- Title
-
Ascl1a/Dkk/ beta -catenin signaling pathway is necessary and glycogen synthase kinase-3 beta inhibition is sufficient for zebrafish retina regeneration
- Authors
- Ramachandran, R., Zhao, X.F., and Goldman, D.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
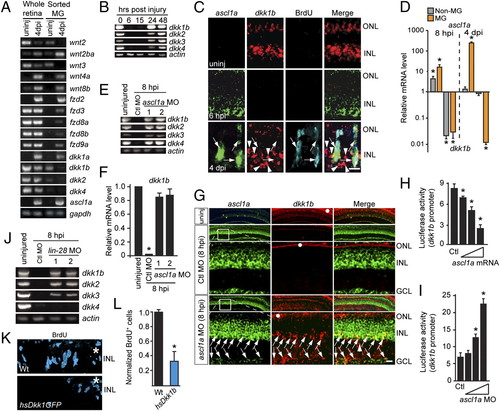
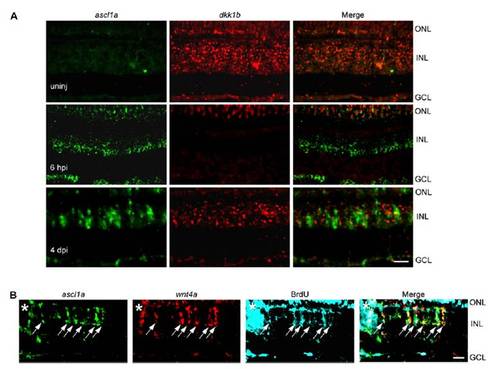
Ascl1a inhibits the expression of dkk genes during retina regeneration. (A and B) Injury-dependent regulation of Wnt signaling component mRNAs. (C) Double in situ hybridization shows mutually exclusive ascl1a and dkk1b gene expression. (D) ascl1a and dkk1b expression in FACS-purified MG and non-MG from injured retinas. Values are relative to uninjured retina. *P < 0.009. (E and F) Ascl1a knockdown prevents injury-dependent dkk gene suppression. (F) Quantification of E by qPCR. Values are relative to uninjured retina. *P < 0.0001. (G) In situ hybridization showing Ascl1a knockdown relieves injury-dependent dkk suppression. Boxed region in low-magnification image is shown in higher magnification in the row below. Arrows point to ascl1a+/dkk1b+ cells. White dots identify autofluorescence in ONL. (H and I) Injection of zebrafish embryos with dkk1b:gfp-luciferase reporter and increasing amounts of ascl1a mRNA (H) or ascl1a-targeting MO (I). *P < 0.005. (J) Lin-28 knockdown differentially affects injury-dependent dkk gene suppression. (K and L) Dkk1b overexpression inhibits cell proliferation at 4 dpi. *P < 0.003. (Scale bars, 10 μm.) ONL, outer nuclear layer; INL, inner nuclear layer; GCL, ganglion cell layer. EXPRESSION / LABELING:
|
|
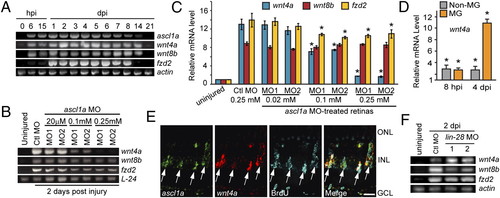
Ascl1a regulates wnt4a expression via a Lin-28 independent pathway. (A) Time course of injury-dependent gene expression. (B and C) MO-mediated Ascl1a knockdown suppresses wnt4a gene induction at 2 dpi. *P < 0.0005. (D) wnt4a expression in MG and non-MG after retinal injury relative to uninjured control retina. *P < 0.0007. (E) ascl1a and wnt4a mRNAs are coexpressed in BrdU+ MG-derived progenitors at 4 dpi. (Scale bar, 10 μm.) (F) Lin-28 knockdown does not suppress injury-dependent wnt4a or fzd2 induction. Abbreviations are as in Fig. 1. EXPRESSION / LABELING:
|
|
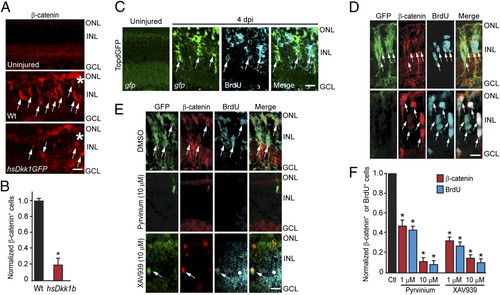
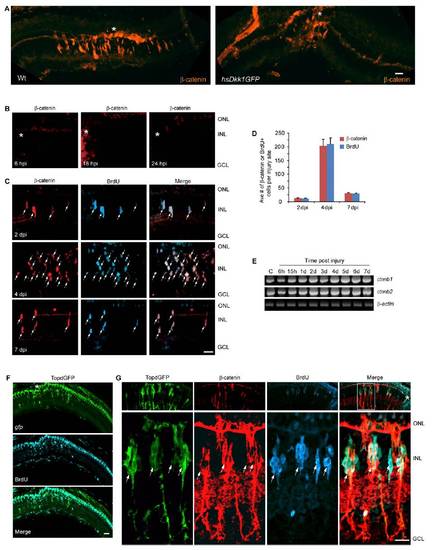
β-Catenin accumulation in MG-derived progenitors is necessary for retina regeneration. (A and B) Injury-induced β-catenin expression at 4 dpi is blocked by Dkk1b overexpression in hs:Dkk1GFP fish. (C) Injury-dependent induction of GFP expression in the β-catenin reporter fish, TOPdGFP. (D) Accumulation of β-catenin in GFP+ MG-derived proliferating progenitors of 1016 tuba1a:gfp fish at 4 dpi. (Upper) Standard immunohistochemical protocol. (Lower) Epitope retrieval protocol. (E and F) Pyrvinium and XAV939 inhibit the generation of GFP+, β-catenin+, and BrdU+ MG-derived progenitors at 4 dpi in 1016 tuba1a:gfp fish. White dot identifies an area of autofluorescence that results from long exposures to detect fluorescent signals. *P < 0.001. (Scale bars, 10 μm.) Abbreviations are as in Fig. 1. EXPRESSION / LABELING:
