- Title
-
Mutation of wrb, a Component of the Guided Entry of Tail-Anchored Protein Pathway, Disrupts Photoreceptor Synapse Structure and Function
- Authors
- Daniele, L.L., Emran, F., Lobo, G.P., Gaivin, R.J., Perkins, B.D.
- Source
- Full text @ Invest. Ophthalmol. Vis. Sci.
|
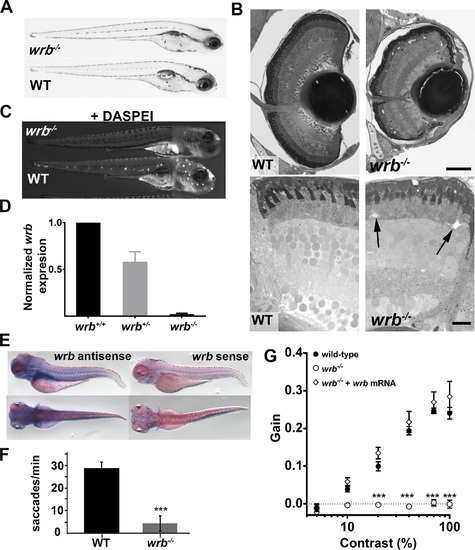
Visual system defects caused by retroviral insertion within the wrb gene. (A) Lateral views of 5 dpf wrb-/- and wild-type larvae. (B) Toluidine blue–stained 1-µm thick retinal sections from 5 dpf wild-type and wrb-/- larvae. Occasional acellular holes were observed in the ONL of wrb-/- mutants (arrows). (C) Live 5 dpf wrb-/- (top) and wild-type (bottom) larvae stained with the vital styryl dye DASPEI to label neuromasts. Two overlapping images, focused at the head or trunk, were stitched together for each panel. (D) Expression levels of wrb mRNA in heterozygous (wrb+/-) and mutant (wrb-/-) larvae at 5 dpf, compared with wild-type larvae, and normalized against beta-actin as measured by qRT-PCR. (E) Whole mount in situ hybridization at 5 dpf with antisense (left) and sense (right) wrb probes. (F) Saccade frequency measured from wild-type and wrb-/- larvae at 5 dpf. (G) Contrast response function measured from smooth pursuit eye movements. Gain versus log contrast for 5 dpf wild-type (black closed circles) wrb-/- (open circles) and wrb-/- rescued with wrb mRNA (open diamonds). Errors bars: SEM. Significance levels are as follows: ***P < 0.0001. Scale bars: 50 µm (B, top) 10 µm (B, bottom). PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|
|
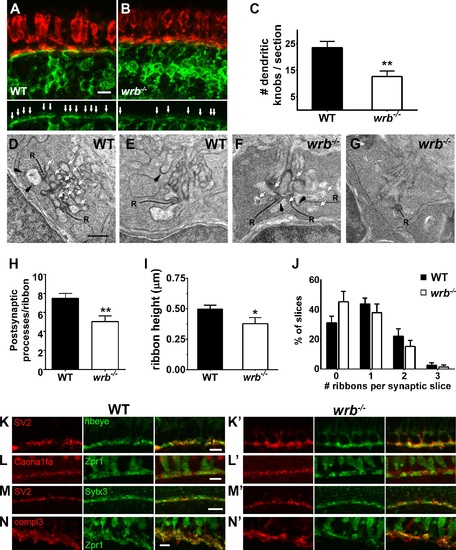
Presynaptic and postsynaptic alterations in wrb-/- mutant photoreceptors. (A, B) Representative images of the OPL from cryosections of 5 dpf Tg(nyx:mYFP) and wrb-/-; Tg(nyx:mYFP) mutant retinas immunostained for red/green double cones (zpr1, red) and GFP (green). Bottom panels show bipolar dendritic projections (white arrows) within the OPL at higher magnification. (C) Quantification of dendritic invaginations inside cone pedicles across several cryosections (n = 4 and 8 sections for wild type and wrb-/-, respectively). (D-F) Transmission electron microscopy images of cone pedicles. Synaptic ribbons (R) were surrounded by postsynaptic processes (white arrows denote representative processes). Horizontal cells could be identified by characteristic densities and electron-lucent cytoplasm (black arrowheads). Quantification of postsynaptic processes per ribbon in wild-type and mutant cone pedicles (n = 26 wild-type, n = 19 wrb-/- synapses). (I) Quantification of photoreceptor ribbon heights in wild-type and mutant synapses (n = 32 wild-type, n = 18 wrb-/- synapses). (G) Quantification of average number of synaptic terminals in which 0, 1, 2, or 3 ribbons were encountered (n = 163 wild-type, n = 216 wrb-/- synapses). (H-K′) Immunohistochemistry of 5 dpf retinal cryosections with indicated photoreceptor presynaptic markers. Images were centered at the OPL of wild-type and wrb-/- mutants. Cacna1fa, pore forming alpha subunit of the presynaptic L-type calcium channel; SV2, synaptic vesicle protein 2; Sytx3, photoreceptor-specific target SNARE syntaxin 3B, compl3- exocytosis regulator complexin 3. * P < 0.05. **P < 0.001. Scale bars: 5 µm (A, B, H-I′, K, K′); 10 µm (J, J′); and 0.5 µm (D-G). |
|
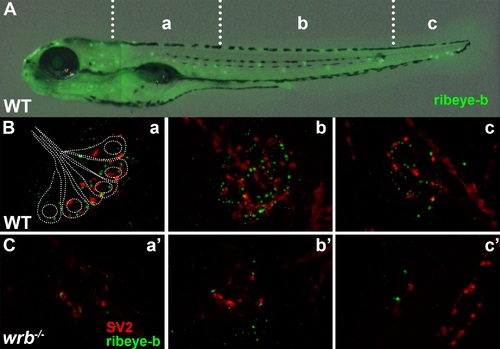
