- Title
-
Loss of Lrp2 in zebrafish disrupts pronephric tubular clearance but not forebrain development
- Authors
- Kur, E., Christa, A., Veth, K.N., Gajera, C.R., Andrade-Navarro, M.A., Zhang, J., Willer, J.R., Gregg, R.G., Abdelilah-Seyfried, S., Bachmann, S., Link, B.A., Hammes, A., and Willnow, T.E.
- Source
- Full text @ Dev. Dyn.
|
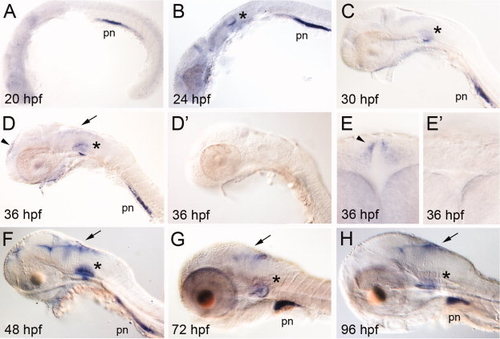
A–H:lrp2 expression in early zebrafish development. Whole mount in situ hybridization (ISH) for lrp2 in zebrafish embryos at the indicated time points. The asterisks mark expression in the otic vesicle. Expression in the developing CNS is highlighted by arrows. The arrowheads indicate lrp2 expression domains in the telencephalon at 36 hpf in lateral (D) and frontal (E) head aspects. D2, E2: ISH using the sense probe as negative control to the respective images shown in D and E. pn, pronephros. EXPRESSION / LABELING:
|
|
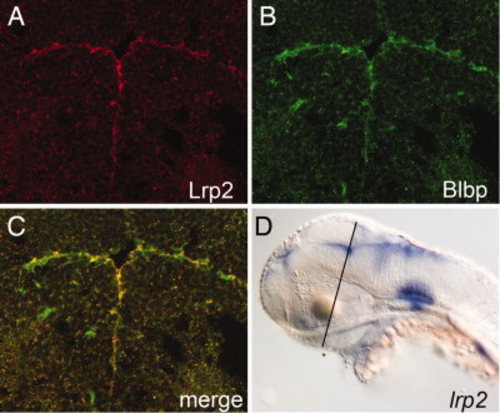
Expression of Lrp2 in the ventricular system of the brain at 48 hpf. A–C: Immunohistology on coronal forebrain sections for Lrp2 (A) and brain lipid-binding protein (Blbp; B) demonstrates co-localization of both proteins in radial glia cells lining the ventricular system (merge; C). D: Whole mount in situ hybridization for lrp2 indicating the plane of section through the diencephalon shown in A–C. EXPRESSION / LABELING:
|
|
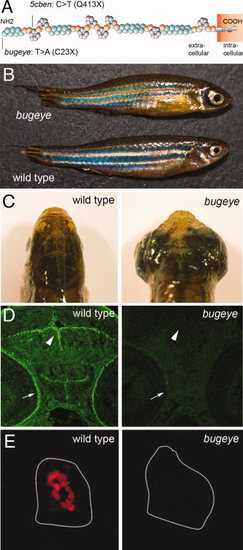
Loss of Lrp2 expression in bugeye mutants. A: Structure of Lrp2 indicating amino acid changes in bugeye and 5cben lines. B, C: Appearance of adult wild type and bugeye zebrafish in lateral (B) and dorsal (C) views highlighting the large eye phenotype in mutants. D: Immunohistological detection of Lrp2 (green) in retinal pigment epithelium (arrow) and ventricular system (arrowhead) in wild type but not in bugeye embryos at 48 hpf using polyclonal antisera directed against multiple epitopes in the extracellular domain of the receptor polypeptide. E: Loss of Lrp2 expression (red) in immunohistological sections of the pronephros in 4-dpf-old bugeye larvae. White lines indicate the position of the pronephric ducts. Complete loss of Lrp2 was also documented in 5cben (data not shown). |
