- Title
-
In vivo imaging of cell behaviors and F-actin reveals LIM-HD transcription factor regulation of peripheral versus central sensory axon development
- Authors
- Andersen, E.F., Asuri, N.S., and Halloran, M.C.
- Source
- Full text @ Neural Dev.
|
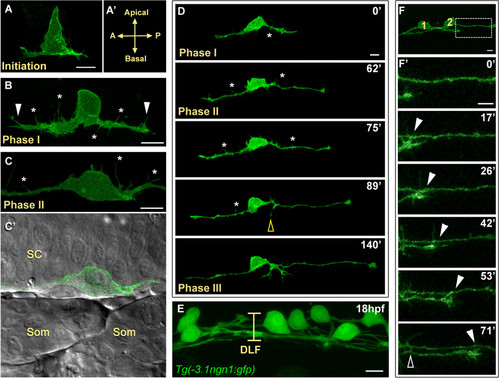
Central axon development in RB neurons. All images are dorsal-lateral views with anterior to the left and are confocal projections unless noted. Individual RB neurons labeled by transient mosaic expression of TagRFP-CAAX (pseudocolored green) (A-D) or GFP-CAAX (F) in wild-type embryos. (A) Central axon initiation. Central axons form at the basal surface of the cell body at the anterior (A) and posterior (P) sides of the cell. (A′) Indicates orientation. (B) RB neuron in phase I of central axon outgrowth. RB has thick central axons with many filopodial protrusions (asterisks) along the axon shafts and growth cones (arrowheads). (C,C′) RB neuron in phase II of central axon outgrowth. (C) The central axon shafts display transient filopodial protrusions (asterisks). Growth cones have extended out of the field of view. (C′) Single XY plane (green and DIC channel overlay) of RB in (C). The central axons grow along the edge of the spinal cord (SC) neuroepithelium. Som, somites. (D) Selected images from an approximately 3.5-hour time-lapse of RB central axon outgrowth, phases I to III. The peripheral axon initiates during phase II (yellow open arrowhead). Asterisks indicate transient filopodial protrusions during phases I and II. See Additional file 1 for a movie. (E) Image showing all RB neurons labeled in a transgenic Tg(-3.1ngn1:gfp) embryo. At 18 hpf, the dorsal longitudinal fascicle (DLF) is loosely bundled (bracket). (F,F′) Central axon fasciculation. (F) Two labeled RB neurons (1 and 2) have begun to extend central axons. Box indicates area shown in (F′). (F′) Time-lapse of RB central axon fasciculation starting at 17 hpf. RB1 growth cone contacts RB2 axon as it extends (arrowheads) and RB1 axon makes transient lateral filopodial contacts (open arrowhead). Time is displayed in minutes. Scale bars = 10 μm. |
|
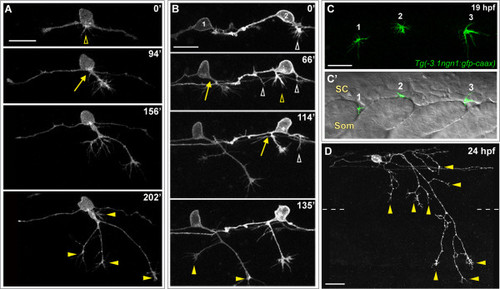
Peripheral axon development in RB neurons. (A,B,D) Individual RB neurons labeled by transient mosaic expression of TagRFP-CAAX (A,D) or GFP-CAAX (B) in wild-type embryos. Dorsal-lateral views, anterior is left. Images are confocal projections. (A) Time-lapse of RB peripheral axon outgrowth from the cell body. Several filopodial protrusions occur in the cell body region during peripheral initiation (yellow open arrowhead). The peripheral axon forms off the cell body (yellow arrow), exits the spinal cord (SC) and grows in a ventral direction while branching (yellow arrowheads indicate branch tips) in the epidermis. (B) Time-lapse of two RB neurons (1 and 2) showing peripheral axon emergence as central axon branches. RB1: a peripheral axon initiates off the descending central axon (yellow arrow), and grows ventrally while extending branches (yellow arrowheads). RB2: multiple neurites initiate as branches off both ascending and descending central axons (open arrowheads); however, only one becomes an established peripheral axon (yellow open arrowhead and arrow), and the others retract (white open arrowheads). See Additional file 2 for movie. Time displayed in minutes (A,B). (C,C′) Lateral views of a 19-hpf transgenic Tg(-3.1ngn1:gfp-caax) embryo showing the anterior-posterior position where peripheral axons exit