- Title
-
Loss of Dnmt1 catalytic activity reveals multiple roles for DNA methylation during pancreas development and regeneration
- Authors
- Anderson, R.M., Bosch, J.A., Goll, M.G., Hesselson, D., Dong, P.D., Shin, D., Chi, N.C., Shin, C.H., Schlegel, A., Halpern, M., and Stainier, D.Y.
- Source
- Full text @ Dev. Biol.
|
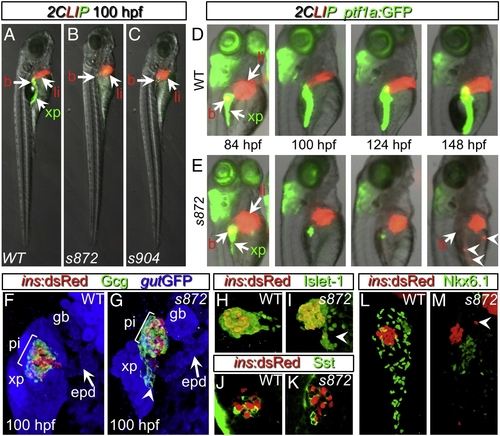
Initial formation and degeneration of the exocrine pancreas in dandelion (ddn) mutants. (A–C) Lateral views of WT (A), ddns872 (B), and ddns904 mutant (C) larvae at 100 hpf in the 2-color Liver, Insulin, acinar Pancreas (2CLIP) transgenic background (see Materials and methods). Acinar cells of the exocrine pancreas (xp) are green and hepatocytes (li) and pancreatic beta cells (b) are red. ddn mutants exhibit minimal acinar tissue, but overall larva morphology is only moderately affected. (D, E) Successive images of individual WT (D) and ddns872 mutant (E) larvae between 84 and 148 hpf in the 2CLIP; Tg(ptf1a:EGFP)jh1 background. The exocrine pancreas and liver grow larger in WT, but degenerate in ddn mutants. Hepatocyte fragments are found in the circulation (arrowheads). (F–K) 3-D projections of confocal stacks showing composition of WT (F, H, J) and ddns872 mutant (G, I, K) endocrine pancreas at 100 hpf in Tg(ins:dsRed)m1081 background. (F, G) A core of Tg(ins:dsRed)m1081+ cells (red) is surrounded by a mantle of Glucagon+ (Gcg) cells (green). Extrapancreatic duct (epd) structure is intact. (H, I) Isl-1, and (J, K) Somatostatin (Sst) positive cells appear unaffected. Arrowheads in G, I, and M point to endocrine cells outside of the primary islet. (L, M) Nkx6.1 immunostaining reveals the presence of intra-pancreatic duct cells in both WT and dnmt1s872 mutant larvae. EXPRESSION / LABELING:
PHENOTYPE:
|
|
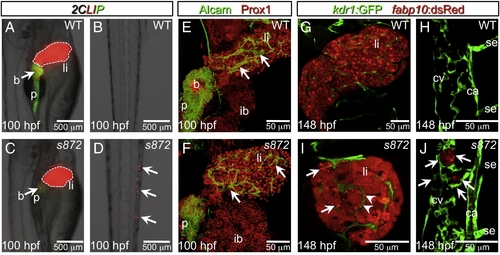
Liver degeneration in dandelion (ddn) mutants. (A–D) Lateral aspect of trunk (A, C) and tail (B, D) regions of 100 hpf WT (A, B) and ddn mutant (C, D) larvae in the 2CLIP transgenic background. The liver is smaller in ddn mutants relative to WT, and cell fragments emitting dsRed fluorescence, derived from the liver, aggregate in clusters in the tail (D, arrows). (E, F) 3-D projections of confocal stacks showing Alcam and Prox1 antibody staining of 100 hpf WT (E) and ddn mutants (F). Initial specification, morphogenesis, and differentiation of the liver appear to be unaffected. Intrahepatic ductal network is indicated by arrows. (G–J) Confocal slices through the liver (G, I) and tail vasculature (H, J) of Tg(kdrl:GFP)s843; Tg(fabp10:dsRed)gz12 WT (G, H) and ddn mutant (I, J) larvae at 6 dpf. Although never present in WT, bright dsRed+ degenerating hepatocyte fragments are observed throughout the ddn mutant liver (I, arrows) and within the hepatic vasculature (I, arrowheads). These particles accumulate in the network of the caudal vein (cv; J, arrows). Other abbreviations: ca, caudal aorta; se, intersegmental vessel; ib, intestinal bulb; p, pancreas; li, liver; b, beta cells. |
|
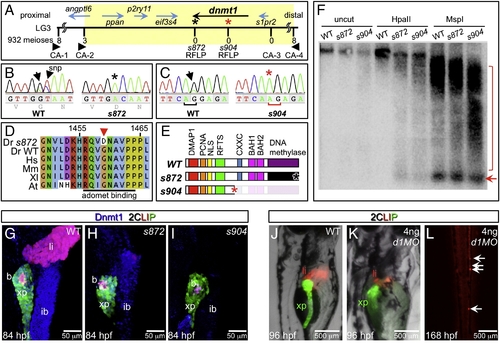
