- Title
-
Nuclear localization of the zebrafish tight junction protein nagie oko
- Authors
- Bit-Avragim, N., Rohr, S., Rudolph, F., Van Der Ven, P., Fürst, D., Eichhorst, J., Wiesner, B., and Abdelilah-Seyfried, S.
- Source
- Full text @ Dev. Dyn.
|
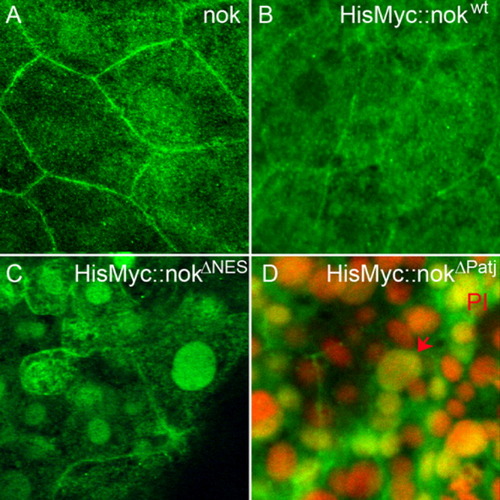
The nok NES motif is required for nuclear export. Images represent reconstructions of confocal Z-stack sections imaged on late gastrula stage whole mounts. A: Endogenous nok mostly localizes to the outer cell membrane of EVL cells whereas nuclear localization is barely detectable. B: HisMyc-tagged nokwt fusion protein detected with anti-Myc antibody, green. The wt protein localizes to the outer cell membrane in EVL cells. C: HisMyc-tagged nokΔNES (green) localizes to the nucleus of EVL cells and lower levels of protein are also present at the outer cell membrane. D: HisMyc-tagged nokΔPatj localizes to the nucleus and lower levels are present at the outer cell membrane (green). Red arrow indicates an EVL cell nucleus, which is large compared with the smaller DL cell nuclei located just underneath. Propidium iodide (PI) nuclear counterstain in red. EXPRESSION / LABELING:
|
|
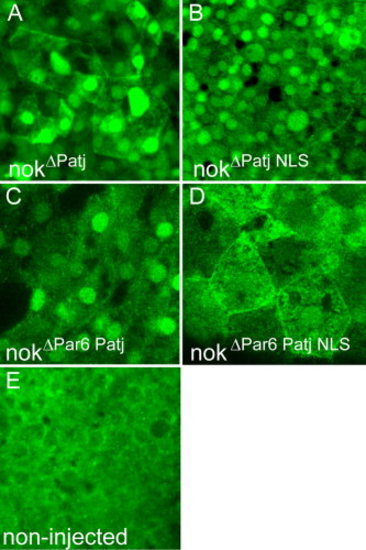
Redundant mechanisms involved in nok nuclear accumulation. Images are reconstructions of confocal Z-stack sections imaged on late gastrula stage whole mounts. HisMyc-tagged nok deletion proteins are detected with anti-Myc antibody (green). A: HisMyc-tagged nokΔPatj strongly localizes to nuclei of EVL and DL cells (recognizable by their size) and to outer cell membranes. B: HisMyc-tagged nokΔPatj NLS protein that lacks the L27N domain and the two nuclear import signals still localizes to the nucleus, which indicates that alternatives routes of nok nuclear import must exist. C: HisMyc-tagged nok nokΔPar6 Patj protein localizes to nuclei of EVL and DL cells indicating that direct association with Par6 is not necessary for nuclear accumulation of nok. D: In contrast, HisMyc-tagged nokΔPar6 Patj NLS protein fails to localize to nuclei of EVL and DL cells suggesting redundancy of the NLS motifs and association with Par6 in nok nuclear accumulation. E: Control non-injected embryos stained with the anti-Myc antibody. |
|
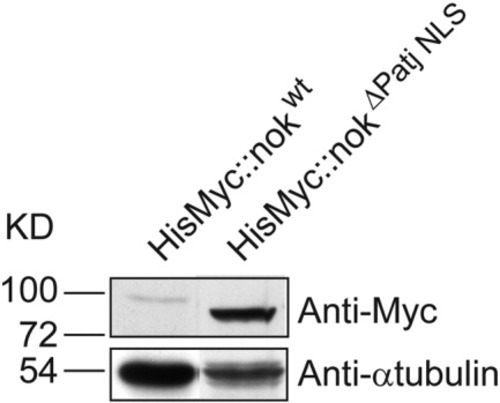
Increased levels of nuclear nok protein. Increased protein levels of HisMyc-tagged nokΔPatj NLS (expected size of 74.1 kD) compared with HisMyc-tagged nokwt (expected size of 82.2 kD) suggest that nuclear accumulation stabilizes nok protein. |