the SC. (C) Confocal projection (green channel) of four 1-μm Z-planes showing newly formed peripheral growth cones of three RB neurons (1, 2, and 3). (C′) Single XY plane (green and DIC channel overlay) reveals sites of peripheral axon emergence. Growth cones 1 and 3 extend between somites (Som). Growth cone 2 grows over the somite. (D) Example of mature RB neuron morphology in a 24-hpf wild-type embryo. RB has an elaborated peripheral axon with many branches (some indicated with yellow arrowheads). Dashed lines indicate the position of the horizontal myoseptum. Scale bars = 25 μm. |
|
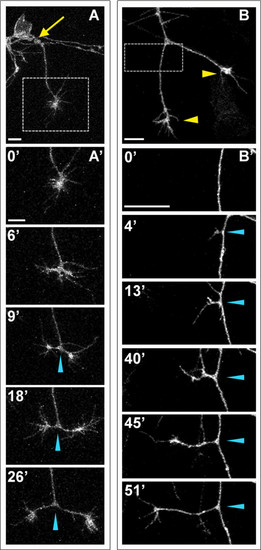
Peripheral axons branch by bifurcation and interstitial branching. Individual RB neurons labeled by transient mosaic expression of GFP-CAAX in wild-type embryos. Lateral views, anterior is left. Images are confocal projections. (A) An unbranched peripheral axon formed at a site posterior to the RB cell body (yellow arrow). Box shows area imaged in (A′). (A′) Time-lapse of peripheral axon bifurcation (blue arrowheads indicate branch point). (B) A branched peripheral axon with growth cones extending ventrally (yellow arrowheads). Box shows area imaged in (B′). (B′) Time-lapse of interstitial branch formation (blue arrowheads indicate branch point). Time is displayed in minutes. Scale bars = 10 μm. |
|
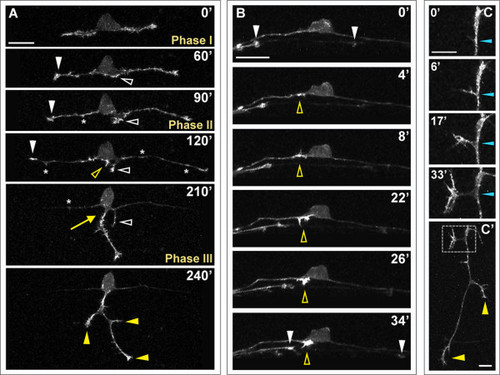
F-actin distribution in developing RB neurons. F-actin labeled in individual RB neurons by transient mosaic expression of mCh-UtrCH in wild-type embryos. Dorsal-lateral views, anterior is left. Images are confocal projections. (A) Time-lapse showing F-actin distribution during RB neuron development. F-actin is concentrated in the growth cones of central axons (white arrowheads) and peripheral axon branches (yellow arrowheads) as they extend. Accumulation of F-actin occurs at the basal edge of the cell body and becomes concentrated at the distal tip of a neurite that initiates posterior to the cell body and later retracts (white open arrowheads). A second neurite initiates directly off the cell body (yellow open arrowhead) and becomes established as the peripheral axon (yellow arrow). F-actin signal is distributed along the central axon shafts during phase I, but is diminished in the axon shaft during phases II and III. Transient F-actin patches occur along the central axon shafts during phases II and III (some indicated with asterisks) and decrease in frequency over time. See Additional file 3 for movie. (B) Time-lapse showing F-actin accumulation during peripheral axon initiation (open yellow arrowheads). White arrowheads indicate central axon growth cones of neighboring labeled RB neurons extending through the field of view. (C) Time-lapse showing F-actin dynamics during interstitial branch formation in a peripheral axon. F-actin accumulates in a filopodium (0′, 6′) then concentrates in the growth cone of the extending branch (17′, 33′). Blue arrowheads indicate branch point. (C′) Low magnification view of branched peripheral axon at the end of the time-lapse. Box indicates region shown in (C). Yellow arrowheads indicate peripheral growth cones. Time is displayed in minutes. Scale bars in (A,B) = 25 μm, in (C) = 10 μm. |
|
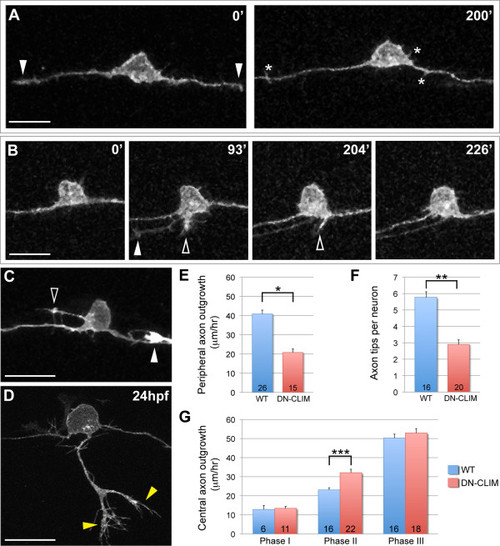