The dandelion phenotype is caused by mutations in dnmt1. (A) Genetic map of the ddn region on linkage group 3; the numbers above the SSLP/RFLP markers indicate the number of recombinants in 932 meioses analyzed. Six genes lie within the critical region (shaded). (B) Sequence traces of pooled phenotypically WT and pooled dnmt1s872 mutant cDNAs; a G to A transversion at nucleotide 4376 results in a G1459D substitution in Dnmt1. (C) Sequence traces of pooled phenotypically WT and pooled dnmt1s904 mutant genomic DNA; a mutation in the exon 15 splice acceptor results in a single base pair frameshift and premature termination following 31 missense substitutions. (D) Peptide sequence alignment of D. rerio (Dr) Dnmt1 from dnmt1s872 mutant and WT with H. sapiens (Hs), M. musculus (Mm), X. laevis (Xl), and A. thaliana (At). (E) Domain map of WT and ddn alleles of Dnmt1. (F) Southern blot of WT and dnmt1s872 and dnmt1s904 mutant genomic DNA either uncut or digested with methylation-sensitive HpaII or methylation insensitive MspI, and hybridized with a radiolabeled probe to the DANA consensus sequence. Appearance of DANA band (arrow) in dnmt1 mutant DNA digested with HpaII indicates hypomethylation of this transposable element, while smearing of high molecular weight DNA indicates widespread genomic hypomethylation (compare bracketed regions). (G–I) Dnmt1 antibody staining in 2CLIP transgenic line. Dnmt1 (blue) is observed in endodermal organs of WT (G) and dnmt1s872 mutants (H), but not dnmt1s904 mutants (I). (J–L) Injection of 4 ng of dnmt1 morpholino (d1MO) into the 2CLIP line phenocopies ddn mutants. Compared to WT (J), d1MO-injected larvae (K) show smaller mass of differentiated pancreatic acinar cells (green) and hepatocytes (red) at 96 hpf. Ectopic dsRed fluorescence is observed in the posterior vasculature of d1MO-injected larvae at 168 hpf (L). EXPRESSION / LABELING:
|
|
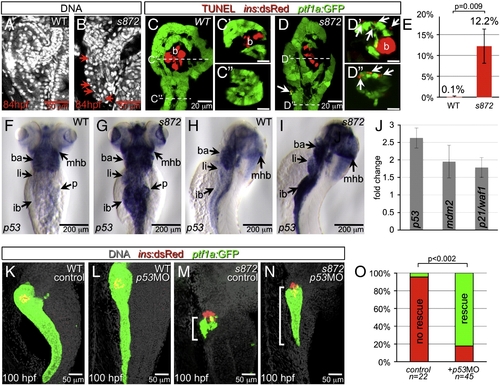
Acinar cell death in dnmt1 mutants occurs in part by p53-dependent apoptosis. (A, B) DRAQ5 staining of DNA in 84 hpf WT (A) and dnmt1s872 mutant (B) larvae. Pyknotic nuclei (arrows) are frequently observed in the pancreas of dnmt1 mutants, but not WT. (C, D) Apoptosis detection by TUNEL labeling of 84 hpf WT (C) and dnmt1 mutant (D) larvae in Tg(ptf1a:EGFP)jh1; Tg(ins:dsRed)m1081 background. C′/C″ and D′/D″ are transverse sections of similar samples at the approximate planes indicated in panels C and D, respectively. (E) Quantification of TUNEL+ acinar cells. (F–I) Expression of p53 mRNA in WT (F, H) and dnmt1s872 mutants (G, I); expression appears dramatically increased in mutants. (J) Fold-change of p53 and target genes mdm2 and p21/waf1 in dnmt1s872 mutants vs. WT by real-time RT-PCR. (K–N) 100 hpf WT (K, L) and dnmt1s872 mutant (M, N) larvae, which are either uninjected controls (K, M) or p53 morpholino-injected (p53MO; L, N). p53MO-injected dnmt1s872 mutants show increased persistence of acinar tissue. (O) More than 82% of p53MO-injected dnmt1s872 mutants showed rescue (green); n = 22 (control), n = 45 (p53MO). Error bars = SEM. Significance assessed by Student′s t-test. EXPRESSION / LABELING:
PHENOTYPE:
|
|
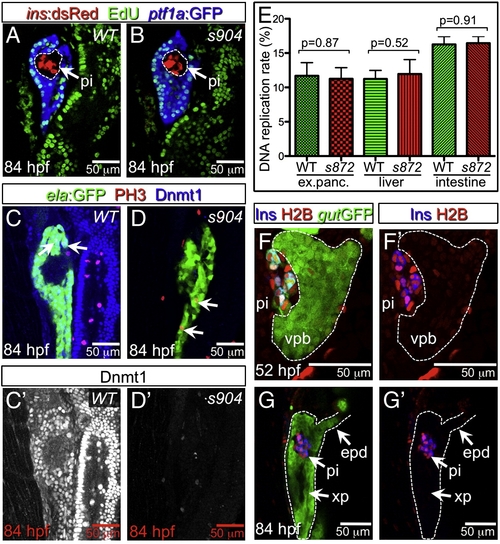
