- Title
-
MicroRNA-30a regulates zebrafish myogenesis through targeting the transcription factor Six1
- Authors
- O'Brien, J.H., Hernandez-Lagunas, L., Artinger, K.B., Ford, H.L.
- Source
- Full text @ J. Cell Sci.
|
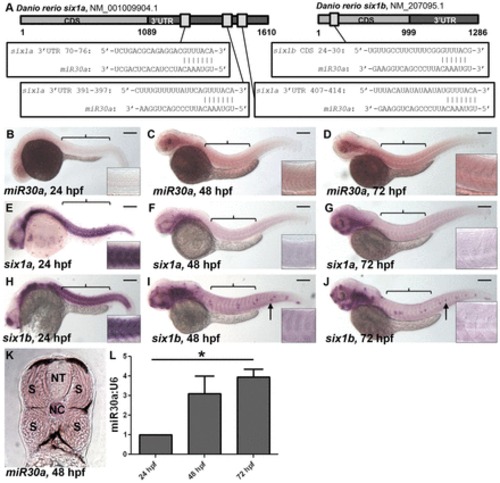
miR30a displays a reciprocal expression pattern with six1a/b. (A) Schematic diagrams show predicted miR30a target binding sites in the 32 untranslated region (UTR) of six1a and the coding sequence (CDS) of six1b. (B–J) In situ hybridization reveals a lack of mature miR30a expression at 24 hpf (B), whereas mature miR30a localizes to the somites (indicated by brackets) at 48 and 72 hpf (C,D). This miR30a expression is reciprocal to expression of six1a (E–G) and six1b (H–J) transcripts observed in the developing somites at 24 hpf (E,H), which is lost at 48 hpf (F,I) and 72 hpf (G,J). Scale bars: 250µm. Anterior is shown to the left; arrows, neuromasts. Insets show a higher magnification of somites above the yolk sac. (K) In situ hybridization for miR30a in 18-µm transverse sections of embryos at 48 hpf reveals miR30a expression specifically in the somites (S), as compared to a lack of expression in the neural tube (NT). NC, notochord. (L) A quantification of miR30a expression performed by real-time PCR analysis shows an increase in miR30a expression from 24 to 72 hpf in tails from wild-type embryos. *P<0.027, ANOVA with Bonferroni′s post-hoc test. EXPRESSION / LABELING:
|
|
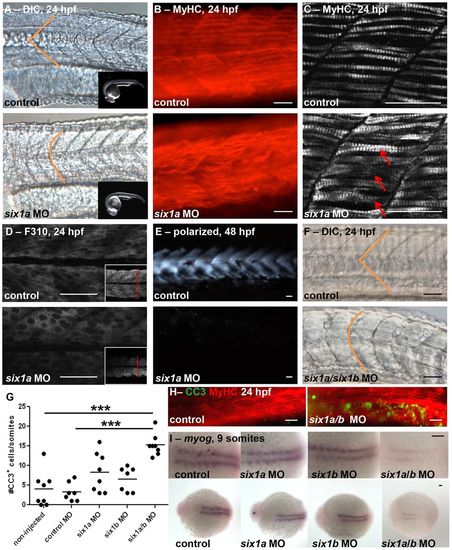
six1a and six1b function together during zebrafish myogenesis. Morpholino (MO)-mediated knockdown of six1a results in disrupted U-shaped versus normal V-shaped (indicated in orange) somite morphology as observed at 24hpf by (A) DIC microscopy (control, n = 35/35; six1a MO, n = 60/83; the inset images demonstrate matched staging of embryos) and (B) Myosin Heavy Chain (MyHC) immunofluorescence (control, n = 7/10; six1a MO, n = 10/16). (C–E) There is disarrayed muscle fiber organization upon six1a knockdown as revealed by confocal imaging of (C) both fast and slow MyHC with A4.1025 antibody (arrows indicate crossover of fibers), (D) fast-muscle-specific fibers with F310 antibody (inset demonstrates decrease in cross-sectional diameter) at 24 hpf, and (E) polarized-light-mediated detection of birefringence (control, n = 17/17; six1a MO, n = 13/19) of embryos at 48 hpf. (F) Dual MO-mediated knockdown of both six1a/b leads to dramatically U-shaped somites at 24 hpf as shown by DIC microscopy (control, n = 75/77; six1a/six1b MO, n = 100/154), and (G,H) increased cleaved caspase-3 (CC3) immunofluorescence staining at 24 hpf. Non-injected, n = 35 total embryos; six1a MO, n = 30; six1b MO, n = 18; six1a/b MO, n = 22. ***P<0.0001, ANOVA with Bonferroni′s post-hoc test. The line represents the mean. (H) CC3 (green) in embryos double-stained with MyHC (red) to