- Title
-
Mechanism of development of ionocytes rich in vacuolar-type H(+)-ATPase in the skin of zebrafish larvae
- Authors
- Esaki, M., Hoshijima, K., Nakamura, N., Munakata, K., Tanaka, M., Ookata, K., Asakawa, K., Kawakami, K., Wang, W., Weinberg, E.S., and Hirose, S.
- Source
- Full text @ Dev. Biol.
|
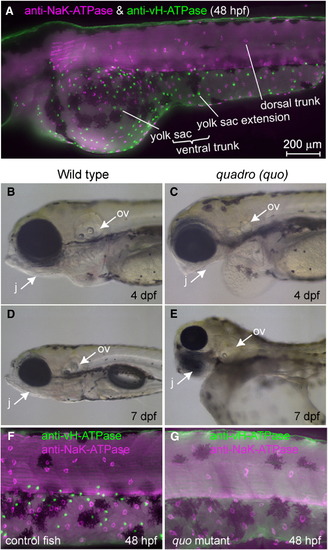
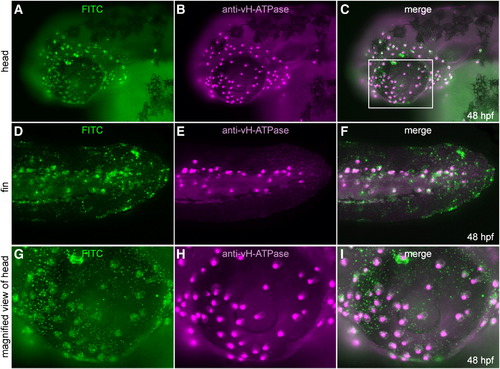
Distribution of two major types of MRC in the skin of zebrafish larvae and a mutant lacking one of them. (A) Double immunostaining of NaK-ATPase (magenta) and vH-ATPase (green) at 48 hpf, viewed from lateral side. (B–E) Phenotypes of quadro (quo) mutant. Defects are seen in otic and jaw development in quo mutant embryos (C, E). All panels show lateral views of live embryos with anterior to the left. j, jaw; ov, otic vesicle. (F, G) Double immunostaining with anti-vH-ATPase and anti-NaK-ATPase antibody at 48 hpf of wild-type control (F) and quo mutant (G). |
|
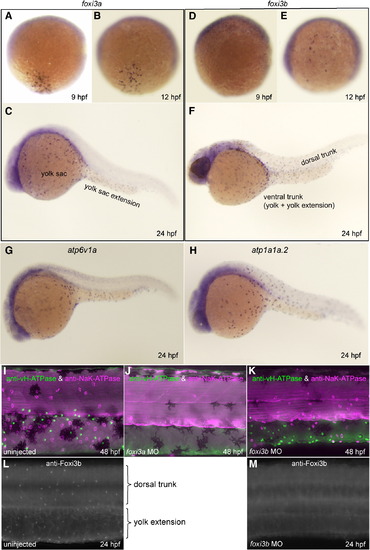
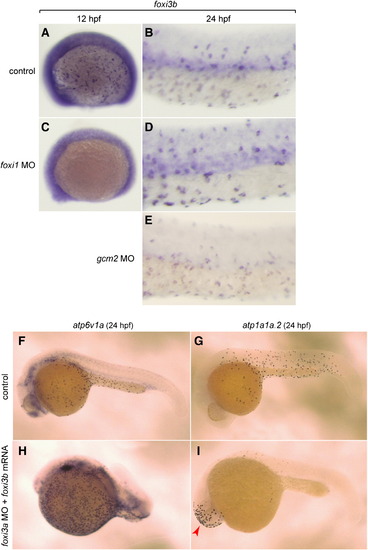
Spatio-temporal expression patterns and knockdown analyses of foxi3a and foxi3b. All panels are lateral views, dorsal side up, anterior to the left. (A–F) Whole mount in situ hybridization with foxi3a probe (A–C) and foxi3b probe (D–F). Expression of foxi3a begins earlier than that of foxi3b (A, D). foxi3b-positive cells are distributed in the dorsal trunk region in addition to the ventral trunk region (F). (G, H) Expression of marker genes for vH-MRC (atp6v1a) and NaK-MRC (atp1a1a.2). (I–K) Double immunostaining with anti-vH-ATPase and anti-NaK-ATPase antibody at 48 hpf of uninjected wild-type control (I), foxi3a morphant (J), and foxi3b morphant (K). (L, M) Immunostaining with anti-Foxi3b antibody at 24 hpf of uninjected wild-type control (L) and foxi3b morphant (M). Knockdown of foxi3b with MO exerted no or little effects on the generation of vH-MRC and NaK-MRC (K) while knockdown of foxi3a resulted in complete loss of both vH-MRC and NaK-MRC (J). EXPRESSION / LABELING:
PHENOTYPE:
|
|
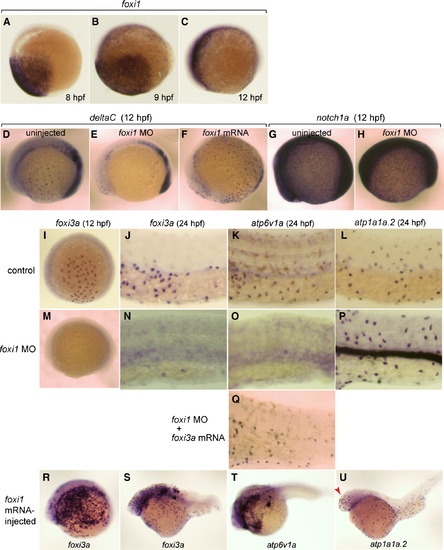
Broad expression of foxi1 in the presumptive ectoderm from 8 hpf and its regulatory role upstream of deltaC and foxi3a. (A–C) Whole mount in situ hybridization with foxi1 probe at 8 hpf (A), 9 hpf (B), and 12 hpf (C). (D–F) Whole mount in situ hybridization with deltaC probe at 12 hpf of uninjected wild-type control (D), foxi1 morphant (E), and foxi1 mRNA-injected embryos (F). (G, H) Whole mount in situ hybridization with notch1a probe at 12 hpf of uninjected (G) and foxi1 MO-injected (H) embryos. (I–U) Whole