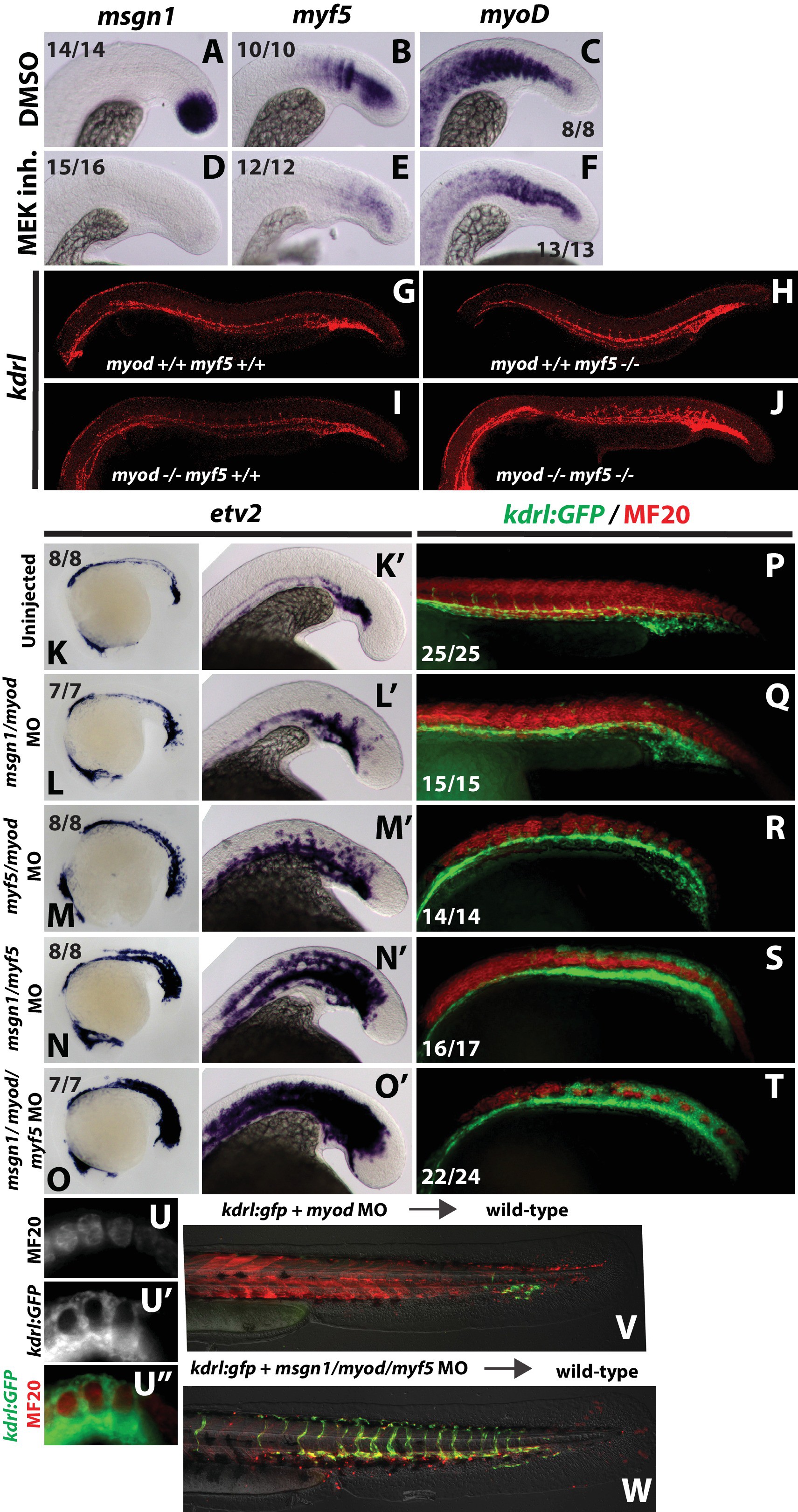
Fig. 7
FGF signaling maintains paraxial mesoderm fate and inhibits endothelial fate through positive regulation of bHLH transcription factors.
Wild-type embryos were treated with the MEK inhibitor or DMSO at the 12-somite stage and fixed five hours later. Expression of msgn1 (D) and myf5 (E) were significantly downregulated compared to controls (A, B), whereas myod (F) exhibited only a minor reduction in expression compared to controls (C). Expression of kdrl (red) is expanded into somitic territories in myod;myf5 double mutants compared to controls (G). n = 44 controls (pooled +/+;+/+, +/+;+/-; +/-;+/+, and +/-;±genotyped embryos). 0/44 controls have expanded kdrl. n = 4 mutants (-/-;-/- genotyped embryos). 4/4 show expanded kdrl (representative embryos shown). MO-mediated loss of function of msgn1/myod (L, L’) or myf5/myod (M, M’) results in a moderate expansion of etv2 expression at the 22 somite stage, whereas loss of msgn1/myf5 causes a broad expansion of etv2 (N, N’). Loss of function of all three genes further enhances etv2 expansion (O, O’). MF20 (muscle, red) antibody staining in 30 hpf kdrl:GFP embryos demonstrates the gain of differentiated vasculature at the expense of differentiated muscle (P–T). U-U’’ are high-magnification views of MF20 staining and kdrl:GFP expression in a msgn1/myf5 loss of function embryo. Transplanted kdrl:GFP cells lacking myod/myf5/msgn1 fail to join host somites and instead contribute predominantly to endothelium (W), whereas cells lacking myod behave normally, with most transplanted cells joining the somites and forming muscle (V).