observe the muscle fibers. (I) Dorsal view of in situ hybridization at 9 somites shows an early delay of myogenin (myog) expression (control, n = 15/18; six1a MO, n = 16/19; six1b MO, n = 21/27; six1a/b MO, n = 8/17), which is strongest in the double six1a/b knockdown. Scale bars: 50µm. Anterior is to the left in all images. For A–F, H and I, n = the number of embryos represented by each image/the total number of embryos analyzed. EXPRESSION / LABELING:
|
|
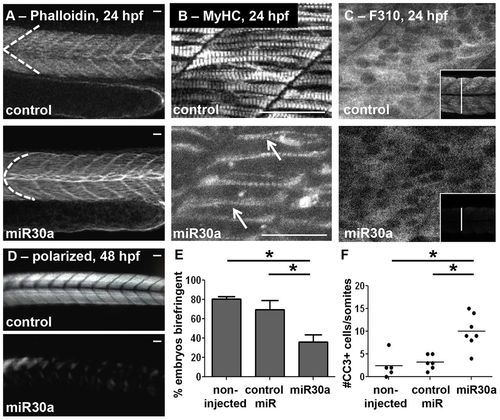
miR30a overexpression functionally phenocopies six1a/b knockdown. Overexpression of miR30a leads to abnormal somite morphology as observed in (A) phalloidin-stained embryos (control, n = 42/49; miR30a, n = 46/77) at 24hpf, and confocal images of (B) both fast and slow MyHC with A4.1025 antibody (arrows indicate crossover of fibers) and (C) fast-muscle specific fibers with F310 antibody (the inset demonstrates a decrease in cross-sectional diameter of the trunk muscle) at 24 hpf. (D) Polarized-light-mediated detection of birefringence of embryos at 48 hpf revealed disarrayed muscle fiber organization upon miR30a overexpression. (E) A quantification (mean±s.e.m.) of the results shown in D. Non-injected, n = 23 total embryos; control miR, n = 25; miR30a, n = 21. *P = 0.008, ANOVA with Bonferroni′s post-hoc test. (F) A quantified increase in cleaved caspase-3 (CC3) is also observed upon miR30a overexpression at 24 hpf. Non-injected, n = 23 total embryos; control miR (1ng), n = 25; miR30a (0.5ng), n = 20. *P<0.05, ANOVA with Bonferroni′s post-hoc test. Scale bars: 50µm. For A, n = the number of embryos represented by each image/the total number of embryos analyzed. EXPRESSION / LABELING:
|
|
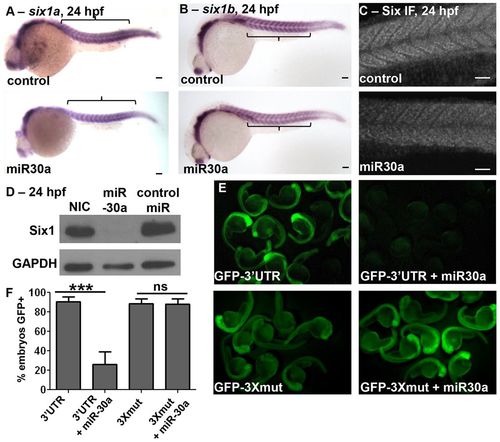
miR30a overexpression directly downregulates Six1 expression. miR30a overexpression results in (A) no substantial decreases in six1a mRNA (control, n = 25/28; miR30a, n = 31/43) and (B) no substantial decreases in six1b mRNA (control, n = 20/23; miR30a, n = 22/28) levels as visualized by in situ hybridization, (C) downregulated SIX family protein expression in the somites as observed in immunofluorescence images (control, n = 8/9; miR30a, n = 12/20), and (D) decreased Six1 protein levels as determined by western blot analysis, all at 24hpf. NIC, non-injected control. Anterior is shown to the left. (E) GFP expression at 24 hpf, imaged at low magnification in multiple embryos, is observed after injection of GFP–six1a-32UTR (GFP–32UTR) mRNA, and this GFP expression is lost when co-injected with miR30a duplex. However, GFP expression remains after co-injection of miR30a with a GFP–six1a-32UTR mRNA that has all three miR30a target seed sites mutated (GFP–3Xmut). (F) The percentage of embryos expressing GFP is quantified (mean±s.e.m.) in embryos expressing GFP–32UTR (n = 79 total embryos), GFP–32UTR + miR30a (n = 37), GFP–3Xmut (n = 90) or GFP–3Xmut (94). ***P = 0.0003, ANOVA with Bonferroni′s post-hoc test; ns, not significant. Scale bars: 50µm. For A–C, n = the number of embryos represented by each image/the total number of embryos analyzed. |
