- Title
-
Altered brain-wide auditory networks in a zebrafish model of fragile X syndrome
- Authors
- Constantin, L., Poulsen, R.E., Scholz, L.A., Favre-Bulle, I.A., Taylor, M.A., Sun, B., Goodhill, G.J., Vanwalleghem, G.C., Scott, E.K.
- Source
- Full text @ BMC Biol.
|
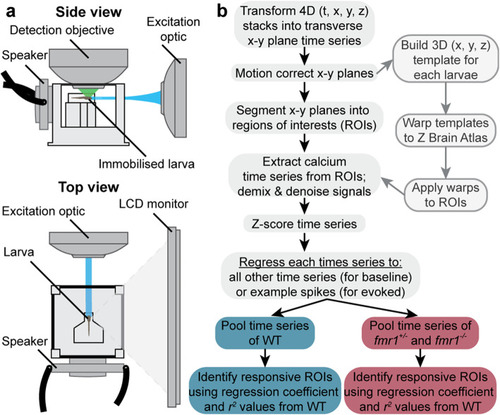
Imaging setup and neuroinformatic workflow. Schematic of the imaging setup ( |
|
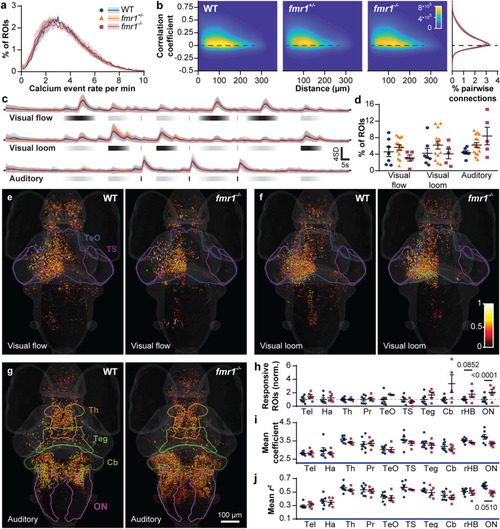
Brain-wide baseline and sensory-evoked neuronal activity. Distribution of brain-wide calcium event rates ( |
|
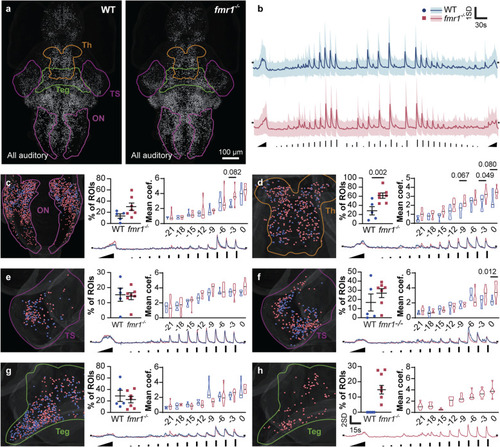
Regional responses to a complex auditory stimulus train. Auditory responsive ROIs ( |
|
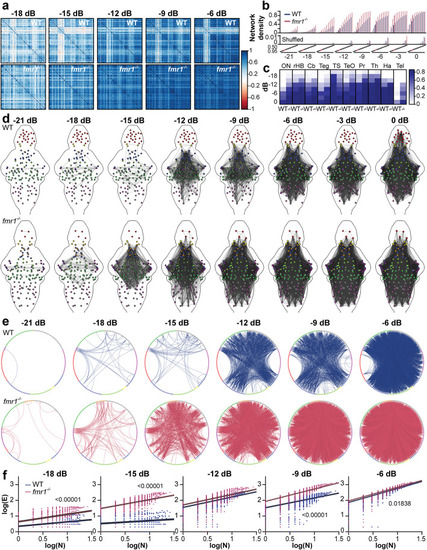
Functional brain-wide auditory networks in WT and |
|
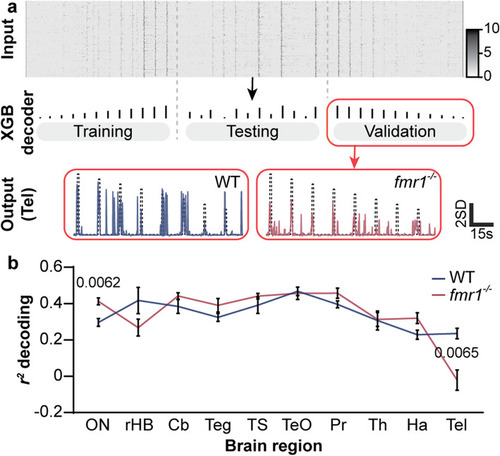
Population decoding of sound amplitude in different WT and PHENOTYPE:
|