- Title
-
Supervillin Is a Component of the Hair Cell's Cuticular Plate and the Head Plates of Organ of Corti Supporting Cells
- Authors
- Pollock, L.M., Gupta, N., Chen, X., Luna, E.J., McDermott, B.M.
- Source
- Full text @ PLoS One
|
Supervillin is expressed in the mouse and zebrafish ear. (A) RT-PCR detection of svila and svilc mRNAs in zebrafish hair cells using two primer pairs to detect svila mRNA [RT plus (lanes 1,3), RT minus (lanes 2,4)] and two primer pairs to detect svilc mRNA [RT plus (lane 5,6), RT minus (lane 7,8)]. Svilb [RT plus (lane 9), RT minus (lane 10)] and svild [RT plus (lane 11), RT minus (lane 12)] mRNAs were detected in zebrafish maculae. (B) RT-PCR of Svil mRNA from mouse hair cells [RT plus (lane 1), RT minus (lane 2)]. (C-H) RNA in situ hybridization. Whole mount 4-dpf zebrafish treated with probes antisense to svila (C) and svilc (F) mRNAs. Both genes are expressed in the otocyst (arrowheads). Controls are displayed. Magnified otocysts show svila (D) and svilc (G) are expressed in the anterior macula (AM). Sense-probed controls (E,H). White dashed lines denote the otic vesicles. Yellow boxes show positions of AM hair cells. PC indicates the region of the posterior cristae (out of focus). |
|
Supervillin localizes to mouse hair cell CPs and cochlear supporting cell head plates. (A-E) Confocal micrographs of mouse vestibular hair cells labeled with anti-SVIL (green) and phalloidin (red) at different developmental stages. (A) A top-down view of several hair cells from a mouse at P1. Supervillin labels the CPs (asterisk) but not the fonticulus (arrowhead). (B,C) Views of hair cells from a 6-month-old mouse. Supervillin labels the CP but not the fonticulus (arrowhead) or stereocilia (arrows). (D,E) Type I (D) and type II (E) vestibular hair cells from a P3 mouse co-labeled with anti-tubulin (blue), which marks somatic microtubules underlying the CPs (arrowheads). Arrow in (E) indicates a kinocilium from a neighboring hair cell. A region of interest (ROI, indicated by the purple line) was selected to span the hair bundle (top portion of the line), CP (middle portion of the line), and underlying microtubules (bottom portion of the line). (F) Fluorescence intensity profile using the ROI from (E). The hair bundle (top portion of the purple ROI line) corresponds to the left region of the plot, showing robust F-actin-associated signal (red), while SVIL- (green) and tubulin-associated (blue) signals are minimal. The middle region of the plot corresponds to the CP (middle portion of the ROI line) and shows overlapping SVIL- and F-actin-associated signals; however, in the right region of the plot, only tubulin-associated signal is seen below the CP (bottom portion of the ROI line). (G-M) Confocal micrographs of mouse cochlear hair cells labeled with anti-SVIL (green) and anti-actin (red). (G-J) Hair cells at the middle (G,H) and apical (I,J) cochlear turns of a P1 mouse. At the middle turn (G,H), SVIL localizes to the CPs (asterisk) and to the region of the hair cell-supporting cell junctions (arrow). At the apical turn (I,J), SVIL co-localizes with actin near the apical surface of the developing hair cells. (K,L) In the middle turn of the P3 mouse cochlea, SVIL localizes to CPs of outer hair cells (OHCs) and inner hair cells (IHCs) and to supporting cell apicolateral margins, including those of Deiters’ cells (DC1, DC2, DC3), outer pillar cells (OP), and inner phalangeal cells (IPC). (M) Side view of two IHCs from the middle turn of a P3 mouse cochlea co-labeled with an antibody to tubulin (blue) demonstrates that SVIL localizes between the hair bundle (arrow) and somatic microtubules (arrowhead), at the region of the cuticular plate (asterisk). (N) Magnification of two OHCs and the Deiters’ cell between them (arrow) from the basal turn of a P1 mouse cochlea labeled with anti-SVIL (green) and anti-β-catenin (red). (O) Magnification of the first two rows of OHCs from the basal turn of a P2 mouse cochlea labeled with anti-SVIL (green) and anti-ZO-1 (red). SVIL strongly localizes to the apicolateral margins of the OPs (arrowhead) and the DCs (arrow). (P) Z-stacks of confocal sections were converted into a 3D model using the Leica Software. A 3D reconstruction of an OHC (asterisk) from the second row flanked by two DCs (arrow) labeled with anti-SVIL (green) and anti-ZO-1 (green) is seen. (Q) Fluorescence intensity profile of the cell in (O) using the ROI indicated by the purple line demonstrates that the signal associated with SVIL (green) is concentrated in the supporting cells, sandwiched between ZO-1-rich bands (red). In graphs in F and Q, intensity scales are linear, but the units are arbitrary. Scale bars, 2 μm. |
|
Supervillin localizes to zebrafish hair cell CPs. (A,B) Confocal micrographs of 4-dpf zebrafish anterior crista hair cells expressing GFP-fascin 2b (red) and labeled with anti-Svila (green). Arrowheads in A indicate hair cells out of focus or bent. Arrow indicates hair-bundle-localized GFP-fascin 2b. Asterisk indicates a CP. (C,D,E) Confocal micrographs of posterior macula hair cells from zebrafish expressing Acf7a-Citrine (red) and labeled with anti-Svila (green) (C,E) or phalloidin (green) (D). Asterisks in (C,D) indicate CPs. Acf7a-Citrine encircles the CP, localizes to the CP base, out of the focal plane in C-E, and is found weakly throughout the CP. (F) Schematic of the zebrafish posterior macula tissue with the location of Svila immunolabeling in green and Acf7a-Citrine indicated in red. Scale bars, 2 μm. EXPRESSION / LABELING:
|
|
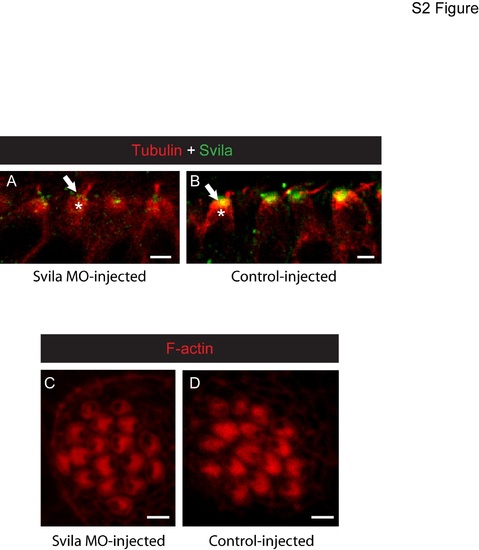
Morpholino knockdown of Svila in zebrafish. Confocal micrographs of 4-dpf zebrafish hair cells injected with a morpholino targeting Svila (A,C) or a 5-bp mismatch control morpholino (B,D). (A,B) Hair cells from the anterior macula labeled with anti-Svila (green) and anti-acetylated tubulin (red) reveal that the intensity of Svila protein at the cuticular plate (arrows) is diminished in Svila morpholino-injected fish (A) compared to fish injected with control (B), but some Svila protein is still detected (A). Fluorescence intensity of anti-Svila at the CP was compared to that associated with anti-tubulin labeling of the underlying microtubules (asterisks). Phalloidin labeling of neuromast hair cells from Svila morpholino-injected (C) and control-injected (D) fish reveals normal gross cuticular plate structure in the morphants. |