Lack of Dnmt1 activity does not arrest the cell cycle. (A, B) Confocal planes showing EdU incorporation in 84 hpf WT (A) and dnmt1 mutant (B) Tg(ptf1a:EGFP)jh1; Tg(ins:dsRed)m1081 larvae. (C, D) Phospho-histone 3 (late G2 interphase through anaphase marker) and Dnmt1 staining of 84 hpf WT and dnmt1s904 mutant Tg(fabp10:DsRed; ela:GFP)gz12 larvae. (C′, D′) Dnmt1 channel only. (E) EdU incorporation rate in pancreatic acinar cells, hepatocytes, and intestine of dnmt1 mutants appears indistinguishable from WT (p = 0.87, p = 0.52, p = 0.91, respectively). Error bars = SEM. Significance assessed by Student′s t-test. (F, G) H2BRFP label retention analysis of endodermal organ forming region in WT Tg(gut:GFP)s854 animals at 52 (F) and 84 (G) hpf. The ventral pancreatic bud (vpb) is demarcated by a dashed line. H2BRFP is diminished in the highly proliferative vpb relative to the dorsal pancreatic bud during outgrowth (F) and pancreas tail formation (G). (F′, G′) H2BRFP (red) and Ins (blue) channels only. Primary islet (pi). EXPRESSION / LABELING:
PHENOTYPE:
|
|
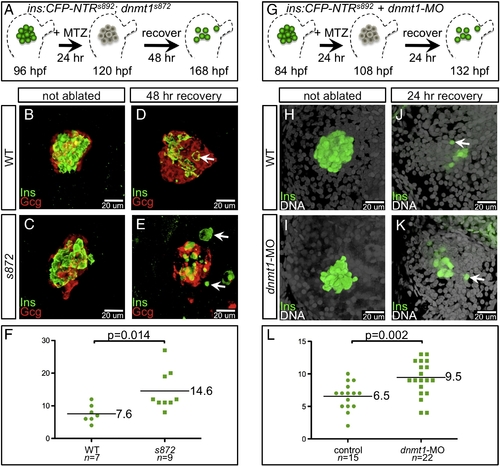
Enhanced recovery of beta cell mass in dnmt1 mutants and morphants. (A) Pancreatic beta cell ablation scheme in dnmt1s872 mutants. 96 hpf Tg(ins:CFP-NTR)s892 WT and dnmt1 mutant larvae were exposed to MTZ for 24 h , then washed for 48 h and analyzed at 168 hpf. (B–E) Three-dimensional projections of the primary islet at 168 dpf in WT (B, D) and dnmt1 mutant (C, E) larvae that were untreated (B, C), or ablated with MTZ (D, E). (B, C) WT and dnmt1 mutant islets have a core of Ins+ beta cells surrounded by a mantle of Gcg+ alpha cells. (D) In recovering WT larvae, new beta cells are observed in the islet (arrow). (E) In recovering dnmt1 mutant islets, nearly double the number of beta cells is present, though some appear morphologically abnormal (arrows; compare with panel D). (F) 7.6 ± 1.0 beta cells were observed in recovering WT islets (n = 7), and 14.6 ± 2.0 beta cells were observed in recovering dnmt1 mutant larvae (n = 9; p = 0.014). (G) Pancreatic beta cell ablation scheme in Tg(ins:CFP-NTR)s892 embryos injected with 2 ng of dnmt1 morpholino (dnmt1-MO). 84 hpf larvae were exposed to MTZ for 24 h , then washed for 24 h and analysed at 132 hpf. (H–K) Three-dimensional projections of the primary islet at 132 hpf in WT (H, J) and dnmt1-MO injected (I, K) larvae that were untreated (H, I), or ablated with MTZ (J, K). (K) dnmt1-MO injected larvae showed a greater degree of beta cell recovery. (L) 6.5 ± 0.5 beta cells were observed in recovering WT larvae (n = 15), and 9.5 ± 0.6 beta cells were observed in recovering dnmt1-MO injected larvae (n = 22; p = 0.002). Error bars = SEM. Significance assessed by Student′s t-test. EXPRESSION / LABELING:
|
Reprinted from Developmental Biology, 334(1), Anderson, R.M., Bosch, J.A., Goll, M.G., Hesselson, D., Dong, P.D., Shin, D., Chi, N.C., Shin, C.H., Schlegel, A., Halpern, M., and Stainier, D.Y., Loss of Dnmt1 catalytic activity reveals multiple roles for DNA methylation during pancreas development and regeneration, 213-223, Copyright (2009) with permission from Elsevier. Full text @ Dev. Biol.