- Title
-
Definitive hematopoietic stem/progenitor cells manifest distinct differentiation output in the zebrafish VDA and PBI
- Authors
- Jin, H., Sood, R., Xu, J., Zhen, F., English, M.A., Liu, P.P., and Wen, Z.
- Source
- Full text @ Development
|
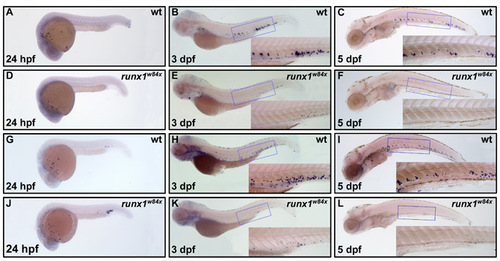
Definitive erythroid cells arise in the PBI but not the VDA. (A-K) Whole-mount in situ hybridization to detect βe1-globin expression in wild-type embryos (A-E) at 22 hpf (A), 2 dpf (B), 3 dpf (C), 4 dpf (D) and 5 dpf (E); runx1w84x mutant (F,G) and morphant (H) embryos at 22 hpf (F) and 4 dpf (G,H); and vltm651 embryos (I-K) at 22 hpf (I), 2 dpf (J) and 4 dpf (K). The blue arrow in E indicates βe1-globin expression in pronephros. The insets are higher magnification (20x) views of the corresponding boxed regions (blue). EXPRESSION / LABELING:
PHENOTYPE:
|
|
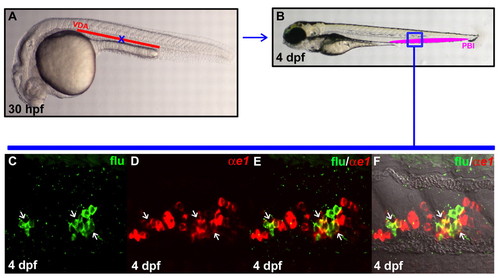
VDA originated HSPCs are capable of giving rise to definitive erythroid cells once homed to the PBI. (A) Lateral view of 30 hpf embryo indicates the uncaged position (blue cross) in the anterior part of VDA. (B) Lateral view of 4 dpf embryo. The boxed region (blue) indicates the relative position in the PBI where flu and βe1-globin RNA double-positive cells are found after uncaging. (C,D) Confocal images of the boxed region in B show the flu signal and βe1-globin RNA staining in the PBI. (E) Merged image of C and D. (F) Superimposed view of E and DIC image. White arrows indicate the co-staining of flu and βe1-globin RNA. EXPRESSION / LABELING:
|
|
Enrichment of definitive myeloid cells in the VDA and the PBI. (A-H) Whole-mount in situ hybridization of l-plastin expression in wild-type embryos at 24 hpf (A), 2 dpf (C), 3 dpf (E) and 5 dpf (G), and runx1w84x mutant embryos at 24 hpf (B), 2 dpf (D), 3 dpf (F) and 5 dpf (H). Insets are high magnification (20X) of the corresponding boxed regions (blue). |
|
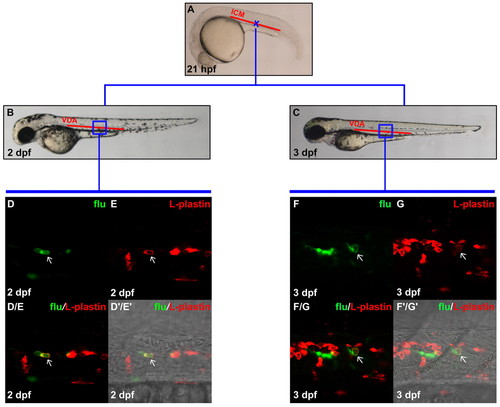
Definitive myeloid cells are generated in the VDA. (A) Lateral view of 21 hpf embryo, indicating the uncaging position (blue cross) in the anterior part of the ICM. (B,C) Lateral view of 2 dpf (B) and 3 dpf (C) embryos. The boxed regions indicate the relative positions in the VDA where flu and L-plastin protein double-positive cells were detected after uncaging. (D,E) Confocal images of the boxed region in B showing the flu signal and L-plastin staining in the VDA at 2 dpf after uncaging at cross in A. (D/E) Merged