- Title
-
Intracellular Localization in Zebrafish Muscle and Conserved Sequence Features Suggest Roles for Gelatinase A Moonlighting in Sarcomere Maintenance
- Authors
- Fallata, A.M., Wyatt, R.A., Levesque, J.M., Dufour, A., Overall, C.M., Crawford, B.D.
- Source
- Full text @ Biomedicines
|
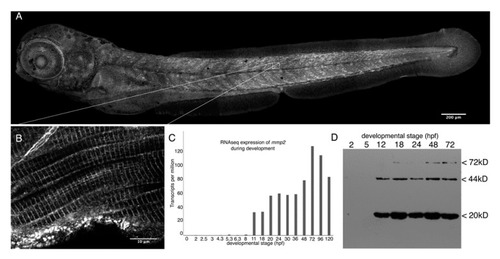
Mmp2 is expressed ubiquitously from early development and accumulates in a striated pattern within the skeletal muscle. ( EXPRESSION / LABELING:
|
|
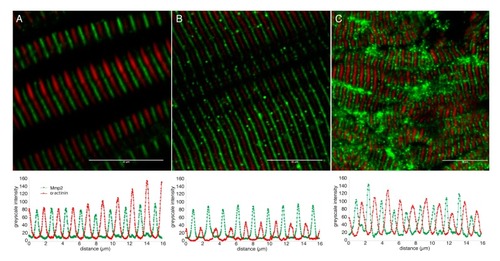
Mmp2 is localized between Z-discs in sarcomeres of embryonic and adult muscle. Confocal micrographs of skeletal muscle from 72 hpf embryos ( EXPRESSION / LABELING:
|
|
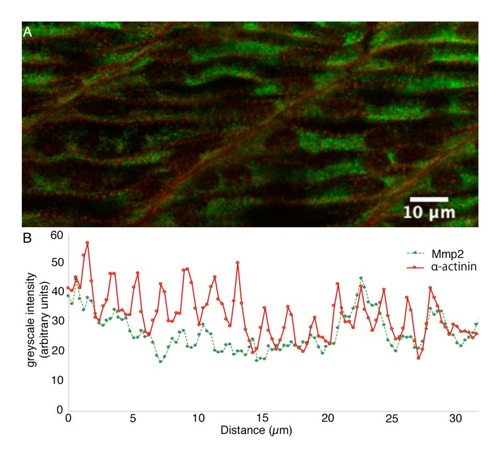
Sarcomeric Mmp2 begins to accumulate subsequent to the assembly of Z-disks. ( |
|
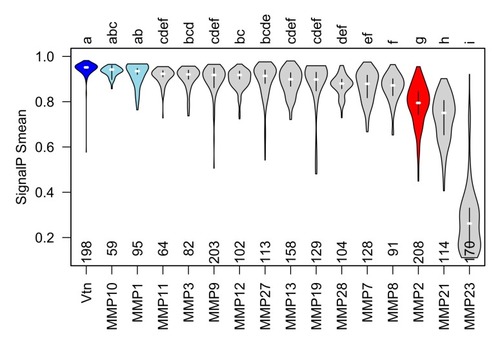
The secretory signal peptide of gelatinase A orthologues is consistently and significantly less likely to be recognized than that of most other type-I secreted proteins. Violin plots of mean ‘S’ scores of the N-terminal secretory signals from orthologues of vitronectin (Vtn) and all secreted MMPs. MMPs with mean S score statistically indistinguishable from vitronectin are shown in blue. Mean S scores for orthologues of gelatinase A (red) are significantly lower than those of vitronectin and other secreted MMPs apart from MMP21 and MMP23, which undergoes type II secretion and therefore, does not have an N-terminal secretory signal. Statistically, indistinguishable groups are indicated with letters at the top of the plot, and the number of orthologues of each protein analyzed is indicated along the |
|
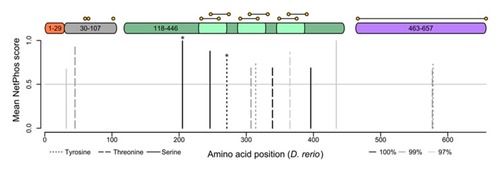
Gelatinase A orthologues have highly conserved phosphorylation sites. Putative serine (solid lines), threonine (dashed lines), and tyrosine (dotted lines) phosphorylation sites conserved in 100% ( |