|
miR30a knockdown leads to abnormal somite and myosin fiber morphology, and to increased Six1 expression. Live images, anterior to the left, at (A) 48 hpf (control, n = 54/58; miR30a MO, n = 49/64; six1a mRNA, n = 36/45) and (B) 72 hpf (control, n = 75/75; miR30a MO, n = 30/40; six1a mRNA, n = 28/33) showing that miR30a knockdown (middle row) disrupts normal somite morphology to phenocopy six1a mRNA overexpression (bottom row). (C) Confocal images of MyHC stained with the A4.1025 antibody (control, n = 23/25; miR30a MO, n = 19/28; six1a mRNA, n = 11/20), and (D) loss of birefringence [mean±s.e.m.; non-injected control (NIC) n = 37 total embryos; control MO, n = 23; miR30a, n = 56; *P = 0.027, ANOVA with Bonferroni′s post-hoc test] demonstrate abnormal muscle fiber morphology upon miR30a loss at 48 hpf. (E,F) Increasing concentrations of miR30a-targeted MO result in a dose-dependent increase in (E) Six1 protein expression as visualized by western blot analysis at 48 hpf and (F) quantified following normalization to GAPDH. Scale bars: 100µm. For A, B, n = the number of embryos represented by each image/the total number of embryos analyzed. EXPRESSION / LABELING:
PHENOTYPE:
|
|
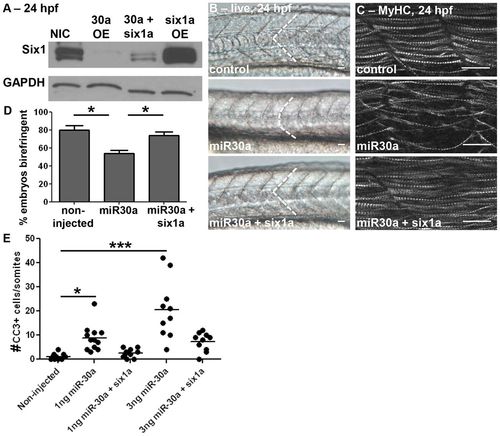
Effects of miR30a on somitogenesis are rescued by non-targetable six1a. (A) Western blot analysis at 24hpf reveals that co-injection of six1a mRNA lacking its 32UTR in combination with miR30a restores Six1 protein expression compared to that observed with overexpression of miR30a alone. NIC, non-injected control; OE, overexpression. (B) Live images (control, n = 54/55; miR30a, n = 50/66; miR30a + six1a mRNA, n = 25/56) at 24 hpf, anterior to the left, reveal that restoration of Six1 levels in the background of miR30a overexpression rescues normal somite morphology. (C) Confocal imaging of MyHC with A4.1025 antibody at 24 hpf (control, n = 24/27; miR30a, n = 24/28; miR30a+six1a: n = 14/18), and (D) birefringence (mean±s.e.m.) at 48 hpf (NIC, n = 38 total embryos; miR30a, n = 44; miR30a+six1a, n = 48; *P<0.05, ANOVA with Bonferroni′s post-hoc test) reveals that the disarrayed muscle fiber organization resulting from miR30a overexpression is rescued by non-targetable Six1. (E) A quantification of cleaved caspase-3 (CC3) shows cell death returns to endogenous levels in rescued embryos in a dose-dependent manner (NIC, n = 11 total embryos; 1ng miR30a: n = 12, 1ng miR30a+six1a, n = 11; 3ng miR30a, n = 10; 3ng miR30a+six1a, n = 11). The line represents the mean. ***P<0.0001, *P<0.05, ANOVA with Bonferroni′s post-hoc test. Scale bar: 50µm. For B, C, n = the number of embryos represented by each image/the total number of embryos analyzed. EXPRESSION / LABELING:
