- Title
-
A dual role for ErbB2 signaling in cardiac trabeculation
- Authors
- Liu, J., Bressan, M., Hassel, D., Huisken, J., Staudt, D., Kikuchi, K., Poss, K.D., Mikawa, T., and Stainier, D.Y.
- Source
- Full text @ Development
|
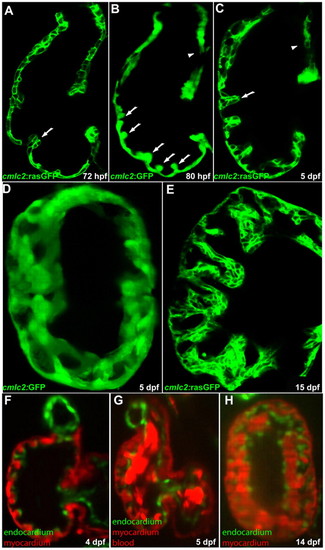
Cardiac trabeculation in zebrafish. (A) Cardiac trabeculae start to form in the ventricle around 72 hpf (arrow). (B) By 80 hpf, cardiac trabeculation has become more pronounced (arrows), with trabeculae distributed exclusively along the luminal side of the outer curvature of the ventricle. (C) The ventricle becomes extensively trabeculated by 5 dpf, but the inner curvature (arrowhead) remains smooth. (D) Cardiac trabeculae form ridges into the lumen as shown by 3D reconstruction. (E) The trabeculae continue to expand as shown at 15 dpf. (F-H) SPIM imaging shows that cardiac trabeculation increases in complexity (see corresponding movies in the supplementary material). EXPRESSION / LABELING:
|
|
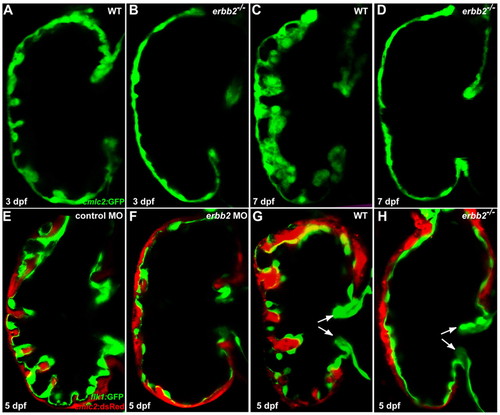
ErbB2 signaling is essential for cardiac trabeculation in zebrafish. (A,B) Trabeculae start to form in wild-type larvae (A) at 3 dpf but are absent in erbb2 mutants (B). (C,D) The wild-type ventricle (C) is extensively trabeculated at 7 dpf, whereas trabeculae remain absent in erbb2 mutants (D) at the same stage. (E,F) Injection of an antisense morpholino oligonucleotide (MO) designed against erbb2 caused a significant reduction in cardiac trabecular formation (F), whereas a mismatch control MO had no effect (E). (G,H) Cardiac valve formation (arrows) appears to be unaffected in erbb2 mutants (H) compared with wild-type siblings (G). PHENOTYPE:
|
|
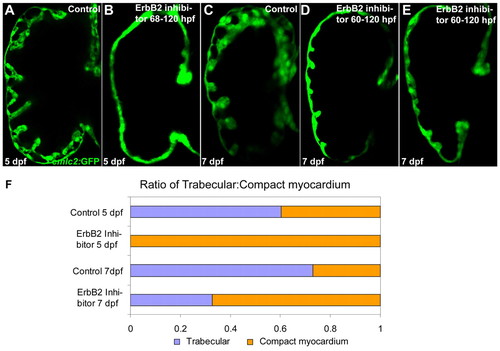
ErbB2 signaling from 68-72 hpf is required for the initiation of cardiac trabeculation. (A,B) Extensive cardiac trabeculation in wild-type control (A) at 5 dpf and absence of cardiac trabeculation in wild-type larvae (B; data not shown) treated with 5 μM AG1478 from 60, 63 or 68 to 120 hpf. (C-E) Some cardiac trabeculae formed in wild-type larvae that were allowed to develop until 7 dpf after treatment with 5 μM AG1478 from 60-120 hpf. (F) Quantification of cardiac trabecular formation in control and AG1478-treated larvae, as determined by measuring the surface area of the trabecular myocardium and the compact myocardium (larvae treated as in A-E). |
|
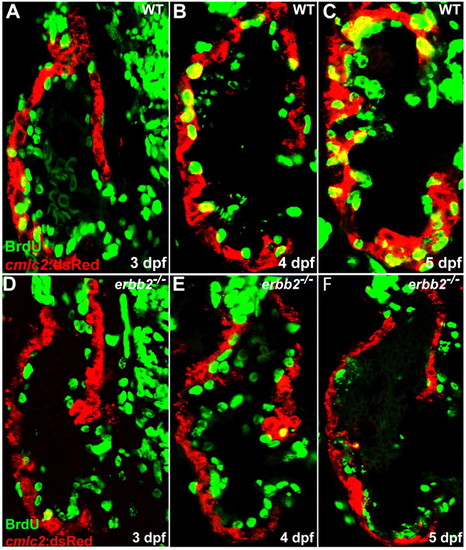
ErbB2 is required for cardiomyocyte proliferation. (A-C) Cardiomyocyte proliferation, as assessed by BrdU incorporation in cardiomyocytes (yellow), increases over time in wild-type animals from 3-5 dpf. (D-F) Cardiomyocyte proliferation appears to be absent in erbb2 mutants during these stages. PHENOTYPE:
|
