- Title
-
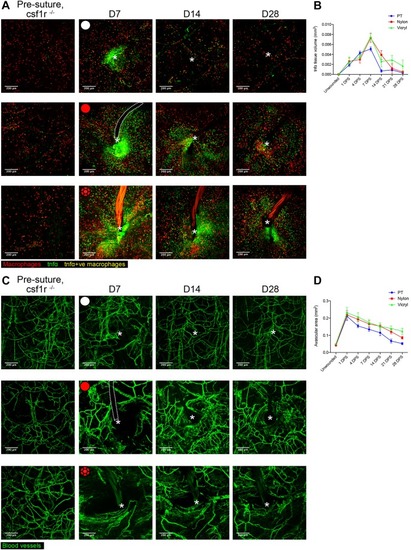
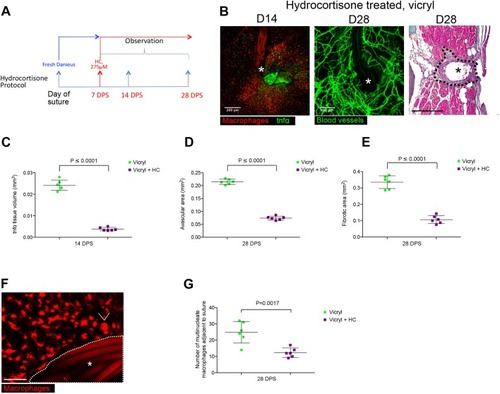
Live imaging the Foreign Body Response reveals how dampening inflammation reduces fibrosis
- Authors
- Gurevich, D.B., French, K.E., Collin, J.D., Cross, S.J., Martin, P.
- Source
- Full text @ J. Cell Sci.
|
|
|
|
|
|
|
|
|
PHENOTYPE:
|
|
PHENOTYPE:
|