- Title
-
Redundant roles of PRDM family members in zebrafish craniofacial development
- Authors
- Ding, H.L., Clouthier, D.E., and Artinger, K.B.
- Source
- Full text @ Dev. Dyn.
|
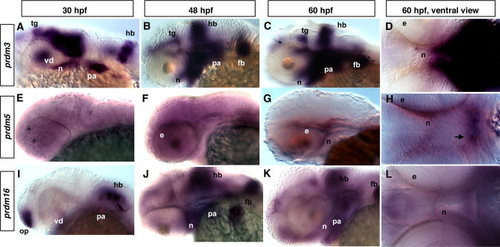
Expression of different prdms in developing zebrafish embryos. In situ hybridization (ISH) using prdm3, 5, and 16 at 30-hpf (A, E, I), 48-hpf (B, F, J), and 60-hpf stages showing lateral (C, G, K) and (D, H, L) ventral views. Note the partially overlapping expression of prdm3 and prdm16. A: Lateral view of whole mount embryo showing the prdm3 expression in the tegmentum, ventral diencephalon, neurocranium, hindbrain, pharyngeal arches at 30 hpf. At 48 hpf (B) and 60 hpf (C, D), prdm3 gradually increases and is highly expressed in pharyngeal arches, and is also expressed in pectoral fin buds, neurocranium, hindbrain, and tegmentum. E–H: prdm5 is not specifically expressed at 30 hpf, but is expressed at a low level in the area behind the eye and neurocranium at 48 and 60 hpf. The arrow in H points to the forming stomodeum in which prdm5 is expressed. I,J: From 30 to 48 hpf, prdm16 expression gradually increases in pharyngeal arches, as well as in neurocranium, pectoral fin buds, hindbrain, and olfactory placode. K,L: At 60 hpf, prdm16 expression decreases in the pharyngeal arches and hindbrain, but is still expressed at a significant level. Anterior is to the left in all panels. e, eye; fb, fin buds; hb, hindbrain; n, neurocranium; op, olfactory placode; pa, pharyngeal arch; tg, tegmentum; vd, ventral diencephalon. |
|
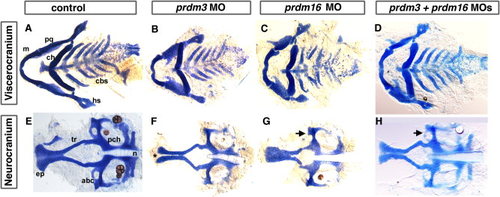
Reduction in prdm3 and prdm16 results in craniofacial defects. Five-dpf uninjected (A, E), prdm3 (B, F), prdm16 (C, G), and double morphant prdm3 and prdm16 (D, H) larvae stained with alcian blue to detect cartilage following dissection of the viscerocranium and neurocranium. As compared to control (A, E), the flat-mounted 5-dpf prdm3 i2e3 (B, F) and prdm16 e3i3 (C, G) morphant larvae have smaller palatoquadrate, including the pterygoid process of the palatoquadrate, and hyosymplectic of the viscerocranium, shortening of the Meckel′s cartilage (m), widening of the angle between ceratohyals (ch) in the viscerocranium, smaller ethmoid plate (ep), shortened trabeculae of the neurocranium, thinning and smaller of the neurocranium. In prdm3 morphant larvae (20–30%), there is a gap in the anterior edge of the ethmoid plate forming a “cleft” as shown in F. D, H: The combination of sub-optimal doses of prdm3 with prdm16 Morpholino. As compared to control (A, E), combination of 3 ng prdm3 i2e3 with 5 ng prdm16 e3i3 Morpholino (D,H) resulted in a slightly more severe phenotype in the neurocranium (much smaller and thinner neurocranium, smaller ethmoid plate, shortened trabeculae, missing anterior basicapsilar commissure, arrow). However, there is only a small gap in the ethmoid plate. Anterior is to the left. abc, anterior basicapsular commissure; cbs, ceratobranchials; ch, ceratohyal; ep, ethmoid plate; hs, hyosymplectic; m, Meckel′s cartilage; n, notochord; pch, parachordal; pq, palatoquadrate; tr, trabeculae; * indicates cleft in the ethmoid plate. PHENOTYPE:
|
|
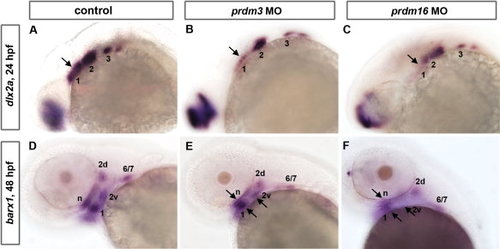
prdm3 and prdm16knockdown causes a reduction in the NCC expression domains of dlx2a at 24 hpf and barx1 at 48 hpf. A-C: Whole mount embryo ISH shows that pharyngeal arch marker dlx2a is expressed in the post-migratory neural crest region in control (A), prdm3 e3i3 (B), and prdm16 ATG (C) morphants at 24 hpf. dlx2a expression in prdm3 and prdm16 morphants shows a significant reduction in the anterior expression domain, and a mild reduction anteriorly relative to controls. D-F: barx1 expression in cranial NCC condensations is significantly reduced in the neurocranium (n) and arch 1 (1), dorsal and ventral domains within arch 2 (2d and 2v), and a domain representing arches 6 and 7 (6/7) in prdm3 (E) and prdm16 (F) morphants at 48 hpf as compared to controls (D). Arrows indicate notably reduced expression domains in prdm3 and prdm16 morphants. Anterior is to the left. |