|
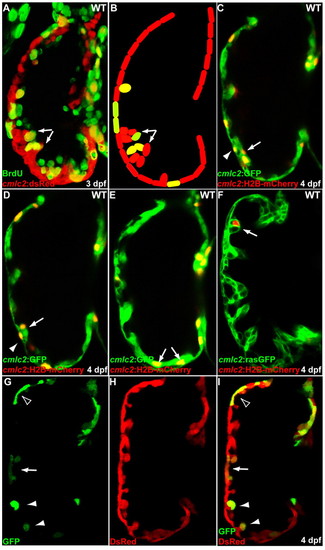
Initiation of cardiac trabeculation appears to involve directional cardiomyocyte migration (delamination) rather than oriented cell division. (A,B) Tg(cmlc2:dsRed) embryos at 60 hpf were exposed to BrdU for 24 hours. BrdU labeling shows that not all trabecular cells (arrows) are BrdU-positive. (C-F) cmlc2:H2B-mCherry reporter construct was injected into one-cell-stage Tg(cmlc2:GFP) (C-E) or Tg(cmlc2:rasGFP) (F) embryos and the distribution of mCherry-positive cardiomyocytes was monitored at 4 dpf. (C,D) Two optical sections of the same heart show that the lineage marker mCherry labels compact layer cardiomyocytes (arrowheads) as well as some of their immediate trabecular neighbors (arrows). (E,F) Trabecular cardiomyocytes (arrows), but not their compact layer neighbors, are labeled in another heart. (G-I) Genetic clones generated by crossing Tg(cmlc2:CreER) fish with Tg(bactin2:loxP-DsRed-STOP-loxP-GFP) reporter animals were found to contain both compact and trabecular cardiomyocytes (arrow), only compact cardiomyocytes (empty arrowhead) or only trabecular cardiomyocytes (arrowheads). |
|
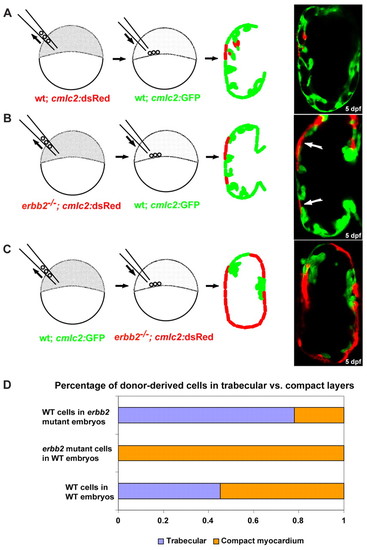
ErbB2 is required cell-autonomously in cardiomyocytes for the initiation of cardiac trabeculation. (A,B) Chimeric ventricles resulting from transplanting wild-type (A) or erbb2 mutant (B) blastomeres from Tg(cmlc2:dsRed) donors into Tg(cmlc2:GFP) wild-type hosts. Wild-type donor cells were detected in both the compact and trabecular layers, whereas erbb2 mutant donor cells failed to incorporate into the trabeculae (arrows). (C) Chimeric ventricles resulting from transplanting wild-type blastomeres from Tg(cmlc2:GFP) donors into Tg(cmlc2:dsRED); erbb2 mutant hosts. Note that the trabeculae only contain wild-type cells. (D) Quantification of the distribution of the wild-type and mutant donor-derived cells in the compact versus trabecular layers in the chimeric ventricles, assessed by measuring the surface area of the donor-derived cells. |
|
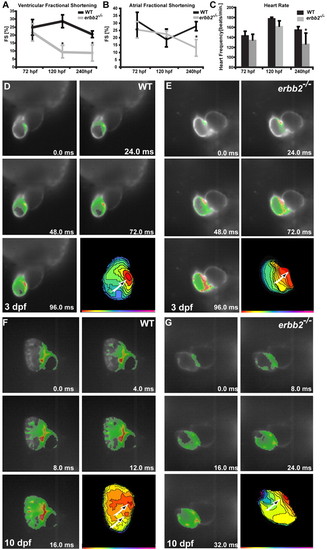
erbb2 mutants display progressive cardiac dysfunction. (A,B) The ventricular fractional shortening of 3 dpf erbb2 mutants is not significantly different from that of wild type (A). A ventricular fractional shortening defect manifests itself at -5 dpf (A) and an atrial fractional shortening defect appears at -10 dpf (B). Data are presented as mean ± s.d. *P<0.05. (C) erbb2 mutant hearts begin to develop bradycardia at <5 dpf. (D-G) Optical mapping of wild-type (D,F) and erbb2 mutant (E,G) hearts at 3 (D,E) and 10 (F,G) dpf. erbb2 mutants at 10 dpf (G) fail to develop the mature conduction system observed in their wild-type siblings (F). |