image of D and E. (D′/E′) Superimposed view of D/E and DIC image. (F,G) Confocal images of the boxed region in C showing the flu signal and L-plastin staining in the VDA at 3 dpf after uncaging at cross in A. (F/G) Merged image of F and G. (F′/G′) Superimposed view of F/G and DIC image. Staining for flu and L-plastin is pseudopainted as green and red, respectively. Arrows indicate co-staining of flu and L-plastin. EXPRESSION / LABELING:
|
|
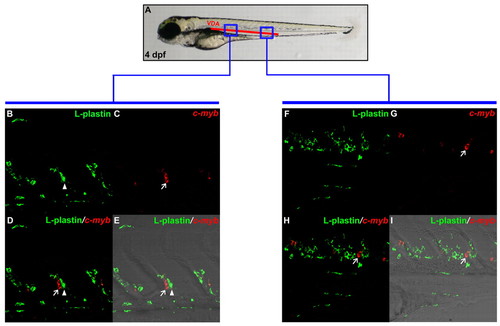
HSPCs are found in the VDA in 4 dpf wild-type embryos. (A) Schematic illustration of 4 dpf embryo, indicating blue boxed regions that are magnified in B-E and F-I. (B,C) Double staining of L-plastin protein (B) and cmyb RNA (C) in anterior VDA portion of 4 dpf wild-type embryo. (D) Superimposed image of B and C. (E) Merged view of D with DIC image. (F,G) Double staining of L-plastin protein (F) and cmyb RNA (G) in posterior VDA region of 4 dpf wild-type embryo. (H) Superimposed image of F and G. (I) Merged view of H with DIC image. Arrows indicate HSPCs that are cmyb-positive only, whereas arrowheads represent L-plastin-positive myeloid cells. EXPRESSION / LABELING:
|
|
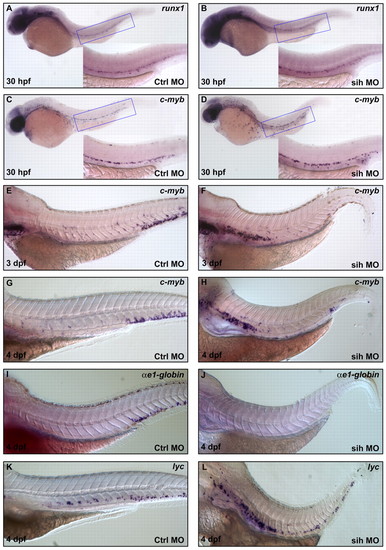
Inhibition of HSPC migration from the VDA to PBI blocks their differentiation into erythroid but not myeloid lineages. (A,B) Whole-mount in situ hybridization of runx1 expression in 30 hpf control MO injected vltm651 embryos (A) and sih MO injected vltm651 embryos (B). (C-H) Whole-mount in situ hybridization of cmyb expression in control MO-injected vltm651 embryos at 30 hpf (C), 3 dpf (E) and 4 dpf (G); sih MO injected vltm651 embryos at 30 hpf (D), 3 dpf (F) and 4 dpf (H). (I,J) Whole-mount in situ hybridization of βe1-globin expression in 4 dpf control MO injected vltm651 embryos (I) and sih MO-injected vltm651 embryos (J). (K,L) Whole-mount in situ hybridization of lyc expression in 4 dpf control MO-injected vltm651 embryos (K) and sih MO-injected vltm651 embryos (L). Insets are higher magnification (20x) views of the corresponding boxed regions (blue). EXPRESSION / LABELING:
PHENOTYPE:
|
|
