- Title
-
Expression profiling during ocular development identifies 2 Nlz genes with a critical role in optic fissure closure
- Authors
- Brown, J.D., Dutta, S., Bharti, K., Bonner, R.F., Munson, P.J., Dawid, I.B., Akhtar, A.L., Onojafe, I.F., Alur, R.P., Gross, J.M., Hejtmancik, J.F., Jiao, X., Chan, W.Y., and Brooks, B.P.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
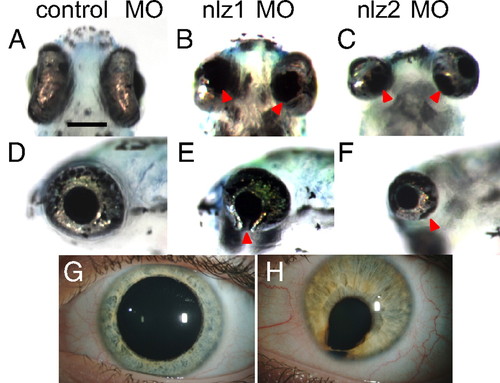
Eye phenotype in nlz1 and nlz2 morphant zebrafish. At 5 dpf, fish injected with control MO show a completely fused optic fissure in the ventral eye (A, ventral; D, lateral) whereas there is an obvious coloboma in the ventral eye of the nlz1 (B, ventral; E, lateral) and nlz2 (C, ventral; F, lateral) morphant fish indicated at arrowheads. This phenotype recapitulates human uveal coloboma (H). Unaffected human eye (G). (Scale bar, 200 μm.) PHENOTYPE:
|
|
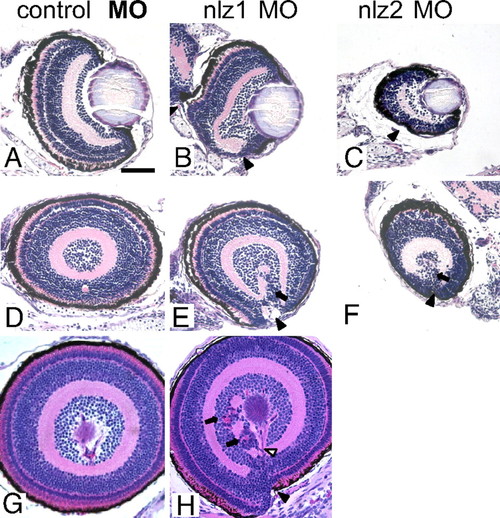
Histopathology of nlz morphant fish. Coronal (A–C) and sagittal (D–H) planes through the zebrafish eye. Normal retinal lamination and a fused ventral fissure can be seen in control MO injected fish at 5 dpf (A and D) and at 6 dpf (G) (Top, dorsal; Bottom, ventral). Fissure closure defects can be seen in nlz1 morphant fish at 5 dpf (B and E) and 6 dpf (H) and nlz2 morphant fish at 5 dpf (C and F). The coloboma in morphant fish is accompanied by discontinuous RPE (black arrowhead), retinal dysplasia/rosettes (arrows), and abnormal vasculature (open arrowhead). H&E staining. |
|
Phenotypic grades of nlz morphant fish 24 hpf. nlz1 MO (A–C) or nlz2 MO (D–F) splice site MO injected fish were scored as having a grade 1 (A and D), grade 2 (B and E), or grade 3 (C and F) optic fissure defect. At 24 hpf, the margins of the optic fissure are apposed in normal eyes (G) and closure is just beginning. Bars delineate edges of optic fissure. (Scale bar, 50 μm.) The results of the phenotype profiling are summarized in the bar graph (J) where lower grade closure defects are observed at low doses of either nlz1 or nlz2 MO, yet an effect which is at least additive is seen when these doses are combined. A higher dose of either morpholino produces strong optic fissure defects in morphant fish, which is partially rescued by coinjection of respective wild-type mRNA. Expression of nlz1 (H) and nlz2 (I) in wild-type zebrafish embryos at 24 hpf shows expression at the optic fissure (arrowheads). |
