- Title
-
The transmembrane inner ear (Tmie) protein is essential for normal hearing and balance in the zebrafish
- Authors
- Gleason, M.R., Nagiel, A., Jamet, S., Vologodskaia, M., Lopez-Schier, H., and Hudspeth, A.J.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
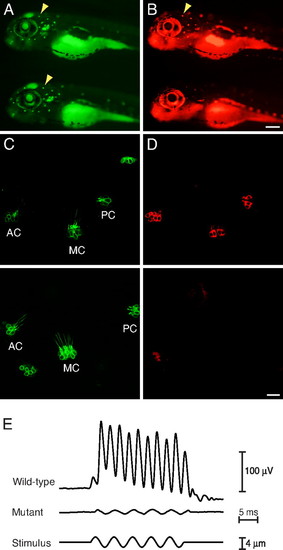
Defective hair-cell function. (A) In a WT (Upper) and a mutant (Lower) larva at 6 dpf, arrowheads indicate neuromasts labeled by Tg(pou4f3:gap43-mGFP) (green). (B) Upon exposure to FM4–64 (red), the WT specimen (Upper) shows robust incorporation of the fluorophore into hair cells but the mutant (Lower) shows none. (C) In the inner ears of WT (Upper) and mutant (Lower) animals at 5 dpf, Tg(pou4f3:gap43-mGFP) (green) marks the anterior (AC), medial (MC), and posterior cristae (PC). (D) Pressure-injection of FM4–64 (red) into the otic cavities labels hair cells in the cristae of WT (Upper) but not mutant (Lower) fish. (E) Averaged extracellular recordings of microphonic potentials measured at 5–7 dpf from the inner ears of eight WT larvae show a clear response at twice the stimulus frequency, whereas the record from eight mutant larvae shows only a stimulus artifact. (Scale bars, 200 μm in A and B; 20 μm in C and D.) |
|
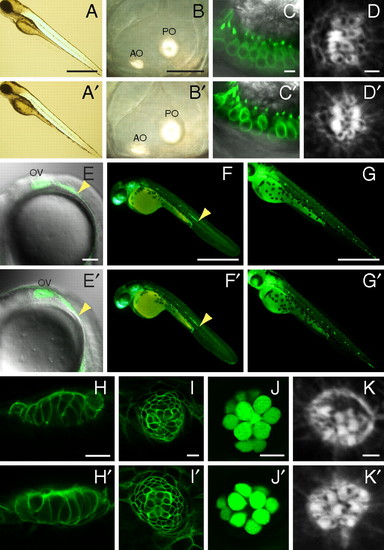
Absence of gross morphological and developmental defects. In each panel, a WT larva (Upper) is contrasted with a mutant (Lower). (A, A′) Mutants display a normal body shape and length at 4 dpf. (B, B′) In a mutant, the semicircular canals develop normally and the anterior (AO) and posterior otoliths (PO) are normal. (C, C′) Labeling of hair cells by Tg(pou4f3:gap43-mGFP) (green) reveals no obvious differences in their number, shape, or localization in the anterior macula. (D, D′) In lateral-line neuromasts at 3 dpf, the hair bundles of mutants display normal planar cell polarity. The kinocilium of each hair bundle is marked by a black notch denoting the absence of actin staining by phalloidin. At 22 hpf (E, E′), 2 dpf (F, F′), and 3 dpf (G, G′), mutants display normal migration of the lateral-line primordium and formation of neuromasts, here labeled by Tg(cldnB:lynGFP) (green). OV, otic vesicle; arrowheads indicate the leading edges of the primordia. (H and I) Supporting cells labeled by Tg(cldnB:lynGFP) (green) are normally arranged in mutants in the anterior (H, H′) and posterior lateral lines (I, I′) of 5-dpf larvae. (J, J′) Fifty hours after treatment with Cu2+, a 7-dpf mutant larva shows normal regeneration of hair cells labeled by Et(krt4:GFP)sqet4 (green). (K, K′) Phalloidin staining shows that regenerated hair cells display planar cell polarity in a mutant. (Scale bars, 500 μm in A, F, and G; 50 μm in B; 5 μm in C; 2 μm in D and K; 100 μm in E; 10 μm in H–J.) PHENOTYPE:
