- Title
-
A role for planar cell polarity during early endoderm morphogenesis
- Authors
- Miles, L.B., Mizoguchi, T., Kikuchi, Y., Verkade, H.
- Source
- Full text @ Biol. Open
|
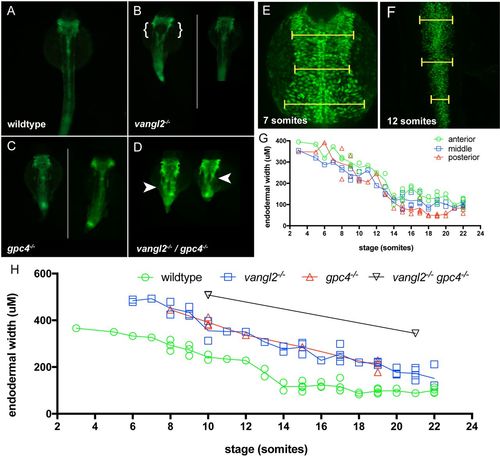
Loss of PCP signalling results in disrupted endoderm morphology during development. (A-D) Fluorescent dissecting microscope images of endoderm morphology at 24 hpf. All views are dorsal. (A) Wild-type and PCP homozygous mutant embryos (B) vangl2, (C) gpc4, and (D) vangl2l/gpc4 double mutants in the Tg(sox17:EGFP) background. Endoderm morphology in PCP mutants (B, C and D) is disorganised and wider than in wild-type (A). Disruption of PCP signalling in the single mutants occasionally results in a splitting of the endoderm posterior to the pharyngeal endoderm as indicated by the brackets in B. vangl2/gpc4 double mutant embryos always have a splitting of the endoderm posterior to the pharyngeal endoderm as indicated by the arrow heads in D. (E-F) Confocal projections of 7 somite (E) and 12 somite (F) wild-type Tg(sox17:EGFP) embryos. Dorsal views. Yellow bars indicate positions of the three width measurements. (G) The rate of change of the width of the endoderm, measured at anterior, middle, and posterior regions of the trunk endoderm, migrate with the same overall rate towards the dorsal midline during somitogenesis stages. (H) The endoderm is wider in homozygote vangl2 and gpc4 mutants and vangl2/gpc4 double mutant lines, although dorsal migration does still take place (n = 1-5 embryos per stage, per genotype). White lines in panel C and E divide grouped individual embryos. |
|
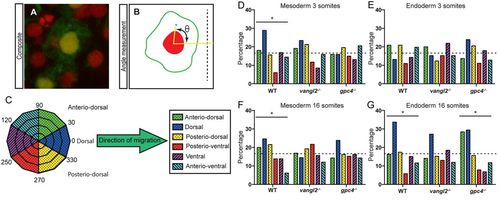
MTOC distribution of mesoderm and endoderm during somitogenesis stages. (A) Composite confocal micrograph demonstrating an EGFP-positive endoderm cell with red nucleus and blue MTOC. (B) Schematic angle measurement (θ) of the MTOC relative to the centre of the nucleus and the dorsal midline (dotted line). (C) Schematic highlighting the grouping of MTOC data into six direction groups. (D-G) Graphs showing the percentage of MTOC in each of the six segments. (D) Mesoderm 3 somites stage. (E) Endoderm 3 somites stage. (F) Mesoderm 16 somites stage. (G) Endoderm 16 somites stage. Dotted line indicates equal (randomised) mean value. Asterisk represents significant difference by χ squared test, *P≤0.05. See Table S1 for χ squared values testing for equal MTOC distributions over the six segments (null hypothesis is randomised data) and with wild type observed values as expected values (null hypothesis is data matching wild type). See Table S2 for cell number and embryo number analysed. |
|
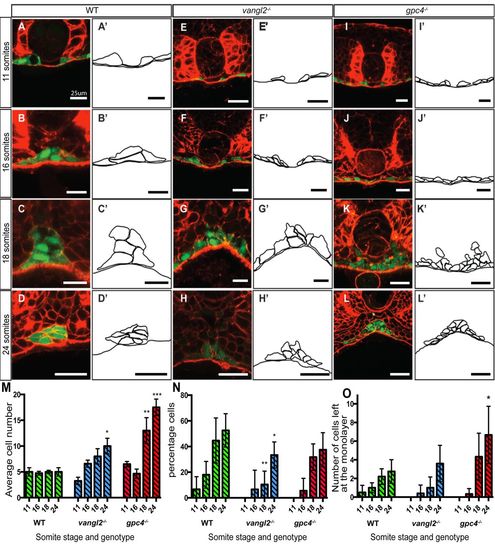
Midline aggregation of endodermal cells in wild-type and PCP mutant lines. (A-L). Confocal images of sections stained with Rhodamine-Phalloidin to show F-actin (red) at 11 somites, 16 somites, 18 somites, and 24 somites. Endoderm labelled in green by Tg(sox17:EGFP). Vibratome sections are in the region of the trunk endoderm to control for A-P position. (A′-L′) Schematic representations of the endoderm at each stage. (A-D) Wild-type embryos. (E-H) vangl2−/− embryos, and (I-L) gpc4−/− embryos, showing a wider endodermal region and an increase in endodermal cell number. Scale bar: 25 μm. (M) Mean cell number at the dorsal midline from 11 somites to 24 somites of wild-type (green bars), vangl2−/− (blue bars), and gpc4−/− (red bars) embryos. (N) Percentage of endodermal cells that have left the monolayer in wild-type (green bars), vangl2−/− (blue bars), and gpc4−/− (red bars) embryos. (O) Total number of endodermal cells that have left the monolayer in wild-type (green bars), vangl2−/− (blue bars), and gpc4−/− (red bars) embryos (n=2-8). Error bars represent s.e.m. Asterisk represents significant as determined by t-test, *P≤0.05, **P≤0.01, ***P≤0.001. |