- Title
-
YB-1 recruitment to stress granules in zebrafish cells reveals a differential adaptive response to stress
- Authors
- Guarino, A.M., Mauro, G.D., Ruggiero, G., Geyer, N., Delicato, A., Foulkes, N.S., Vallone, D., Calabrò, V.
- Source
- Full text @ Sci. Rep.
|
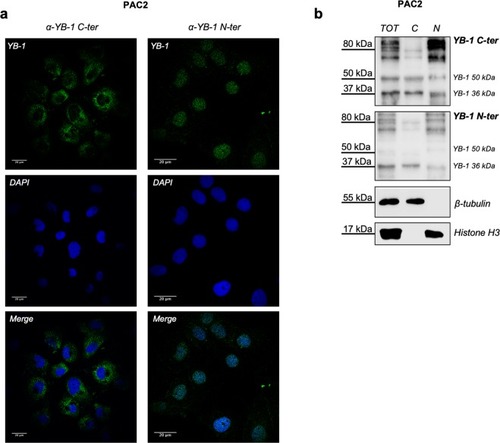
Immunoreactivity of human YB-1 antibodies against zebrafish YB-1. ( |
|
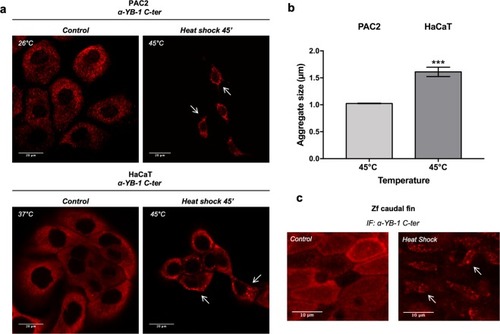
Heat shock promotes assembly of YB-1 positive aggregates in zebrafish and mammalian cells. |
|
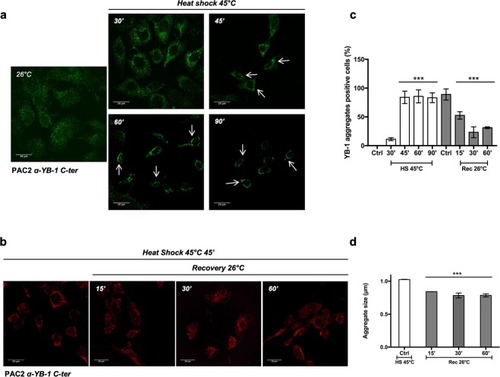
Kinetics of YB-1 aggregate formation. ( |
|
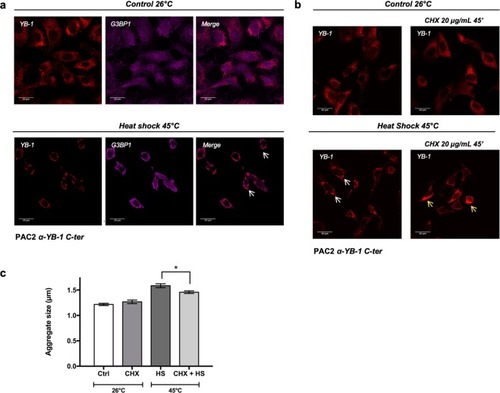
Heat shock induced YB-1 aggregates represent |
|
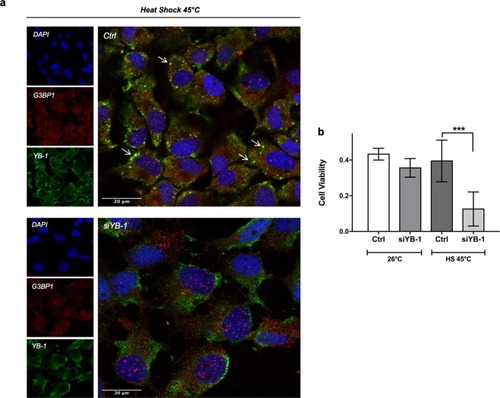
YB-1 is essential for SG formation and PAC2 cell viability upon thermal stress. ( |