- Title
-
Reactivation of Notch signaling is required for cardiac valve regeneration
- Authors
- Kefalos, P., Agalou, A., Kawakami, K., Beis, D.
- Source
- Full text @ Sci. Rep.
|
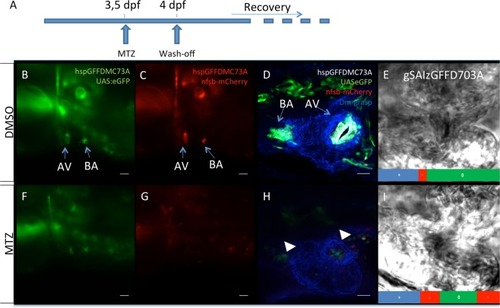
Chemically induced genetic cell ablation of zebrafish larval cardiac valves. ( |
|
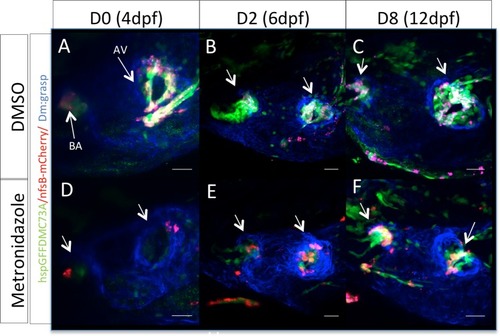
Zebrafish embryonic valves can regenerate. ( |
|
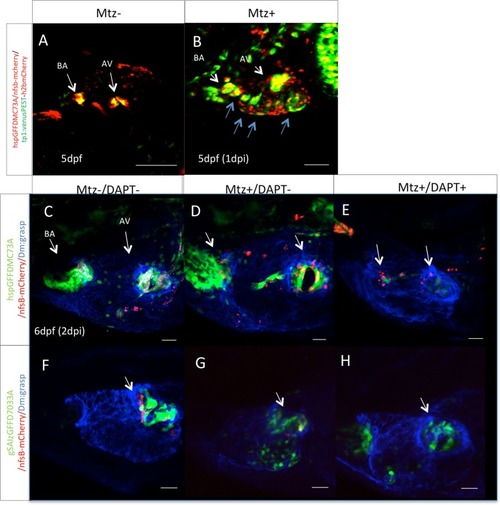
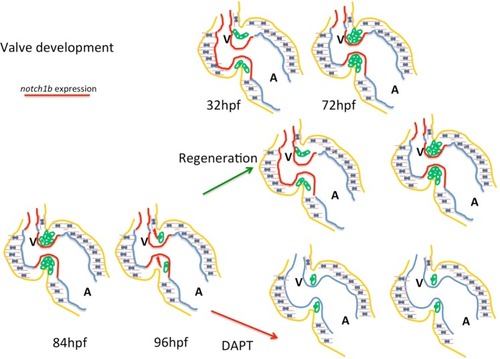
Notch signaling is activated during valve regeneration. |
|
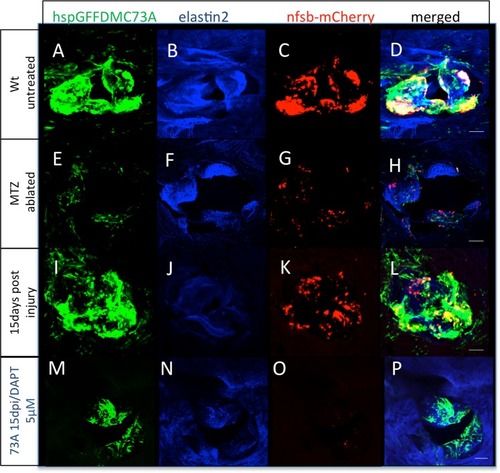
Zebrafish adult valves retain the ability to regenerate through Notch signaling reactivation. ( |
|
Sensing of immature intracardiac flow patterns activates a Notch mediated regenerative program, recapitulating cardiac valve development. At embryonic developmental stages a feedback loop where intracardiac flow dynamics regulate |