|
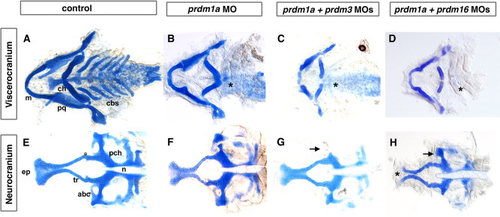
Combination of sub-optimal doses of prdm1a with prdm3 or prdm16 Morpholino results in severe craniofacial defects. Alcian blue staining on 5-dpf larvae to detect cartilage formation. Alcian blue–stained uninjected (A, E), prdm1a (B, F), prdm3, prdm1a-prdm3 double (C, G), and prdm1a-prdm16 double (D, H) morphant larvae. As compared to control (A), prdm1a knockdown alone has an inverted ceratohyal (ch) and missing ceratobranchial 2-5 (*) in the viscerocranium (B). In addition, prdm1a morphants have a smaller and narrower neurocranium and slightly shortened trabeculae (F). The combination of sub-optimal doses of prdm1a with prdm3 e3i3 Morpholino resulted in a more severe phenotype in viscerocranium (C, shortening of the Meckel′s cartilage and loss of ceratobranchial 2–5). In the neurocranium, the double morphants have an increasingly smaller and thinner neurocranium, a smaller ethmoid plate, and shorter trabeculae. The combination of suboptimal doses of prdm1a and prdm16 ATG Morpholino led to a more severe phenotype in the neurocranium (H, much smaller and thinner neurocranium with clefting observed in 20-30% of embryos, smaller ethmoid plate, shorter trabeculae) and viscerocranium (D, shortening of the Meckel′s cartilage and absence of ceratobranchial 2–5). Anterior is to the left. abc, anterior basicapsular commissure; cbs, ceratobranchials; ch, ceratohyal; ep, ethmoid plate; m, Meckel′s cartilage; n, notochord; pch, parachordal; tr, trabeculae; * in B–D illustrates missing cbs, in H indicates area of clefting. PHENOTYPE:
|
|
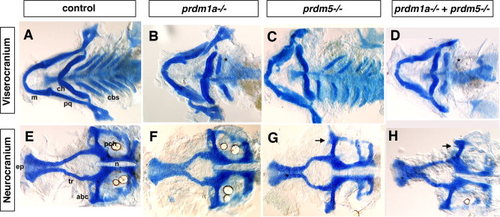
The viscerocranium and neurocranium of prdm5hi61Tg-prdm1am805 double mutant exhibit more severe phenotypes than single mutants. Double prdm1am805 prdm5hi61Tg heterozygotes were crossed to obtain the prdm5hi61Tg-prdm1am805 double mutant embryos. A-D: Flat-mounted Alcian blue staining of the viscerocranium and neurocranium in control, prdm1am805, prdm5hi61Tg mutant, and prdm5hi61Tg-prdm1am805 double mutant. As compared to control (A), the prdm1am805 (B) mutant larva is missing the ceratobranchial 2–5 cartilages (cb2–5) as was also observed in the prdm1a morphant. The prdm5hi61Tg (C) mutant larva has a slightly shortened Meckel′s cartilage (m) in the viscerocranium. The prdm5hi61Tg-prdm1am805 double mutants (D) have a more severe phenotype in the viscerocranium (shortening of the Meckel′s cartilage, greatly widening of the angle between ceratohyals [ch], and absence of ceratobranchials 2–5). E–H: Flat mount of the neurocranium in the control (E), prdm1am805 mutant (F), prdm5hi61Tg mutant (G), and the prdm5hi61Tg-narrowmindedm805 double mutant (H). The neurocranium and ethmoid plate (ep) are slightly smaller and trabeculae are shortened in prdm1am805 mutant and prdm5hi61Tg mutant (F,G). In prdm5-/-, the hypophyseal fenestra is significantly wider and the anterior basicapsilar cartilage is missing (G, arrow). The double mutant shows further reduction of the neurocranium, smaller ethmoid plate, shorter trabeculae, and missing basicapsilar cartilage (H, arrow). Anterior is to the left. abc, anterior basicapsular commissure; cbs, ceratobranchials; ch, ceratohyal; ep, ethmoid plate; m, Meckel′s cartilage; n, notochord; pch, parachordal; tr, trabeculae; * in B,D illustrates missing cbs. PHENOTYPE:
|