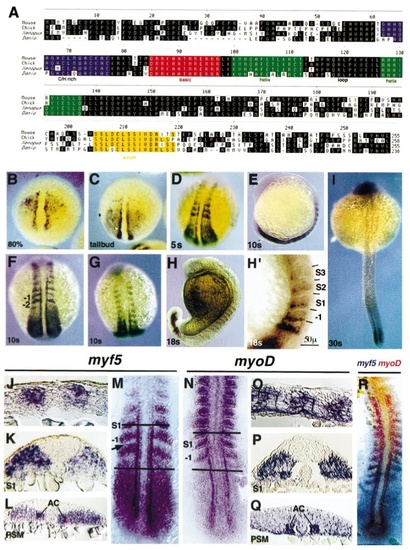
CLUSTAL alignment of the conceptual translation product of the zebrafish myf5 gene with representatives of other major vertebrate groups (mammals, birds, amphibia) (A). Localisation of myf5 (B–M) and myoD (N–Q) transcripts by in situ hybridisation at different developmental stages. Myf5 transcripts first appear in the presegmentation embryo in two stripes adjacent to the notochord and in broad triangular-shaped domains on each side of the embryonic shield at 9 hpf (80% epiboly) (B). The triangular expression domains appear to transit towards the posterior end in 10-hpf (tailbud stage) embryos (C). At the onset of segmentation lateral stripes of expression are present in the presomitic mesoderm and in the first 2–3 somites at 12 hpf (5– 6 somites); dorsal view (D). The maximal extent of myf5 expression, including the first 5– 6 somites, is reached at 14 hpf (10 –11 somites), including the lateral presomitic cells; lateral view (E), dorsal posterior view (F), dorsal anterior view (G). At subsequent stages, myf5 is rapidly down-regulated in rostral somites, 18 hpf (18 somites) (H, lateral view, and at higher magnification showing the location of myf5 expression in the posterior region of somites -I to IV, H′). Expression in the tail adjacent to the notochord is visible at 24 hpf (30 somites) (I). Flat-mounted dorsal view of 15-hpf (12–13 somite) embryo (M), showing down-regulation of myf5 in presomitic mesoderm in the region of somite -I (arrow). Sections from similar 15-hpf embryos: parasagittal in nascent somites (J, anterior to left), transverse through somite 1 (K), and transverse through presomitic mesoderm (L). Localisation of myoD: 15-hpf (12–13 somite) at mount (N) and sections: parasagittal (O, anterior to left), transverse through somite 1 (P), and transverse through presomitic mesoderm (Q). Both myf5 and myoD signals are most intensely reactive in adaxial cells in presomitic mesoderm and in lateral posterior cells of forming somites, in which myoD persists after myf5 declines. Neither myf5 nor myoD is highly expressed in the medial tail bud. Two-colour in situ mRNA hybridisation (R) with a red myoD probe and a purple myf5 probe at 13 hpf (7– 8 somites) dorsal view shows that myf5 and myoD are expressed in partially overlapping domains, in both the posterior somites and the adaxial cells. EXPRESSION / LABELING:
|
|
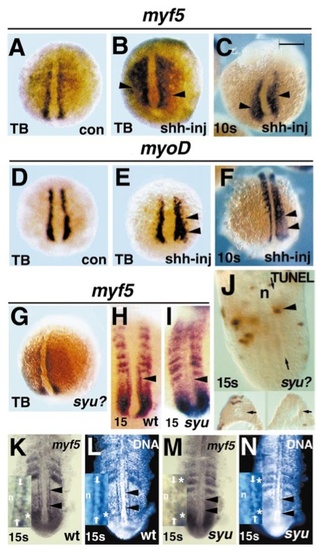
myf5 is maintained in adaxial cells by Sonic hedgehog from the ventral midline. Injection of RNA encoding shh into two to four cell zebrafish embryos causes widespread ectopic expression of both myf5 (A–C arrowheads) and myoD (D–F arrowheads) in the lateral presomitic mesoderm at tailbud (A, B, D, E) and 10 somite (C, F) stages. myf5 expression appears unaffected by lack of Sonic hedgehog in clutches of tailbud stage embryos from a syu heterozygote cross (G, a randomly selected embryo from 45 in