|
|
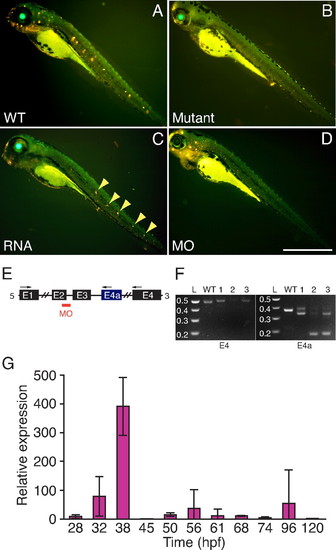
Confirmation of the mutation. (A) Exposure of a WT 4-dpf larva to 4-Di-2-ASP (yellow) labels neuromasts. (B) Labeling is absent in a mutant. (C) Injection of capped RNA coding for normal Tmie partially rescues a mutant, restoring labeling in some neuromasts (arrowheads). (D) Injection of a WT embryo with a morpholino against tmie spares fluorophore incorporation in some neuromasts of the anterior lateral line but not in those of the posterior lateral line. (Scale bar, 500 μm.) (E) The morpholino (MO) targets the splice junction between exon 2 and intron 2. The arrows show the locations of the primers used to amplify the splicing products in RT-PCRs. (F) The RT-PCR products amplified from one WT and three morphant fish (1-3) confirm that the injected morpholino disrupts normal splicing of tmie (Left) and of its alternative transcript (Right), producing various aberrant products. L, DNA ladder in kilobases. (G) Endogenous expression of Tmie is sparse. The relative expression of tmie at different developmental stages was measured by qRT-PCR with RNA extracted from whole larvae. The results are presented as the mean and standard deviation of the ratio of each measurement to that at 24 hpf, the lowest value observed. Expression peaks at 38 hpf, declining sharply thereafter and remaining low through 120 hpf. |
|
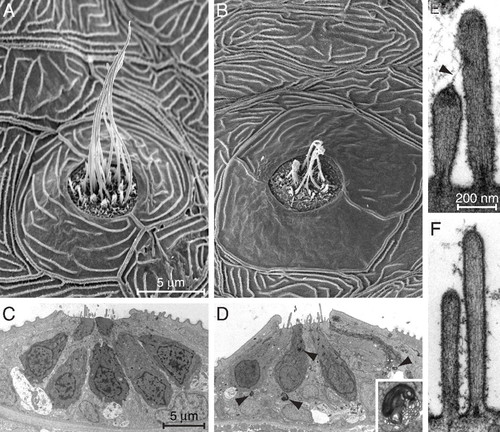
Ultrastructural features of mutant and normal hair cells. (A) A scanning electron micrograph of a midtail neuromast from the posterior lateral line of a mutant at 6 dpf shows a dozen long kinocilia emerging from short clusters of stereocilia. (B) A neuromast from a similar position in an age-matched, homozygously mutant fish displays shrunken kinocilia and a diminished number of hair bundles. The semilunar periderm cells surrounding the neuromast are abnormally smooth. (C) A low-power transmission electron micrograph of a neuromast in a 6-dpf control larva shows parts of seven hair cells, some contacting large nerve terminals. (D) A mutant neuromast is characterized by several lysosomes (arrowheads) both within them and in supporting cells. A representative lysosome is enlarged fivefold in the inset. (E) A high-power micrograph from a control animal demonstrates a pair of sterocilia connected by a tip link (arrowhead) that terminates at its upper end in a prominent insertional plaque. (F) In a mutant larva of the same age, the stereocilia are narrow and display little basal tapering. Neither tip links nor insertional plaques are evident, and the shorter stereocilia lack dense material beneath their tips. The magnifications are identical for A and B, for C and D, and for E and F. PHENOTYPE:
|

Unillustrated author statements PHENOTYPE:
|