- Title
-
Biallelic CHP1 mutation causes human autosomal recessive ataxia by impairing NHE1 function
- Authors
- Mendoza-Ferreira, N., Coutelier, M., Janzen, E., Hosseinibarkooie, S., Löhr, H., Schneider, S., Milbradt, J., Karakaya, M., Riessland, M., Pichlo, C., Torres-Benito, L., Singleton, A., Zuchner, S., Brice, A., Durr, A., Hammerschmidt, M., Stevanin, G., Wirth, B.
- Source
- Full text @ Neurol Genet
|
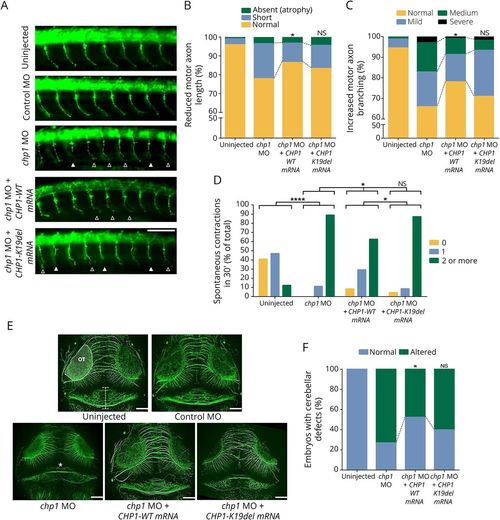
Chp1 downregulation in zebrafish leads to CaP-MN, cerebellar, and movement defects rescued with WT, but not mutant CHP1 mRNA (A) Lateral views of whole-mount embryos stained with synaptotagmin antibody (znp1). Truncated and absent motor axons are indicated with solid arrowheads and terminally branched axons with open arrowheads. Scale bar: 25 μm. (B and C) Based on overall appearance, CaP-MNs were classified as follows: normal, short (truncated axonal projection), or absent (total axonal atrophy). Based on terminal branching, axons were classified as normal, mild (branching ventral from midline), medium (2–3 or more branches at ventral or midline), or severe (>3 branches ventral or dorsal from midline). chp1-depleted zebrafish larvae display CaP-MN truncations and increased terminal branching in comparison with control (non-targeting) Morpholino (MO) and uninjected fish. Quantitative analysis of CaP-MN demonstrates that coinjection of chp1 MO and CHP1-WT mRNAs significantly improves axonal defects. Coinjection of chp1 MO and CHP1-K19del mRNAs fails to ameliorate CaP-MN defects in comparison with CHP1-WT mRNA. Results are presented in percentages from n ≥ 150 analyzed axons representing 3 independent experiments. * denotes statistical significance in comparison with the chp1 MO group (p ≤ 0.05 χ2 test). (D) chp1-depleted ∼34 hpf morphants show an increased frequency of spontaneous contractions. For graphic representation, spontaneous contractions were categorized as follows: 0 (no contraction), 1 (1 contraction in 30 seconds), and 2 or more (2 or more contractions in 30 seconds). Results are presented as the percentage of total from n ≥ 50 observed zebrafish larvae representing 3 independent experiments. Coinjection of chp1 MO and CHP1-WT mRNAs, but not CHP1-K19-del mRNA, significantly reduced spontaneous contractions in 30 seconds “****” denotes statistical significance (p < 0.0001 Fisher exact test). (E) Dorsal views of whole-mount embryos labeled with acetylated tubulin at ∼72 hpf. The structure depicted between white horizontal lines in the uninjected morphant indicates the cerebellar area of observation. Optic tectum (OT) is outlined. Severe cerebellar hypoplasia is observed in chp1-depleted zebrafish in comparison with control MO and uninjected fish. “*” in chp1 MO image indicates trochlear decussation. Scale bar: 25 μm. (F) Quantitative analysis of cerebellar defects demonstrates that coinjection of chp1 MO and CHP1-WT mRNAs significantly ameliorates this phenotype. Coinjection of chp1 MO and CHP1-K19del mRNAs fails to improve cerebellar defects to WT levels. Results are presented in percentages from n ≥ 40 observed zebrafish morphants from 3 independent experiments. “*” denote statistical significance (p ≤ 0.05 Fisher exact test). CaP-MN = caudal primary motor neuron; mRNA = messenger RNA; NS = not significant; WT = wild type. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|