such a cross, shown in three-quarter dorsal view, posterior at bottom). In adaxial cells in the presomitic mesoderm of the future yolk extension region of 15 somite syu embryos, myf5 mRNA levels are reduced compared to wild type (H, I arrowheads), although lateral presomitic mesoderm and somitic expression appears unaffected. (J) TUNEL staining revealed few apoptotic cells in 15 somite embryos from a syu heterozygote cross; dorsal view at mount. Single TUNEL-reactive cells (arrowhead) were detected in adaxial positions (between arrows) adjacent to the notochord (n) in about 10% of embryos. Serial transverse sectioning of TUNEL-stained embryos confirmed the presence of adaxial cells (insets, arrows, dorsal to top) and the low frequency of adaxial apoptosis. The decline in adaxial myf5 mRNA in a syu embryo (M) compared to wild-type sibling (K) is not due to loss of the expressing cells, because visualisation of DNA-containing nuclei in the same individual embryos reveals cells with normal adaxial morphology and position (L, N arrowheads). Insets in K–N show anterior presomitic mesoderm of 15 somite stage embryos, notochord at left (n). Rows of adaxial cell nuclei (between arrows) are clearly discernible in wild type, with myf5 signal outlining the nuclei (K, L). In syu mutant (M, N) a row of adaxial-like nuclei (between arrows) does not have cytoplasmic myf5 signal, but is morphologically distinguishable from nuclei in both nonexpressing notochord (n) and myf5-expressing lateral paraxial presomitic mesoderm (asterisks). All images show posterior at bottom in either wholemount (A–I, dorsal view) or at mount (K–N, ventral view after removal of yolk cells) embryos. Bar, 200 μm in A–G, 120 μm in H, I, 67 μm in K–N, 48 μm in J, 30 μm insets K–N. |
|
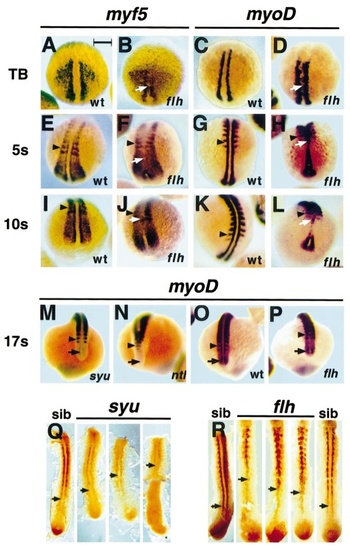
The adaxial cell phenotype of oating head and sonic you embryos is similar: failed caudal myf5 and myoD maintenance and delayed terminal differentiation. myf5 (A, B, E, F, I, J) and myoD (C, D, G, H, K–P) mRNAs are reduced in adaxial cells, but not elsewhere in homozygous h embryos (B, D, F, H, J, L, P), in which notochord fails to mature, compared to wild-type or heterozygous siblings. As in syu embryos, h embryos show good adaxial expression at tailbud stage, but h additionally has ectopic mesodermal midline myf5 and myoD mRNA (arrows, B, D). At 5 somites, much myf5 mRNA is lost in adaxial cells of anterior presomitic mesoderm (F), and myoD mRNA-containing cells are converging into the midline (H). This trend is accentuated at 10 somites, when both myf5 and myoD mRNAs (J, L) appear reduced in anterior presomitic adaxial cells, except for rare clusters of cells that appear to reexpress in the anterior midline (arrows, F, H, J, L). By 17 somites, myoD mRNA is undetectable in anterior presomitic mesoderm of h (P) and syu (M) embryos, but nevertheless is detected at wild-type levels anking the notochord anlagen in