FIGURE SUMMARY
- Title
-
A noncanonical function of histidyl-tRNA synthetase: inhibition of vascular hyperbranching during zebrafish development
- Authors
- Ni, R., Luo, L.
- Source
- Full text @ FEBS Open Bio
|
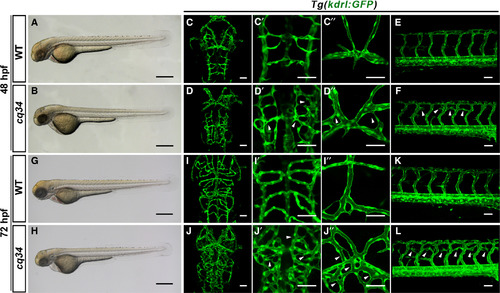
The cq34 mutants exhibit hyperbranching cranial and intersegmental blood vessels. (A,B,G,H) Bright‐filed micrographs of the cq34 mutant reveal a decreased head and thin trunk at 48 hpf, and a pericardial edema at 72 hpf. (C–F, I–L) Blood vessels are visualized using the Tg(kdrl:GFP) transgenic line. The cq34 mutants display ectopic branching (arrowheads) in the head and dorsal part of the trunk at 48 hpf, which become more severe at 72 hpf. The aberrant branching of CtAs (C′, D′, I′, J′) and dorsal cranial vasculature (C″, D″, I″, J″) can be observed clearly under higher magnification. Scale bars, 100 μm (A,B,G,H), 50 μm (C–F, I–L).
PHENOTYPE:
|
|
The gene responsible for the cq34 encodes Hars. (A) The cq34 mutation is located on the LG 14. Numbers below the SSLP markers indicate the genetic distance and the recombination events seen in 180 mutant embryos. (B) The hars genomic DNA sequence of WT and hars−/− shows a point mutation in the first base pair of intron 7–8 (substitution from G to A), causing deletion of exon 7. Exon bases are indicated in blue and intron bases in black. (C) Schematic representation of the secondary structure of Hars, which consists of WHEP, aminoacylation, and anticodon‐binding domains. The hars−/− mutant presents a partial deletion of the aminoacylation domain (amino acids: 211–243). (D) Sequencing the hars cDNA of WT and hars−/− shows the exon 7 deletion in mRNA level in hars−/−. (E) Western blotting shows the deletion in protein level in hars−/−, and α‐Tubulin serves as a loading control. (F) Whole‐mount in situ hybridization shows that the hars transcript is expressed in one‐cell stage embryos and thus is maternally supplied. It is expressed ubiquitously in the embryo at later stages. (G–K) Injection of WT hars mRNA prevents the appearance of body defects and ectopic brain vascular branching typical of the cq34 mutant at 72 hpf. (L,M) Inhibition of protein synthesis by CHX treatment between 36 and 48 hpf leads to reduced and thinner brain vasculature, and DMSO treatment was used as control. (N) In vivo Hars activity measured by ELISA assay. n = 3 tubes of lysates, mean ± SD, NS, not significant, Student's t‐test. Scale bars, 50 μm.