Effects of LIM-HD disruption on RB axon development. (A-D) Individual neurons labeled by transient mosaic expression of TagRFP-CAAX in DN-CLIM-expressing embryos. Dorsal-lateral views, anterior is left. Images are confocal projections. (A) Two images from a time-lapse in a DN-CLIM embryo. Arrowheads indicate central growth cones at 0′. No peripheral axons initiate. Transient filopodia form off cell body and axon shafts (asterisks). See Additional file 4 for movie. (B) Time-lapse in a DN-CLIM-expressing embryo. A peripheral axon initiates, but fails to extend. Transient neurites form in two locations (open arrowheads) and subsequently retract. Arrowhead at 93′ indicates growth cone of neighboring central axon growing through the field of view. (C) Ectopic neurite formation in a DN-CLIM-expressing embryo. A long filopodial protrusion extends from an ectopic apical location on the cell body (open arrowhead). The arrowhead indicates the growth cone of neighboring central axon that enters the field of view. (D) Example of a peripheral axon in a 24-hpf DN-CLIM-expressing embryo. The axon is shorter and less branched than the wild type (compare to Figure 2D; horizontal myoseptum in Figure 5D is out of frame). Yellow arrowheads indicate branches. See Additional file 5 for movie. (A-D) Time displayed in minutes. Scale bars = 25 μm. (E) Quantification of average central axon extension rates in wild-type (WT) versus DN-CLIM-expressing embryos. Central axon extension rates are significantly faster in DN-CLIM embryos during phase II. (F,G) Quantifications of average peripheral axon extension rates (F) and average number of branches per peripheral axon (G) in wild-type versus DN-CLIM-expressing embryos. *P < 10-7, **P < 10-6, and ***P = 0.0004 (two-tailed t-test). Error bars represent standard error of the mean. Numbers in bars signify number of neurons. |
|
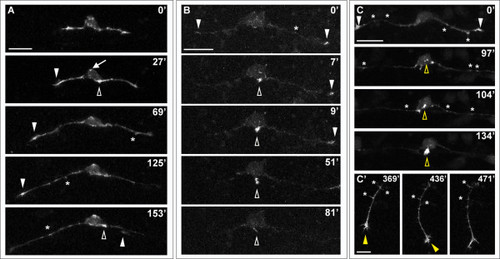
Effects of LIM-HD disruption on F-actin distribution in developing RB neurons. Time-lapse sequences of F-actin distribution in DN-CLIM-expressing embryos. F-actin is labeled by transient mosaic expression of mCh-UtrCH. Dorsal-lateral views, anterior is left. Images are confocal projections. (A) Neuron in which no peripheral axon initiates. F-actin accumulates transiently near the cell body (open arrowheads) and in patches along central axons (some indicated with asterisks). F-actin also localizes to central growth cones (white arrowheads; including neighboring central growth cone extending through field at 153′). Ectopic F-actin signal occurs at the apical edge of the cell body (example indicated with arrow at 27′). See Additional file 6 for movie. (B) Neuron in which peripheral neurite with strong F-actin signal initiates from the cell body (open arrowhead), but retracts. F-actin is also visible in central growth cones (arrowheads) and transiently along the central axon shaft (example indicated with asterisk). See Additional file 7 for movie. (C,C′) Neuron in which peripheral axon initiates and extends into the periphery. (C) F-actin accumulation is visible in growth cones of central axons (arrowheads), which extend out of the field of view, and transiently along the central axon shaft (some indicated with asterisks). Accumulation of F-actin occurs in an ectopic apical location of the cell body and becomes concentrated at the initiating neurite tip (open yellow arrowheads). (C′) Shifted focal plane shows established peripheral axon extending ventrally without branching (yellow arrowhead indicates growth cone). Asterisks indicate some F-actin-rich filopodial protrusions along the peripheral axon shaft. Time is displayed in minutes. Scale bars in (A-C) = 25 μm, in (C′) = 10 μm. |