Loss of Wrb disrupts endogenous ribeye localization at synaptic ribbons in hair cells at 5 dpf. (A) Lateral image of 5 dpf larvae immunostained with ribeye b antibodies to label lateral line hair cells. Hair cells in rostral (a, a′), middle (b, b′), and caudal (c, c′) regions of the trunk were imaged by fluorescence microscopy. (B, C) Fluorescence images showing individual neuromasts immunolabeled with SV2 (red) and ribeye-b (green) antibodies in wild-type and wrb-/- mutant larvae. Dotted outline in (B) depicts the orientation of individual hair cells within a single neuromast, as it relates to synaptic immunoreactivities. |
|
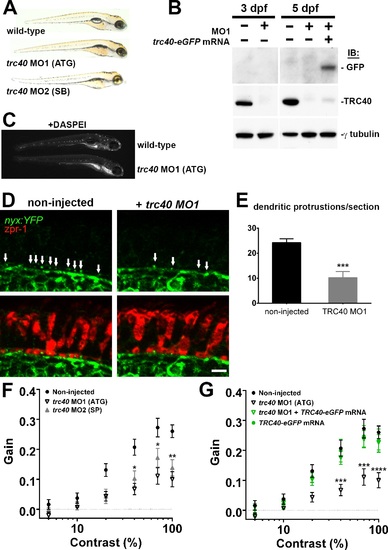
Morpholino knockdown of trc40 results in wrb-/--like phenotypes. (A) Lateral view of 5 dpf uninjected larvae (top) and trc40 MO1 (translation-blocking, ATG), and trc40 MO2 (splice-blocking, SB) injected larvae. (B) Western blots of 15 µg protein lysates collected from whole larvae at 3 and 5 days after injection with 3 ng trc40 MO1, or MO1 with human trc40-eGFP mRNA. Membranes were immunoblotted with antibodies to detect GFP (top), TRC40 (middle) and γ-tubulin (bottom). (C) Live 5 dpf wild-type and trc40 morphants (MO1) larvae stained with the vital styryl dye DASPEI. (D) Immunohistochemistry of 5 dpf cryosections of Tg(nyx:mYFP) and Tg(nyx:mYFP) + trc40 MO1 morphant larvae using antibodies to detect YFP (green) and red/green double cones (zpr1, red). White arrows indicate bipolar cell dendritic protrusions into cone pedicles. (E) Quantification of dendritic protrusions observed within cone pedicles from cryosections of 5 dpf larvae (n = 11 wild-type, 8 trc40 MO1). (F) Gain of OKR versus log contrast for noninjected wild-type larvae (closed circles), 3 ng trc40 MO1 morphants (open down triangles) and 10 ng trc40 MO2 morphants (filled upward triangles) all at 5 dpf. Significance values indicated for trc40 MO2 morphants only. (G) Gain of OKR versus log contrast for noninjected larvae (filled circles), or larvae injected with 3 ng trc40 MO1 (downward gray triangles), 3 ng trc40 MO1 + trc40-eGFP mRNA (downward green triangles) and human trc40-eGFP mRNA alone (filled green circles). Significant values noted for only for trc40 MO1 morphants. Significance levels are as follows: *P < 0.05. **P < 0.01. ***P < 0.001. ****P < 0.0001. Scale bar: 5 µm. EXPRESSION / LABELING:
PHENOTYPE:
|
|
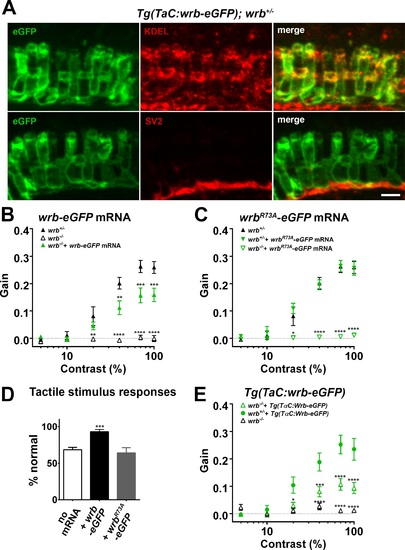
Photoreceptor wrb expression is critical for normal visual sensitivity. (A) Immunofluorescent images 10 µm-thick transverse cryosections of 5 dpf retinas from Tg(TaC:wrb-eGFP) stained antibodies to GFP (green), KDEL (top, red) or SV2 (bottom, red) to label ER and synapse respectively. (B) OKR gain versus log contrast plots for 5 dpf wrb+/- (closed triangles), wrb-/- mutants (open triangles), or wrb-/- mutants injected with mRNA encoding wrb-eGFP (green triangles). (C) Gain of OKR versus log contrast plots for 5 dpf wrb+/- heterozygous larvae (closed triangles), wrb+/- larvae injected with mRNA encoding wrbR73A-eGFP (green closed triangles), or wrb-/- mutants injected with mRNA encoding wrbR73A-eGFP (green open triangles). (D) Quantification of the percentage of larval offspring from a wrb heterozygous mating showing normal avoidance responses to light tail touch. Roughly 25% of larvae offspring (homozygous wrb-/- mutants) failed to show normal responses. Almost 100% of larvae exhibited normal touch responses following injection of mRNA encoding wrb-eGFP into 1-cell embryos. 25% of larvae failed to show a normal response following injection of mRNA encoding wrbR73A-eGFP. (E) OKR gain versus log contrast plots from 5 dpf heterozygous and homozygous wrb mutants carrying the Tg(TαC:wrb-eGFP) transgene. *P < 0.05. **P < 0.01. ***P < 0.001. ****P < 0.0001. Scale bar: in (A): 5 µm. |