mount in situ hybridization with (i) foxi3a probe at 12 hpf (I, M, R) and 24 hpf (J, N, S), (ii) atp6v1a probe at 24 hpf (K, O, Q, T), and (iii) atp1a1a.2 probe at 24 hpf (L, P, U) of uninjected wild-type controls (I–L), foxi1 morphants (M–P), foxi1 morphant injected with foxi3a mRNA (Q), and foxi1 mRNA-injected embryos (R–U); for the controls of R–U, also see Figs. 2C, G, H. atp1a1a.2 and atp6v1a are marker genes for NaK-MRC and vH-MRC, respectively. Red arrow in panel U indicates foxi1-induced ectopic generation of NaK-MRC in the head region. Embryos are shown with their ventral side down and animal pole to the right. EXPRESSION / LABELING:
PHENOTYPE:
|
|
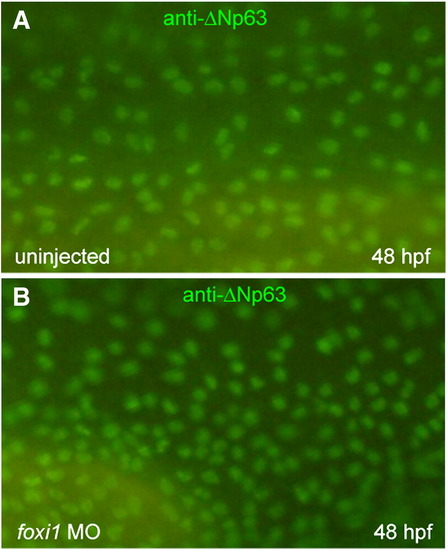
Foxi1 is not necessary for keratinocyte differentiation. Control (A) and foxi1 morphant (B) embryos were immunostained at 48 hpf for ΔNp63, a marker for keratinocyte precursor cells. |
|
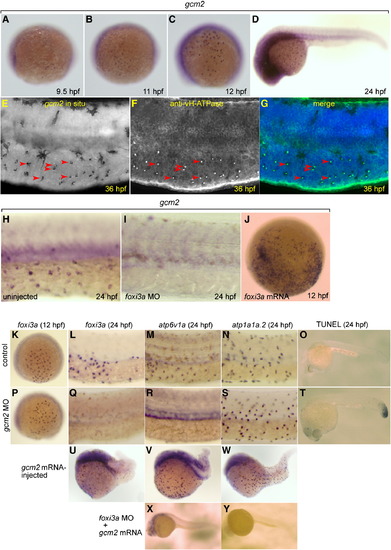
Expression of gcm2 in vH-MRC and under the influence of foxi3a. (A–D) Whole mount in situ hybridization with gcm2 probe at 9.5 hpf (A), 11 hpf (B), 12 hpf (C), and 24 hpf (D). (E–G) Co-staining of gcm2 (E, in situ hybridization) and vH-ATPase (F, immunostaining), and their merged image (G). gcm2-positive cells (arrowheads in E) completely overlap with vH-MRCs (arrowheads in F). (H, I) Whole mount in situ hybridization with gcm2 probe at 24 hpf of uninjected wild-type control (H), and foxi3a morphant (I). (J) Whole mount in situ hybridization with gcm2 probe at 12 hpf of foxi3a mRNA-injected embryo. (K–N, P–S, U–Y) Whole mount in situ hybridization with (i) foxi3a probe at 12 hpf (K, P) and 24 hpf (L, Q, U), (ii) atp6v1a probe at 24 hpf (M, R, V, X), and (iii) atp1a1a.2 probe at 24 hpf (N, S, W, Y) of uninjected wild-type controls (K–O), gcm2 morphants (P–T), gcm2 mRNA-injected embryos (U–W), and foxi3a morphant injected with gcm2 mRNA (X, Y). (O, T) TUNEL assay of control (O) and gcm2 MO-injected (T) embryos. Injection of gcm2 MO induced apoptosis in the tail region but not in the natural ionocyte domains (T). The densities of vH-MRC and NaK-MRC seem to be slightly increased by gcm2 mRNA injection (V, W) if compared with controls (Figs. 2G, H), but it is difficult to draw a definite conclusion because of morphogenetic defects caused by misexpression of gcm2. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Transplantation experiment to show cell-autonomous nature of foxi3a. (A–F) foxi3a mRNA and FITC-dextran injected cells were transplanted into the animal pole of a foxi3a MO-injected embryo at 4–5 hpf. Transplanted cells (A, D, G; FITC-green) and cells expressing vH-ATPase (B, E, H; immunostaining, purple) completely overlapped (C, F, I; merged images). (G–I) High-magnification view of ectopic expression of vH-ATPase. Magnified area is indicated by a white box (C). |