|
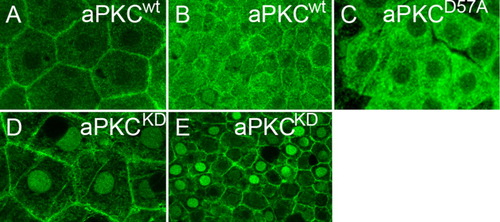
aPKC kinase catalytic activity prevents nuclear accumulation. Images are reconstructions of confocal Z-stack sections imaged on late gastrula stage whole mounts. Anti aPKC (A,B,D,E) and anti-Myc (C) stainings are false-colored. A: Endogenous aPKC localization in EVL cells is mostly at the outer cell membrane whereas (B) weak levels of wt protein are also detectable throughout the cytoplasm and nuclei of DL cells. C: HisMyc-tagged aPKCiD57A within EVL cells localizes to the cytoplasm and is complete absent from outer cell membranes or nuclei. D: aPKCiKD strongly localizes to the nuclei of EVL and (E) DL cells. All images are apical views onto the tissue. EXPRESSION / LABELING:
|
|
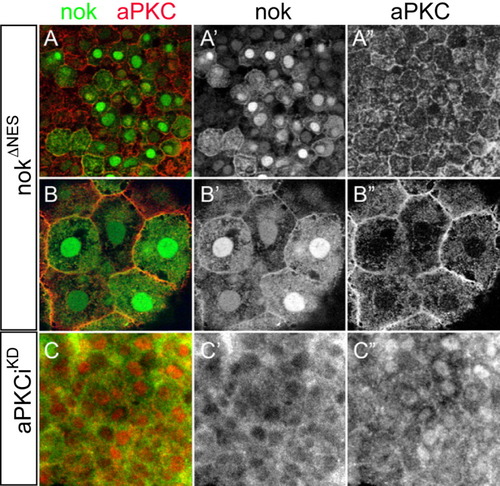
nok and aPKCi are not sufficient to tether each other within the nucleus. Images are reconstructions of confocal Z-stack sections imaged on late gastrula stage whole mounts. Myc and aPKC stainings are false-colored. A,B: Embryos expressing high levels of HisMyc-tagged nokΔNES were counterstained with anti aPKC. A,A′: Within DL cells, high levels of nuclear HisMyc-tagged nokΔNES are present whereas aPKC localizes to outer cell membranes and no increased levels of nuclear aPKC are detectable (A“). B,B′: Within epithelial EVL cells, HisMyc-tagged nokΔNES is at outer cell membranes and within the nucleus. B”: aPKC is predominantly associated with outer cell membranes. C: Embryos overexpressing HisMyc-tagged nokwt (green) and non-tagged aPKCiKD (red), whereas nokwt is absent from the nucleus (C′), aPKCiKD is nuclear. All images are apical views onto the tissue. |
|
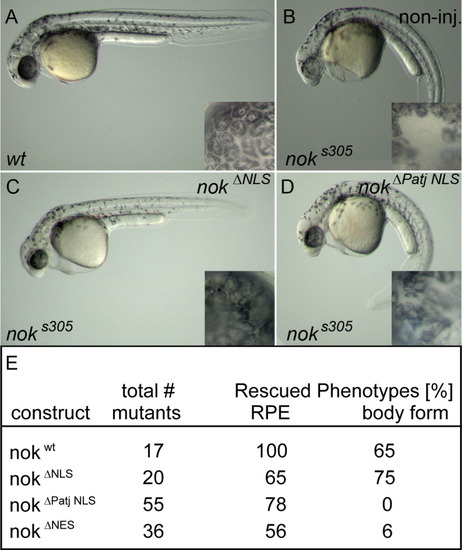
Loss of nok nuclear import and export signals does not affect epithelial maintenance. Phenotypes of (A) wt, (B) noks305 mutant, (C) noks305 mutant rescued with nokΔNLS mRNA or with (D) nokΔPatj NLS mRNA at 36 hpf. The straight body form is rescued in nokΔNLS mutants (C) but not in nokΔPatj NLS mutants (D). Insets depict details of RPE from each corresponding embryo. The cobble-stone like appearance of RPE tissue surrounding the neural retina is severely disrupted and displays wholes in (B) noks305 but this phenotype is partially complemented in (C) nokΔNLS or (D) nokΔPatj NLS mutants. E: Summary of rescue efficiencies of the different deletion mutants. Mutant phenotypes for nokΔNES are similar to nokΔPatj NLS (not shown). PHENOTYPE:
|