- Title
-
Gli2a protein localization reveals a role for Iguana/DZIP1 in primary ciliogenesis and a dependence of Hedgehog signal transduction on primary cilia in the zebrafish
- Authors
- Kim, H.R., Richardson, J., van Eeden, F., and Ingham, P.W.
- Source
- Full text @ BMC Biol.
|
A Gli2a-GFP fusion protein is capable of rescuing adaxial F59 expression in you-too (yot) mutant embryos. Newly fertilised eggs from a yot/+ incross were injected with modified BAC DNA containing GFP tagged Gli2a. The injected embryos were fixed at the 12 somite stage and labelled with anti-F59 (red) in order to detect slow lineage muscle fibres (a), the expression of which is eliminated in yot mutant embryos (b). F59 expression was restored specifically in the Gli2a-GFP expressing cells of transient transgenic mutant embryos (c), indicating that GFP tagged Gli2a protein is functional. Dorsal view, anterior to the left. Nuclei: DAPI in blue. Scale bar: 20 μm |
|
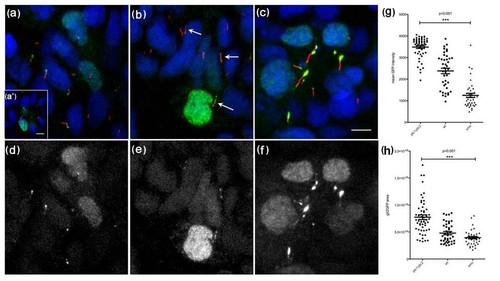
Gli2a-GFP localizes to the distal tips of primary cilia in paraxial mesodermal cells (18 somite stage) and its localization is modulated by the activity of Hedgehog signalling. Gli2a-GFP injected embryos were labelled with mAb acetylated tubulin (red in a, b and c) and gamma tubulin (red in a′). This revealed that Gli2a-GFP was localized at the distal tip of primary cilia (a) and that its expression was excluded from the basal bodies (a′). In smoothened (smo) mutant embryos, levels of Gli2a-GFP were diminished (b) and the signal was dispersed along the cilia or localized both at the distal tip and at the basal bodies (arrows). In ptc1:ptc2 double mutant embryos, by contrast, high levels of Gli2a-GFP accumulated at the distal tip of the cilia (c). Panels (d-f) show the green channel images of (a-c), respectively. The intensity and the area of Gli2a-GFP at the tip of the cilia were measured in wild type and mutant embryos and the difference between the three groups was analysed by one way ANOVA test. This revealed a significant difference in protein levels between wild type, smo and ptc1;ptc2 mutant groups with P < 0.001 (g and h). Scale bars: 5 μm |
|
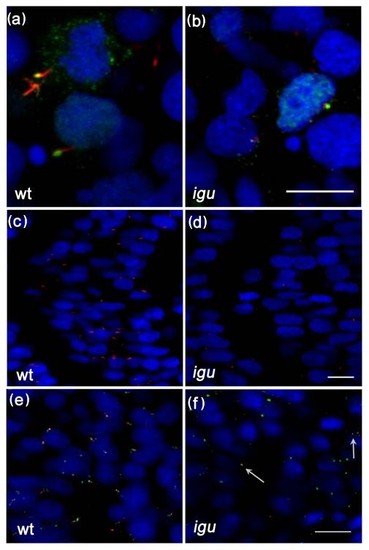
Primary cilia are truncated or missing from somitic cells in Iguana (igu) mutant embryos. Gli2a-GFP co-localized with acetylated tubulin (red) in igu mutant embryos in presumptive axonemes (b) that were severely truncated relative to those of wild type primary cilia (a). Truncated cilia (red) were only occasionally present in igu embryos (d) in contrast to wild type (c). Basal bodies (green: γ-tubulin in e, f) were present in all cells in igu mutant embryos (f) as in wild type (e) and were associated with the truncated axonemes (arrows). Scale bars: 10 μm |
|
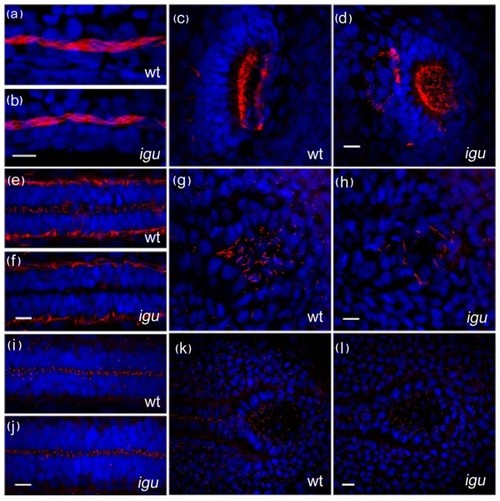
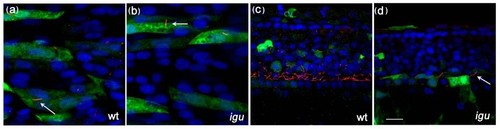