the tip of the tailbud (arrows, M, O, P). In contrast, this expression is entirely lost in no tail embryos (arrow, N). Arrowheads mark the most posterior left somite boundary in each panel. Wholemount immunohistochemical detection of sarcomeric MyHC (Q, R) reveals that, whereas adaxial cell terminal differentiation precedes or is contemporaneous with somite formation in siblings, differentiation is substantially and variably delayed in syu (Q) or h (R), with the caudalmost detectable MyHC being in older somites (arrows). Bar, 200 μm in A–P and 177 μm in Q, R. EXPRESSION / LABELING:
PHENOTYPE:
|
|
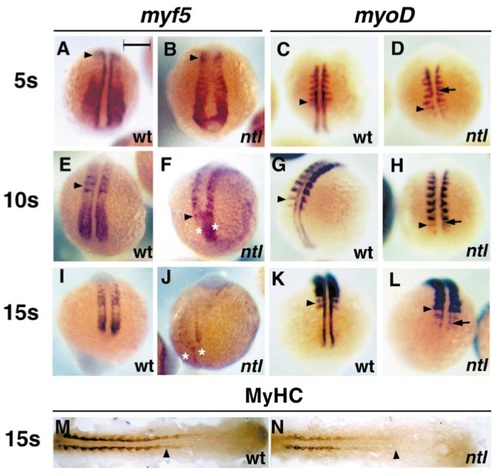
Failure of adaxial myogenesis in posterior no tail embryos occurs at the Mrf induction stage. Analysis of 5 somite (A–D), 10 somite (E–H), and 15 somite (I–N) ntl embryos (B, D, F, H, J, L, N) reveals that adaxial myf5 (A, B, E, F, I, J) and myoD (C, D, G, H, K, L) expression is severely reduced at all stages and positions examined, compared to wild-type or heterozygous siblings (A, C, E, G, I, K, M). In contrast, lateral somitic expression levels of both myf5 and myoD appear unaffected by the ntl mutation at early stages. Note the delayed appearance of substantial medial myoD mRNA at somitic levels (arrows, D, H, L). myf5 mRNA accumulation in the tailbud tip is decreased at later stages of ntl development (asterisks, F) and is almost lost caudally in ntl embryos as outgrowth ceases (asterisks in J, dorsal three-quarter view, tailbud at bottom). Wholemount visualisation of all sarcomeric MyHC shows that adaxial muscle differentiation is substantially delayed in the 15 somite stage ntl embryo (M, N dorsal view, anterior to left). Bar, 200 μm in A–L and 164 μm in M, N. EXPRESSION / LABELING:
PHENOTYPE:
|
|
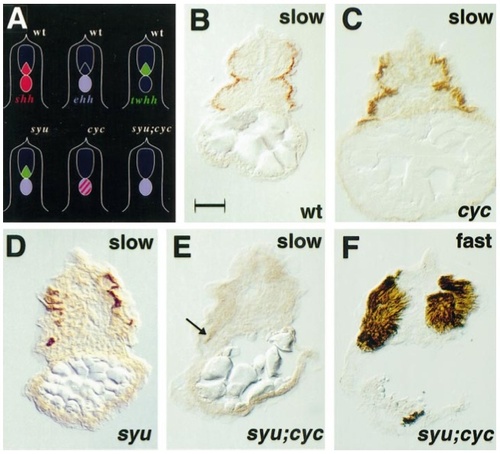
Exacerbation of slow muscle deficits in sonic you;cyclops double mutant. Schematic representation of hedgehog gene expression in wild-type and mutant zebrafish embryos (A). Slow MyHC-expressing muscle cells (B–E) in the yolk extension region of 24-hpf wild-type (B) and cyc (C) embryos are indistinguishable. syu embryos show slow muscle reduction (D). A syu;cyc double mutant embryo has only isolated residual slow muscle cells (E, arrow), yet expresses substantially normal fast muscle in an adjacent section (F). All panels show transverse sections, dorsal at top. Each of three syu;cyc mutants from 91 cross progeny, identified by lack of oorplate and U-shaped somites,were serially sectioned and individuals were found to contain two, five, and eight slow cells. Bar, 50 μm. |
Reprinted from Developmental Biology, 236(1), Coutelle, O., Blagden, C.S., Hampson, R., Halai, C., Rigby, P.W.J., and Hughes, S.M., Hedgehog signalling is required for maintenance of myf5 and myoD expression and timely terminal differentiation in zebrafish adaxial myogenesis, 136-150, Copyright (2001) with permission from Elsevier. Full text @ Dev. Biol.