- Title
-
Exome Sequence Analysis Suggests that Genetic Burden Contributes to Phenotypic Variability and Complex Neuropathy
- Authors
- Gonzaga-Jauregui, C., Harel, T., Gambin, T., Kousi, M., Griffin, L.B., Francescatto, L., Ozes, B., Karaca, E., Jhangiani, S.N., Bainbridge, M.N., Lawson, K.S., Pehlivan, D., Okamoto, Y., Withers, M., Mancias, P., Slavotinek, A., Reitnauer, P.J., Goksungur, M.T., Shy, M., Crawford, T.O., Koenig, M., Willer, J., Flores, B.N., Pediaditrakis, I., Us, O., Wiszniewski, W., Parman, Y., Antonellis, A., Muzny, D.M., Baylor-Hopkins Center for Mendelian Genomics, Katsanis, N., Battaloglu, E., Boerwinkle, E., Gibbs, R.A., Lupski, J.R.
- Source
- Full text @ Cell Rep.
|
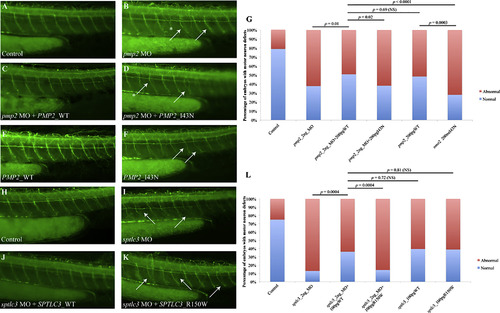
Suppression of pmp2 and sptlc3 in Zebrafish Causes Defects in Motor Axon Pathfinding and Outgrowth (A–F) Lateral views of a control embryo, an embryo injected with pmp2 morpholino (MO) and embryos injected with pmp2 MO+PMP2_WT and pmp2 MO+PMP2_I43N, PMP2_WT and PMP2_I43N cocktails, respectively, at 2 dpf (days post fertilization). Controls showed even spacing and normal branching of the motor neuron axons (A). In the pmp2 MO-injected embryos, the spacing of neuronal axons is perturbed by exiting the periphery but failing to extend (asterisks) or presenting pathfinding errors (arrows; B). Co-injection of pmp2 MO with human PMP2_WT resulted in restoration of the normal neuronal phenotype (C), but PMP2_I43N did not (D). Overexpression of human PMP2_WT causes mild pathfinding errors (E), suggesting dose sensitivity for PMP2. However, the human PMP2 mutant p.I43N was significantly more severe than PMP2_WT when overexpressed (F) and had similar effects to suppression of pmp2 by MO knockdown. (G) Percentage of normal versus abnormal embryos under the conditions being evaluated above. (H–K) Wild-type embryos (H) and sptlc3 morphants (I) in which secondary axons fail to migrate appropriately (white arrows). The phenotype induced by suppression of sptlc3 could be rescued by co-injection with SPTLC3_WT (J) but not SPTLC3_R150W (K). (L) Quantification of normal embryos versus embryos with motor neuron axon defects. For statistical analyses, Χ2 tests were performed. |
|
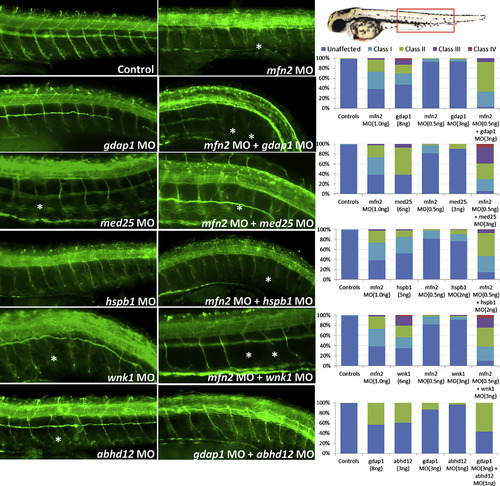
Functional Assessment of Mutation Burden Hypothesis in a Zebrafish Model First and second column panels show representative images of acetylated-tubulin (α-AcTub) staining of peripheral neurons in 2-day MO knockdown, single or in pairwise combinations, zebrafish larvae. Third column panel shows qualitative assessment of morphant fish evaluated as defects in peripheral neuron axon extension, branching or pathfinding according to the scoring system developed. For pairwise combinations, sub-effective concentrations of each of the gene-specific MOs were injected as shown in the graphs by the number of abnormal larvae in each category. However, when injected together increased severity in the phenotype was observed for all the pairwise combinations, suggesting in vivo epistatic effects between these pairs of genes as observed in the α-AcTub fluorescence images and quantified in the graphs. Asterisks highlight some evidently affected axons. The scoring system used for assessing PNS defects in zebrafish was developed ad hoc and implemented here in order to best reflect the observations resulting from our experiments. Class I category refers to single axon defects; class II category refers to two or more axons exhibiting defects with the presence of some normal axons; class III category refers to generalized axonal defects; class IV category refers to complete absence of axonal extension. |
|
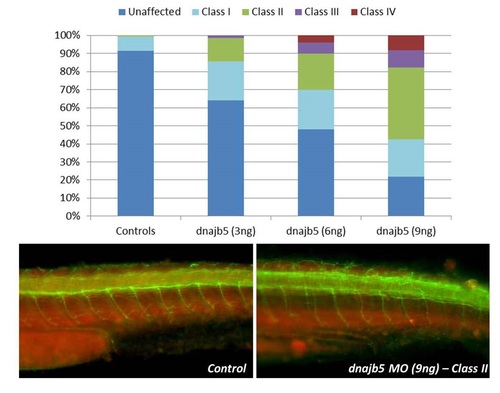
Evaluation of dnajb5 MO knockdown phenotype in zebrafish by Acetylated Tubulin staining of neurons and axons (green) and muscle (red). Gross axonal defects are observed in MO injected embryos at 2dpf. (Related to Figure 2) |