Circulating definitive proerythroblasts are present in the bloodstream from 4 dpf onwards. (A,B) May-Grunwald Giemsa staining of proerythroblasts isolated from 5 dpf bloodstream (A) and adult kidney marrow (B). (C) The profile of definitive proerythroblasts present in the blood during various developmental stages. The vertical axis indicates the percentage of definitive proerythroblasts in the blood. The horizontal axis indicates the stage of embryos sacrificed for analysis. The blue bars represent wild-type embryo. The green bars represent runx1 morphant. For counting the number of definitive proerythroblasts, circulating blood cells are pooled from 30 embryos of 30 hpf and 20 embryos of the other stages. |
|
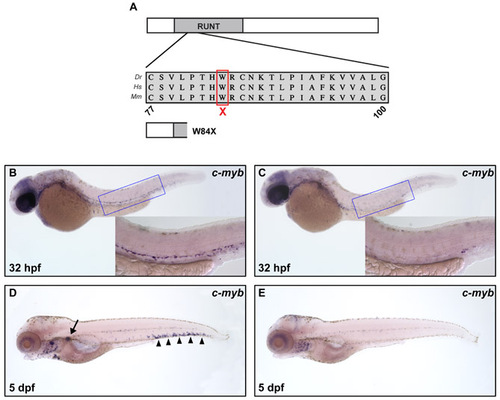
Characterization of the runx1 null mutation W84X. (A) Schematic representation of Runx1 protein with runt homology domain marked and multispecies alignment of the region containing the mutation W84X shown by red box. A schematic of the predicted mutant protein is depicted underneath. Abbreviations: Dr, Danio rerio; Hs, Homo sapiens; Mm, Mus Musculus. (B-E) Whole-mount in situ hybridization of cmyb expression in 32 hpf wild-type (B), 32 hpf runx1w84x mutant (C), 5 dpf wild-type (D) and 5 dpf runx1w84x mutant (E) embryos. The insets are high magnification (20x) of the corresponding boxed regions (blue). Black arrow and arrowheads indicate cmyb expression in pronephros and PBI, respectively. |
|
Expression of lyc and mpo in wild-type and runx1w84x mutant embryos. (A-F) Whole-mount in situ hybridization of lyc expression in 24 hpf (A), 3 dpf (B) and 5 dpf (C) wild-type embryos, and 24 hpf (D), 3 dpf (E) and 5 dpf (F) runx1w84x embryos. (G-L) Whole-mount in situ hybridization of mpo expression in 24 hpf (G), 3 dpf (H) and 5 dpf (I) wild-type embryos, and 24 hpf (J), 3 dpf (K) and 5 dpf (L) runx1w84x embryos. Inserts are higher magnification views of boxed regions (blue). |
|
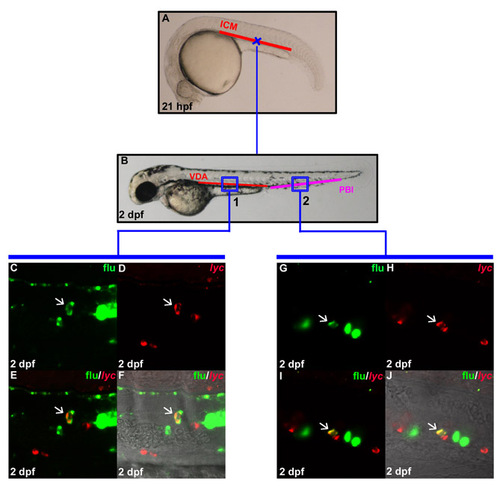
In situ generation of lyc positive myeloid cells in the VDA. (A) Lateral view of 21 hpf embryo indicates the uncaging positions (cross) in the anterior part of the ICM. (B) Lateral view of 2 dpf embryo. The boxed regions show the relative positions in the VDA (region 1) and PBI (region 2) where flu and lyc RNA double-positive cells are found after uncaging. (C,D) Confocal images of the boxed region 1 in B show the flu signal and lyc staining in the VDA at 2 dpf after uncaging at cross A. (E) Merged image of C and D. (F) Superimposed view of E and DIC image. (G,H) Confocal images of the boxed region 2 in B show the flu signal and lyc RNA staining in the PBI at 2 dpf after uncaging at cross in A. (I) Merged image of G and H. (J) Superimposed view of I and DIC image. White arrows indicate the co-staining of flu and lyc RNA. |