- Title
-
HDAC8: A Promising Therapeutic Target for Acute Myeloid Leukemia
- Authors
- Spreafico, M., Gruszka, A.M., Valli, D., Mazzola, M., Deflorian, G., Quintè, A., Totaro, M.G., Battaglia, C., Alcalay, M., Marozzi, A., Pistocchi, A.
- Source
- Full text @ Front Cell Dev Biol
|
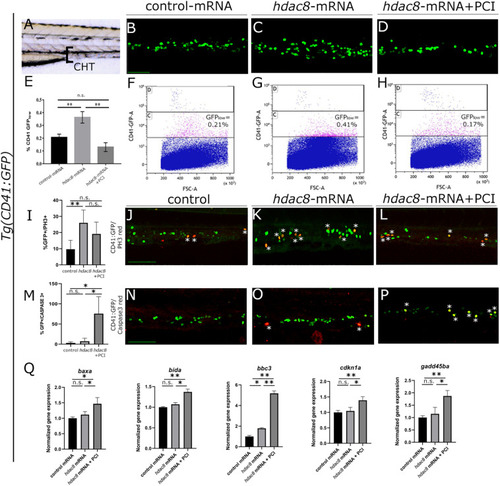
Analysis of Hdac8 overexpression in zebrafish and apoptosis induced by PCI treatment. |
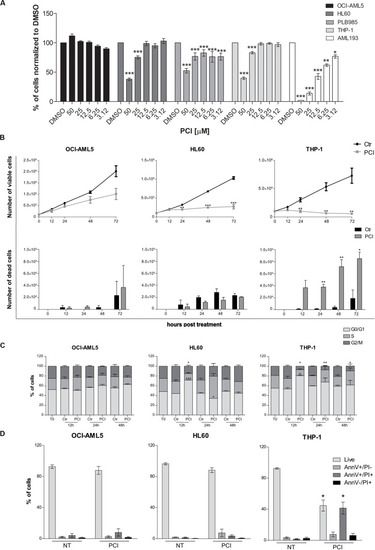
|
Cytostatic and cytotoxic effect of PCI in AML cell lines. |
|
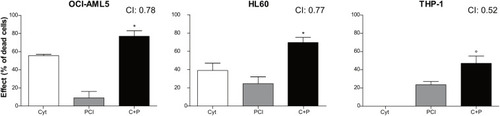
Combination treatment with cytarabine + PCI. OCI-AML5, HL60, and THP-1 cell lines were treated for 72 h with single compound and their combination. CTG assay was used to assess the effect of each treatment on the cell culture viability. The bar graph represents the effect of one of the eight concentrations tested. CI values are shown for each combination. The results are presented as mean ± SEM from two independent experiments. Asterisk (*) indicates significance of combination treatment versus PCI, circle (°) indicates significance of combination treatment versus cytarabine. One symbol, |
|
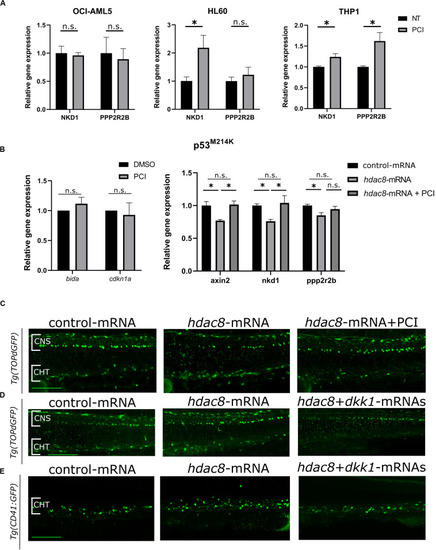
Canonical Wnt pathway modulation by HDAC8. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. EXPRESSION / LABELING:
|