|
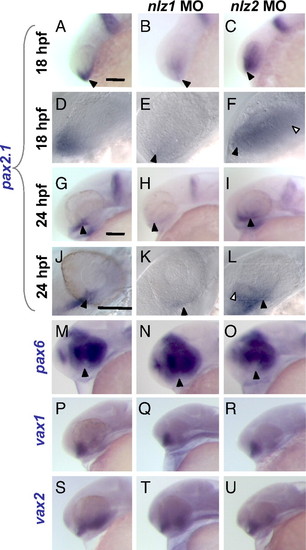
Misregulation of pax2.1 and pax6 in nlz morphant fish in the setting of normal vax1 and vax2 expression at 24 hpf. In situ hybridization at 18 hpf (A–F) and 24 hpf (G–U). At 18 hpf, pax2.1 is normally expressed in the optic stalk and ventral optic primordium (A and D, arrowheads) and at 24 hpf is expressed distinctly in the optic stalk and fissure (G and J, arrowheads), midbrain/hindbrain boundary, and otic vesicle. In nlz1 morphant fish, pax2.1 expression is drastically reduced in the optic stalk and fissure (B, E, H, and K). In nlz2 morphant fish, the expression domain is expanded (C, F, I, L, open arrowheads). A higher magnification view shows the expression of pax2.1 in normal and nlz morphant fish at 18 hpf (D–F) and 24 hpf (J–L). (M–O) pax6 expression shows a subtle increase in the ventral eye in nlz1 morphant fish (N) and slight decrease in nlz2 fish (O, arrowheads). vax1 (P–R) and vax2 (S–U) are both expressed at normal levels and in a normal distribution in the optic stalk and ventral retina. (Scale bar, 100 μm.) EXPRESSION / LABELING:
|
|
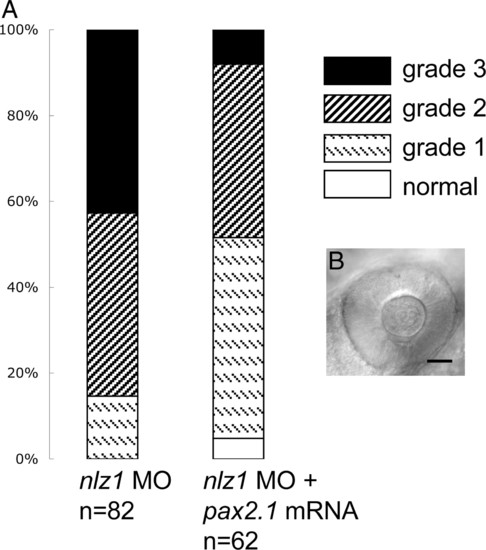
pax2.1 partially rescues nlz1 morphant phenotype. (A) Coinjection of pax2.1 mRNA shifts the coloboma phenotype profile toward normal, decreasing the percentage of embryos in cohort with severe defects (grade 3) and increasing the percentage of mildly affected (grade 1) and normal eyes. (B) Representative picture of nlz1 morphant eye at 24 hpf rescued by pax2.1 injection. PHENOTYPE:
|
|
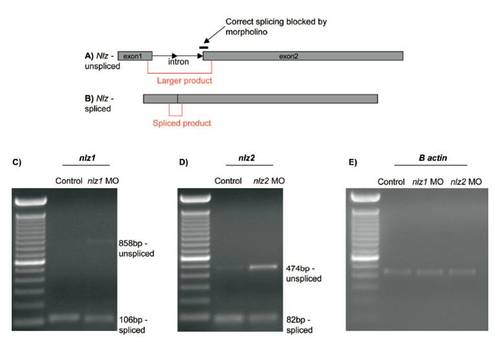
RT-PCR confirms correct targeting of splice site blocking morpholinos and knockdown of target transcript at 24 hpf. Diagram depicting the target site of nlz1 and nlz2 splice blocking morpholinos at the intron 1–exon 2 boundary and PCR primer positions (red) spanning the intron in unspliced (A) and spliced (B) transcripts. (C) Agarose gel showing reduction of smaller spliced nlz1 product (106 bp) and appearance of unspliced product (858 bp) in embryos injected with nlz1 MO.(D) Agarose gel showing reduction of smaller spliced nlz2 product (82 bp) and appearance of more robust unspliced product (474 bp) in embryos injected with nlz2 MO. (E) Actin control RT-PCR in control, nlz1, and nlz2 MO injected embryos. |