|
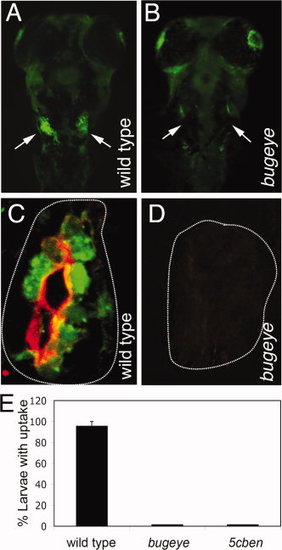
Lrp2 deficiency disrupts clearance pathways in the pronephric duct. A, B: Whole mount fluorescence microscopy detects accumulation of fluid phase marker FITC-dextran (green) in the wild type but not in the Lrp2-deficient pronephros (arrows in A and B) upon injection into the common cardinal vein. C, D: Immunohistological detection of Lrp2 (red) and FITC-dextran (green) in the pronephros of the indicated genotypes. White lines highlight the position of the pronephric ducts. E: Wild type as well as bugeye and 5cben larvae at 4 dpf were injected with FITC-dextran and the number of larvae with tubular accumulation of tracers evaluated by fluorescence microscopy. Data are given as % ± SEM of all larvae injected. N= 53 (wild type), 15 (bugeye), or 23 (5cben). PHENOTYPE:
|
|
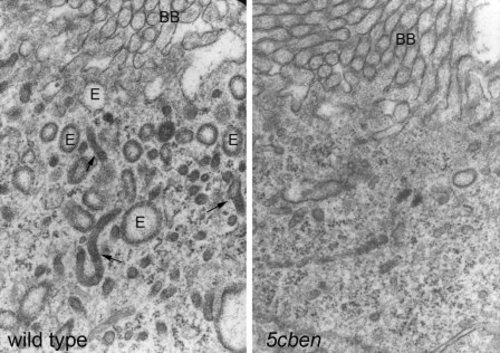
Loss of endocytic apparatus in Lrp2-deficient pronephros. Electronmicroscopical analysis documents almost complete loss of the endocytic apparatus including endosomes (E) and dense apical tubules (arrows) in the 5cben pronephros compared with the wild type control at 4 dpf. BB, brush border of tubular epithelium. PHENOTYPE:
|
|
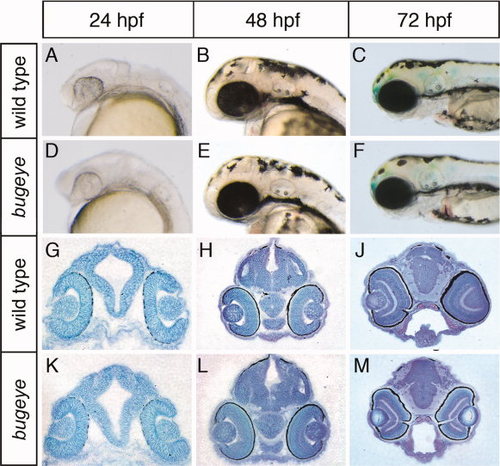
Appearance of head and brain structures in bugeye embryos. Lateral head aspects (A–F) and coronal diencephalic sections (G–M) stained with toluidine blue are shown. No obvious malformations in head or brain anatomy are seen at the indicated time points comparing wild type and bugeye embryos. PHENOTYPE:
|
|
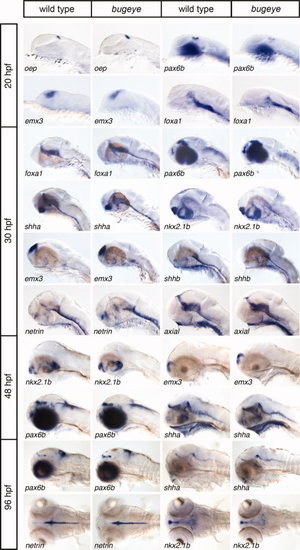
Analysis of forebrain development in bugeye embryos. Whole mount in situ hybridization of wild type and bugeye embryos at 20, 30, 48, and 96 hpf using the indicated markers of forebrain development. Lateral or dorsal head aspects are shown. EXPRESSION / LABELING:
|
|
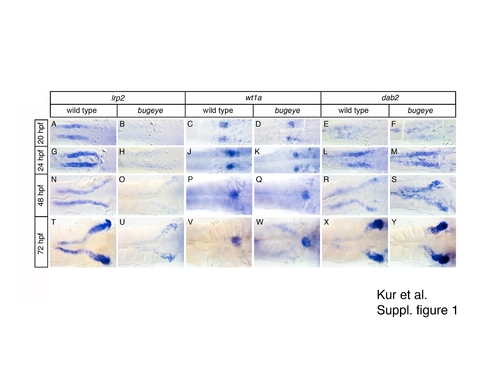
Loss of lrp2 does not affect development of the larval pronephros. Whole mount in situ hybridization for lrp2, wt1a, and dab2 in wild type and bugeye embryos at the indicated time points. EXPRESSION / LABELING:
|
|
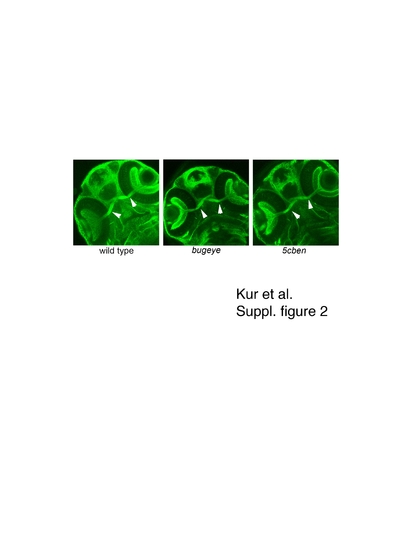
Formation of optic chiasm in wild type and mutant zebrafish embryos. Immunohistolological detection of Zn5 visualizes the optic chiasm (arrowhead) in wild type and mutant embryos at 53 hpf. EXPRESSION / LABELING:
|
|
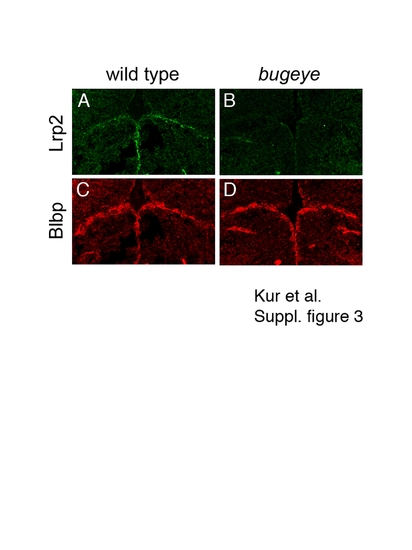
Appearance of radial glia in the ventricular system of wild type and mutant zebrafish embryos. Immunohistology on coronal forebrain sections for Lrp2 (A, C) and brain lipid-binding protein (Blbp; B; D) in wild type and bugeye embryos at 48 hpf. Lack of Lrp2 expression in bugeye (C) does not affect the appearance of radial glia lining the ventricular system as shown by staining for Blbp (D). EXPRESSION / LABELING:
|
|
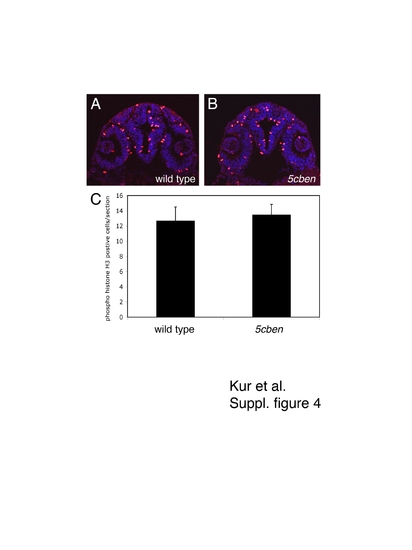
Quantification of proliferation in the developing zebrafish forebrain. (A, B) Immunohistological detection of phospho-histone H3 (pH3) in the diencephalon of wild type and 5cben embryos at 24 hpf. (C) Quantification of the number of pH3 positive cells per section (± SEM). n=3-6 sections each of a total of 10 embryos per genotype. |
|
ATG morpholino knockdown of lrp2b in bugeye. Bugeye embryos were injected with buffer (A, D) or with two different lrp2b ATG morpholinos (lrp2bMO1; lrp2bMO2) (B - F). In addition, all embryos received a test RNA encoding mCherry targeted by lrp2bMO1 and lrp2bMO2. Successful knockdown of mCherry expression was evaluated 3 hr after injection by whole mount fluorescence microscopy (A-C). Proper formation of head structure was documented at 24 hpf in lateral head aspects (D – F). PHENOTYPE:
|

Unillustrated author statements |