|
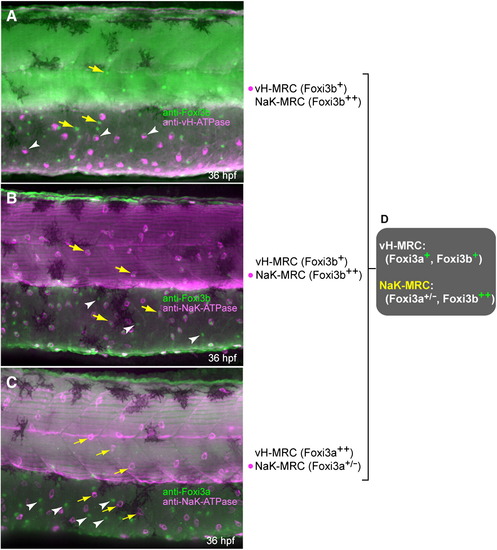
Expression profiles of protein products of foxi3a and foxi3b in vH-MRC and NaK-MRC revealed by immunohistochemistry. All panels are lateral views of yolk sac extension and dorsal trunk, dorsal side up, anterior to the left. Yellow arrows and white arrowheads represent typical NaK-MRC and vH-MRC, respectively. (A) Double immunostaining of Foxi3b and vHA-TPase. (B) Double immunostaining of Foxi3b and NaK-ATPase. (C) Double immunostaining of Foxi3a and NaK-ATPase. (D) Summary of the expression profiles. EXPRESSION / LABELING:
|
|
Complex interplay between the foxi1–foxi3a/gcm2 pathway and foxi3b. (A–E) Whole mount in situ hybridization, with a foxi3b probe, of control (A, B), foxi1 MO-injected (C, D), and gcm2 MO-injected (E) embryos. (F, H) Whole mount in situ hybridization, with an atp6v1a probe, of control embryo (F) and foxi3a morphant injected with foxi3b mRNA (H). (G, I) Whole mount in situ hybridization, with an atp1a1a.2 probe, of control embryo (G) and foxi3a morphant injected with foxi3b mRNA (L). foxi3b-induced formation of NaK-MRC was only observed ectopically in the head region (red arrow) in foxi3a morphants. EXPRESSION / LABELING:
|
|
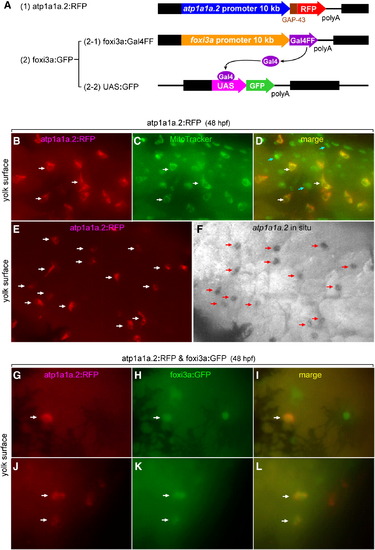
Lineage tracing showing a foxi3a-positive nature of NaK-MRC precursor. (A) Structures of the three expression vectors used for in vivo RFP labeling of atp1a1a.2-positive cells and in vivo GFP labeling of foxi3a-positive cells. (B–F) Demonstration that RFP is specifically expressed in the NaK-MRC subtype in the atp1a1a.2:RFP transgenic zebrafish line. In C, MitoTracker stains all types of MRCs. Blue arrows in panel D indicate MRCs other than NaK-MRC. (G–L) Examples of coexpression of RFP and GFP in the same cell under the influence of the promoters of the atp1a1a.2 and foxi3agenes in double transgenic zebrafish embryos. Dark areas in panels G–I represent spider-shaped melanocytes. EXPRESSION / LABELING:
|
Reprinted from Developmental Biology, 329(1), Esaki, M., Hoshijima, K., Nakamura, N., Munakata, K., Tanaka, M., Ookata, K., Asakawa, K., Kawakami, K., Wang, W., Weinberg, E.S., and Hirose, S., Mechanism of development of ionocytes rich in vacuolar-type H(+)-ATPase in the skin of zebrafish larvae, 116-129, Copyright (2009) with permission from Elsevier. Full text @ Dev. Biol.