|
|
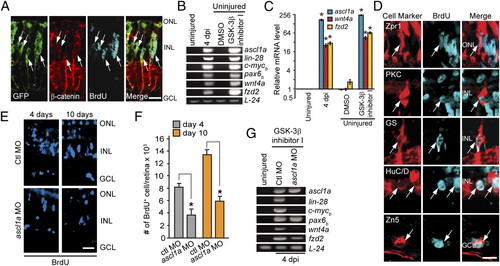
β-Catenin stabilization stimulates MG dedifferentiation and the generation of multiple retinal cell types in the uninjured retina. (A) Intravitreal injection of GSK-3β inhibitor induces GFP, β-catenin, and cell proliferation in the uninjured retina of 1016 tuba1a:gfp transgenic fish. (B and C) Expression of regeneration-associated genes in uninjured retinas treated with the GSK-3β inhibitor. *P < 0.0001. (D) GSK-3β inhibitor-induced retinal progenitors proliferate and generate all major retinal cell types. (E and F) Effect of Ascl1a knockdown on MG proliferation in the GSK-3β inhibitor-treated retina. *P < 0.002. (G) Effect of Ascl1a knockdown on genes associated with MG dedifferentiation in the GSK-3β inhibitor-treated retina. [Scale bars, 10 μm (A and D); 20 μm (E).] Abbreviations are as in Fig. 1. EXPRESSION / LABELING:
|
|
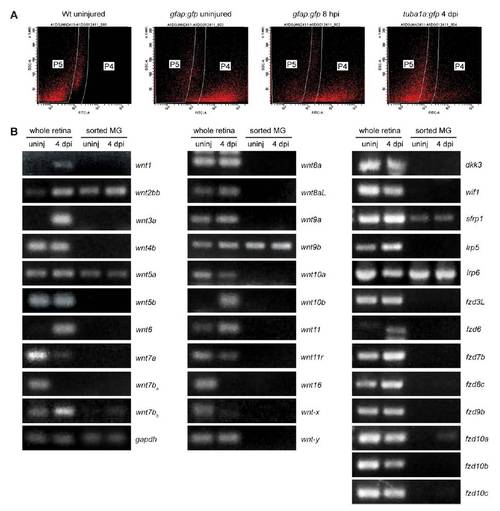
FACS and Wnt component gene expression. (A) Representative scatter plots used to FACS purify GFP-expressing cells. Wild type (Wt) retinal cells do not fall into the P4 window, whereas GFP+ retinal cells from gfap:gfp and 1016 tuba1a:gfp transgenic fish retinas readily sort into this window. (B) RT-PCR analysis of injury-dependent Wnt signaling component gene expression using total retina RNA or RNA from FACS-purified MG and MG-derived progenitors. Shown are Wnt signaling component genes that do not exhibit injury-dependent regulation in purified MG. |
|
Ascl1a is restricted from dkk1b expressing cells but coexpressed with wnt4a in proliferating progenitors in the injured retina. (A) Double in situ hybridization assay for ascl1a and dkk1b gene expression in the uninjured and injured retina. (B) At 4 dpi in situ hybridization reveals ascl1a and wnt4a are both expressed in proliferating MG-derived progenitors. (Scale bar, 10 μm.) |
|
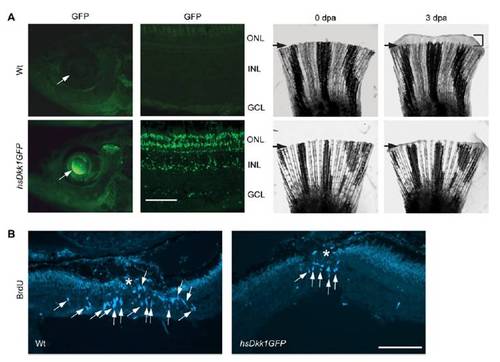
Dkk1b overexpression suppresses injury-dependent cell proliferation. (A) Heat shock in hs:Dkk1GFP fish, but not Wt fish, stimulates Dkk1b–GFP fusion protein expression in the retina and inhibits fin regeneration. Arrows in fin panels indicate the amputation plane. (B) Heat shock in hs:Dkk1GFP fish, but not Wt fish, inhibits injury-dependent MG proliferation. Asterisk indicates the injury site. (Scale bar, 50 μm.) dpa, days post amputation; INL, inner nuclear layer. |
|