|
|
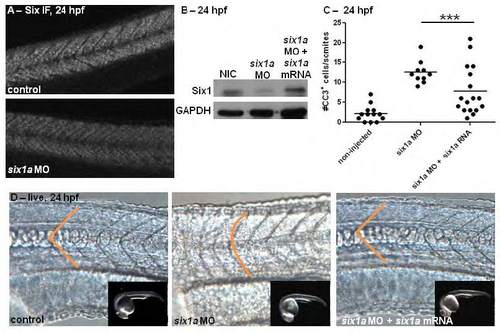
Rescue of six1a morphant phenotypes is observed with overexpression of six1a mRNA. Immunofluorescent (IF) staining for multiple SIX family members at 24 hours post-fertilization (hpf) reveals a decrease in protein expression in the somites when six1a morpholino (MO) is injected in 1-4 cell embryos (A). Injection of sixla MO leads to downregulation of Six1 protein specifically as observed by Western blot analysis (B); with this protein expression rescued by co-injection of six1a mRNA with six1a MO. Quantification of cleaved-caspase-3 IF staining (C) at 24 hpf demonstrates that cell survival is also rescued with six1a mRNA overexpression in six1a morphants, ***p<0.0001, ANOVA with Bonferroni’s post-hoc test. Live images (D) at 24 hpf, anterior to the left, show that the U-shaped somite morphology observed in six1a morphants is rescued to normal V-shape with six1a mRNA overexpression (control: n=13/13, 13 ng six1a MO: n=8/10, 13 ng six1a MO + 100pg six1a mRNA: n=12/17). Inset images confirm stage-matching across groups. |
|
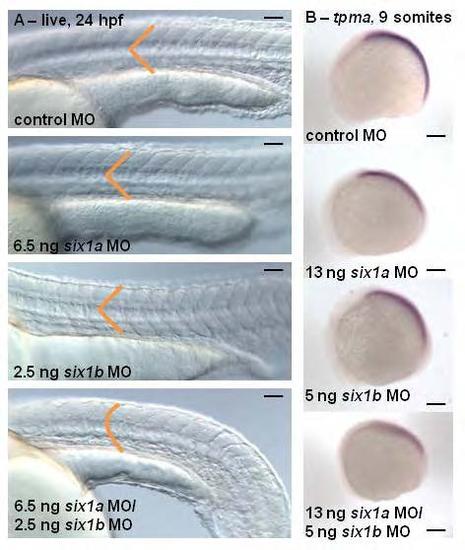
Morpholino studies reveal that six1a and six1b function together during zebrafish myogenesis. Live images (A) at 24 hours post fertilization (hpf), anterior to the left, show that lower doses of six1a MO and six1b MO injected alone do not disrupt normal somite morphology individually, only when injected together are U-shaped somites observed (control: n=75/77, 6.5 ng six1a MO: n=61/64, 2.5 ng six1b MO: n=50/56, six1a/b MO: n=100/154). Lateral views of in situ hybridization (B) at 9 somites shows an early delay of tropomyosin-α (tpma) (control: n=25/27, six1a MO: n=21/25, six1b MO: n=20/25, six1a/b MO: n=10/14), which is strongest in the double six1a/b knockdown. Scalebar = 100 µm. EXPRESSION / LABELING:
PHENOTYPE:
|
|
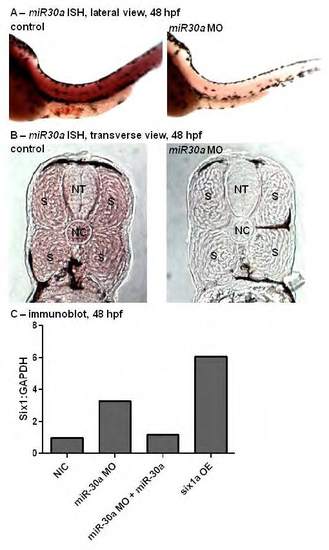
microRNA-30a targeting morpholino specificity confirmed by in situ hybridization and immunoblot analyses. In situ hybridization (ISH) analysis (A, control: n=17/17 total embryos, miR30a MO: n=14/18) for microRNA-30a (miR30a) at 48 hours post fertilization (hpf) reveals a decreased expression of miR30a upon injection of miR30a-targeting morpholino (MO). Transverse cross-sections at 18 µm of 48 hpf embryos stained by ISH for miR30a (B) confirm miR30a is expressed in the somites (S), (NT=neural tube, NC=notochord). Western blot analysis (C) quantified at 48 hpf following normalization to GAPDH demonstrates that miR30a-targeted MO injection into 1-4 cell stage embryos leads to an increase in Six1 protein expression, which is lost if miR30a is co-injected with miR30a MO. Further, six1a mRNA injection does result in increased levels of Six1 protein expression. |