Motile cilia are largely unaffected by the loss of Iguana (Igu) function. Motile cilia (red: acetylated tubulin in a-h) in the pronephros at 48 hpf in igu mutant embryos appeared normal (b), as did those in the olfactory pits (d). By contrast motile cilia were largely absent from the floorplate at 28 hpf (f) and those in Kupffer′s vesicle were significantly reduced in number (h) in 10 somite stage igu mutant embryos. Basal bodies (red: γ-tubulin in i-l) in the igu mutants, by contrast, were formed normally at all stages examined in the floorplate (j) and Kupffer′s vesicle (l). Scale bars: 10 μm |
|
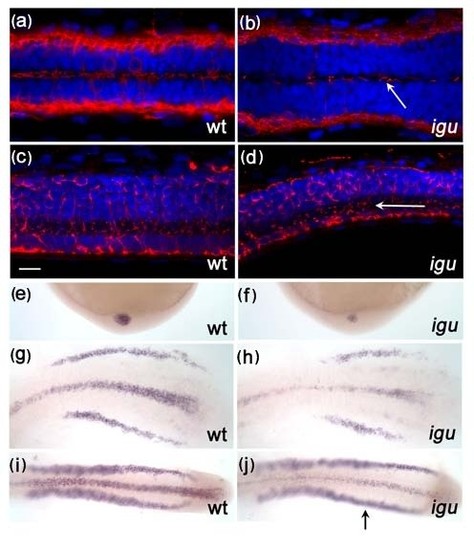
Motile cilia in the floorplate recover in Iguana (igu) mutant by 5 dpf. A few motile cilia (red: acetylated tubulin) were apparent in the floorplate of igu mutants at 48 hpf (b, arrow), and were almost completely recovered by 5 dpf (d, arrow). In situ hybridization reveals that foxj1a expression is down-regulated in Kupffer′s vesicle (f) and the floor plate (h, j) at the end of gastrulation and early somitogenesis (h:8 somite, j:14somite), respectively. In contrast, expression in the pronephros was normal (j, arrow). Scale bar: 10 μm. Dorsal view: a-b & g-j, lateral view: c-f EXPRESSION / LABELING:
|
|
Ectopic expression of foxj1a in somites results in ectopic motile cilia formation in both wild-type and mutant embryos and rescues motile cilia formation in the floorplate of Iguana (igu) mutant embryos. Ectopic motile cilia were induced in somitic cells expressing foxj1a (marked by green fluorescent protein expression) both in wild type and igu mutant embryos (a and b, arrows). Motile cilia were restored in the floorplate of igu mutant embryos in cells expressing foxj1a (d, arrow). Scale bar: 10 μm |
|
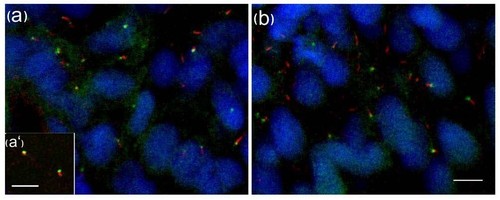
GFP-tagged Iguana (Igu) localizes to basal bodies of primary cilia in zebrafish embryos. GFP-igu mRNA was injected into 1-cell stage embryos and injected embryos fixed and labelled with anti-acetylated tubulin (red). Punctate GFP-Igu localization was excluded from the axoneme (a). Co-injection of mRNA encoding tdTomato fused GTAP3 (γ-tubulin associated protein 3) and GFP-Igu, revealed co-localization of both proteins at the basal bodies in live early somitogenesis stage embryos (a′). The basal body localization of GFP-Igu was un-altered in embryos expressing dnPKA (b). Scale bars: 5 μm |
|
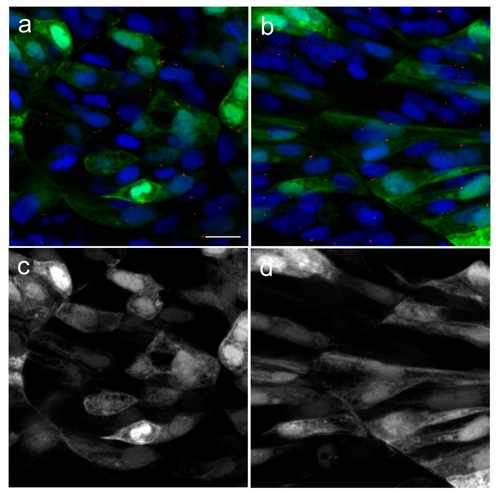
Green fluorescent protein (GFP) does not localize to primary cilia. In order to confirm that GFP alone does not localize to the primary cilia, we cloned GFP into pCS2 vector and injected into newly fertilised zebrafish eggs. Injected embryos were fixed at 18 hpf. Primary cilia and nuclei were visualised with acetylated tubulin (red) and DAPI (blue) respectively. GFP can be seen to localize to the cytoplasm and nuclei in both slow (b) and fast muscle (a) cells, but was not detected in the primary cilia. Green channels from (a) and (b) are shown in (c) and (d), respectively. Scale bar:10 μm |

Unillustrated author statements |