β-catenin stabilization in the injured retina. (A) At 4 dpi epitope retrieval reveals β-catenin stabilization localized to the injury site (asterisk) in Wt fish. Overexpression of Dkk1b in hsDkk1GFP transgenic fish suppressed proliferation of MG-derived progenitors in the injured retina. (B) Epitope retrieval shows β-catenin is undetectable at 6–24 hpi. (C) Epitope retrieval shows β-catenin is first detected at 2 dpi in proliferating MG-derived progenitors (BrdU+). (D) Quantification of β-catenin+ and BrdU+ cells in the injured retina. (E) The two different β-catenin mRNA isoforms, ctnnb1 and ctnnb2, are expressed in the uninjured and injured retina and show marginal increases following injury. (F) Low magnification view of retinal section in TOPdGFP transgenic fish showing transgene gfp expression localized to the injury site (in situ hybridization) overlaps with BrdU+ cells (immunofluorescence). (G) At 4 dpi retinal progenitors (BrdU+) coexpress β-catenin and GFP in TOPdGFP transgenic fish. Upper shows a low magnification view of the retina, and Lower shows a high magnification view of the boxed area in the low magnification merge panel. Asterisks indicate injury sites. (Scale bar, 10 μm.) ONL, outer nuclear layer; INL, inner nuclear layer; GCL, ganglion cell layer. |
|
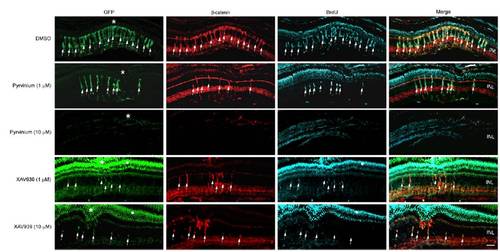
β-catenin destabilization inhibits MG dedifferentiation and proliferation in the injured retina. Retinas in 1016 tuba1a:gfp transgenic fish were injured and injected with either DMSO, pyrvinium, or XAV939. Pyrvinium and XAV939 inhibited GFP expression, β-catenin stabilization, and cell proliferation. Asterisks indicate injury sites. White dot identifies areas of GFP autofluorescence that also bleeds over into the blue channel, due to the long exposures that are necessary to visualize cells harboring very low levels of GFP. INL, inner nuclear layer. (Scale bar, 20 μm.) |
|
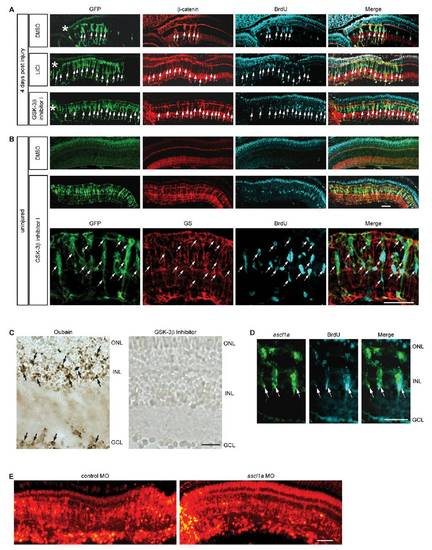
Pharmacological stabilization of β-catenin stimulates MG dedifferentiation and proliferation in the injured and uninjured retina. (A) LiCl and GSK-3β inhibitor injection into the eye at the time of retinal injury increases the regenerative zone as indicated by the increased number of GFP transgene expressing cells and the increased number of cells labeled with BrdU in 1016 tuba1a:gfp transgenic fish. (B) Intravitreal injection of the GSK-3β inhibitor into uninjured eyes stimulates MG dedifferentiation and cell proliferation in 1016 tuba1a:gfp transgenic fish examined at 4 d postinjection. Upper and Middle are a low magnification view of the retina; Bottom is a close-up view to show that glutamine synthetase (GS)-expressing MG have dedifferentiated (GFP+) and begun to proliferate (BrdU+) in response to GSK-3β inhibition. Asterisks indicate the injury sites. (C) TUNEL stain shows a lack of cell death in GSK-3β inhibitor-treated retinas. As a positive control retinas were treated with oubain, which resulted in widespread cell death. (D) In situ hybridization and BrdU immunofluorescence show GSK-3β inhibition-dependent induction of ascl1a in the uninjured retina is in proliferating MG-derived progenitors. (E) Retinal sections showing lissamine- labeled control and ascl1a-targeting MOs are widely incorporated into most cells of the retina. ONL, outer nuclear layer; INL, inner nuclear layer; GCL, ganglion cell layer. (Scale bars, 20 μm.) |