|
|
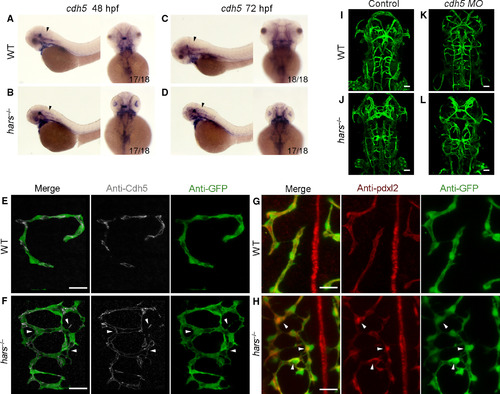
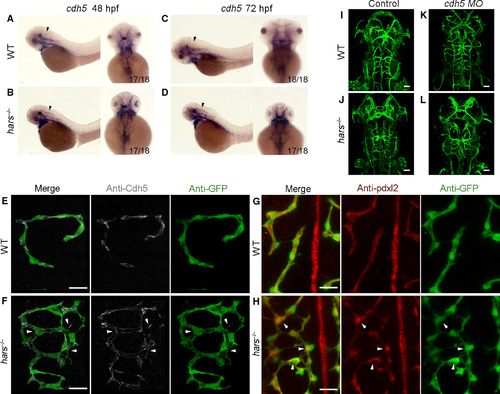
The hars mutants display disordered connections. (A–D) In situ hybridization shows that cdh5 expression is upregulated in the brain of hars−/− at 48 and 72 hpf (arrowheads). (E,F) Antibody staining shows the Cdh5 distribution overlapping with the abnormal branching connections of CtAs, and additionally, Cdh5 appears as punctate clusters at some sprouting sites (arrowheads) in hars−/− at 48 hpf. (G,H) Pdxl2 is seen to accumulate at sprouting and connection disordered sites (arrowheads) in hars−/− at 48 hpf. (I–L) Injection of 2.5 ng cdh5 MO reduces the abnormal brain vascular connections in hars−/− at 48 hpf, but the vasculature becomes immature. Scale bars, 20 μm (A,B,G,H), 50 μm (I–L).
EXPRESSION / LABELING:
PHENOTYPE:
|
|
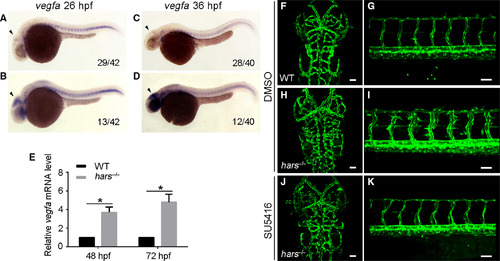
Noncanonical function of Hars represses vegfa expression. (A–D) In situ hybridization reveals that the expression of vegfa in the head is upregulated in about 25% of embryos at 26 and 36 hpf (arrowheads). (E) Quantitative real‐time PCR shows that vegfa mRNA levels in hars−/− are significantly upregulated at 48 and 72 hpf. Target transcript levels were normalized to the reference gene elf1a and to levels in WT controls, which were set to 1; n = 3 pooled biological replicates of ≥ 20 embryos, with three technical replicates per n. (mean ± standard error of the mean, *P < 0.05, two‐tailed t‐test). (F–K) SU5416 treatment suppresses ectopic branching in the brain and trunk of hars−/− at 60 hpf. Scale bars, 50 μm.
EXPRESSION / LABELING:
PHENOTYPE:
|
|
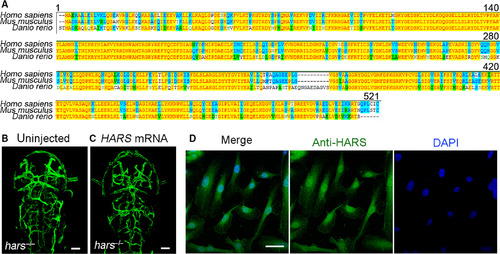
The role of Hars in regulating vascular development is functionally conserved between zebrafish and humans. (A) Hars protein sequence alignment shows high homology between the three species tested. (B,C) Injection of human HARS mRNA rescues ectopic brain vascular branching in zebrafish hars−/−, indicating a conserved regulatory function of Hars in vascular development between zebrafish and humans. (D) Confocal immunofluorescence microscopy shows nuclear localization of endogenous HARS in HUVECs. Scale bars, 50 μm.
|
Acknowledgments
This image is the copyrighted work of the attributed author or publisher, and
ZFIN has permission only to display this image to its users.
Additional permissions should be obtained from the applicable author or publisher of the image.
Full text @ FEBS Open Bio