- Title
-
FGF signaling enforces cardiac chamber identity in the developing ventricle
- Authors
- Pradhan, A., Zeng, X.I., Sidhwani, P., Marques, S.R., George, V., Targoff, K.L., Chi, N.C., Yelon, D.
- Source
- Full text @ Development
|
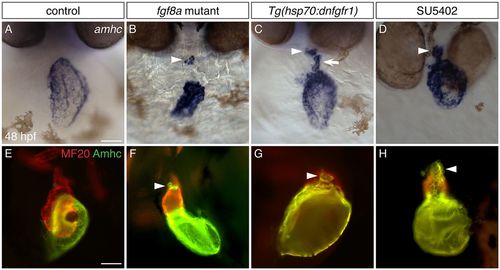
Inhibition of FGF signaling results in ectopic amhc expression in the ventricle. (A-D) In situ hybridization showing amhc expression in frontal views at 48 hpf. (A) Wild-type embryos express amhc in the atrium. Similar phenotypes are seen in all controls examined: wild-type siblings of fgf8a mutants, nontransgenic siblings following heat shock, and DMSO-treated siblings. (B) In fgf8a mutants, amhc expression is found not only in the atrium but also in a small cluster of ventricular cells (arrowhead; n=32/46 mutants). (C) More ectopic amhc-expressing cells are induced in Tg(hsp70:dnfgfr1) embryos following heat shock at 18 hpf (n=15/15); these cells are typically seen along the inner curvature (arrow) and at the arterial pole (arrowhead) of the ventricle. (D) Similarly, ectopic amhc-expressing cells can be induced by exposure to SU5402 starting at 18 hpf (arrowhead; n=15/15). The relatively mild phenotype of fgf8a mutants (B), compared with the more striking presence of ectopic cells in Tg(hsp70:dnfgfr1)-expressing and SU5402-treated embryos (C,D), suggests that additional FGF ligands might work together with fgf8a to prevent inappropriate amhc expression. (E-H) Immunofluorescence with MF20 (marks the myocardium; red) and S46 (recognizes Amhc; green) showing cardiac morphology and Amhc localization in lateral views at 48 hpf. In contrast to wild type (E), ectopic Amhc (arrowheads) is seen in fgf8a mutants (F), Tg(hsp70:dnfgfr1) embryos after heat shock at 18 hpf (G) and embryos treated with SU5402 from 18-30 hpf (H) (n>10 each). Scale bars: 50 μm. |
|
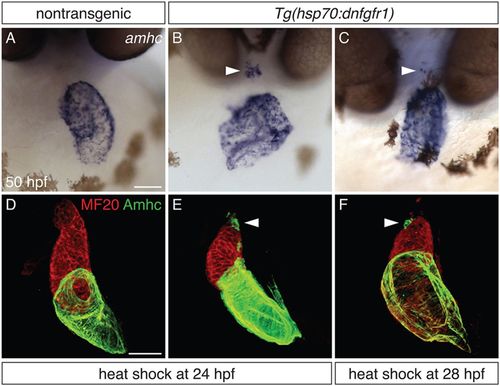
Defining a time interval during which FGF signaling prevents ectopic amhc expression. (A-C) In situ hybridization depicts amhc expression in frontal views of nontransgenic (A) and Tg(hsp70:dnfgfr1) (B,C) embryos at 50 hpf. Heat shock at 24 hpf (A,B) induces ectopic amhc-expressing cells in nearly all Tg(hsp70:dnfgfr1) embryos (B; n=22/24), whereas ectopic cells are induced less frequently with heat shock at 28 hpf (C; n=6/36). Heat shock at 18 hpf gives rise to approximately 20-30 ectopic amhc-expressing cells (Fig. 1C), heat shock at 24 hpf induces approximately five to ten ectopic amhc-expressing cells (B, arrowhead), and approximately two to three ectopic cells are produced with heat shock at 28 hpf (C, arrowhead). (D-F) Three-dimensional reconstructions of immunofluorescence, as in Fig. 1E-H, of nontransgenic (D) and Tg(hsp70:dnfgfr1) (E,F) embryos. Whereas Tg(hsp70:dnfgfr1) embryos heat shocked at 18 hpf (Fig. 1G) typically display ectopic amhc-expressing cells in the ventricular inner curvature and at the arterial pole, Tg(hsp70:dnfgfr1) embryos heat shocked at 24 (E) and 28 hpf (F) primarily exhibit ectopic cells at the arterial pole (arrowheads) [n=15 for nontransgenic, n=10 per condition for Tg(hsp70:dnfgfr1)]. Scale bars: 50 μm. |
|
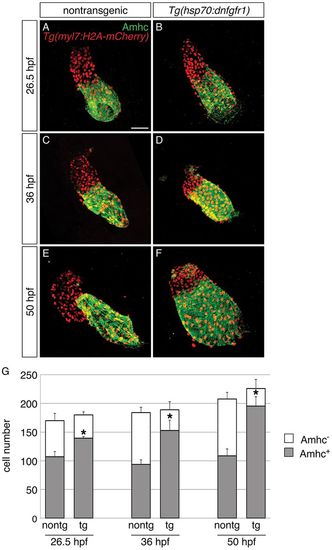
Progressive accumulation of Amhc+ cells and decrease in Amhc− cells in embryos with reduced FGF signaling. (A-F) Immunofluorescence for Amhc (green) and mCherry (red) allows counting of Amhc+ and Amhc− cardiomyocytes in nontransgenic (A,C,E) and Tg(hsp70:dnfgfr1) embryos (B,D,F) carrying Tg(myl7:H2A-mCherry). Images depict hearts flattened to facilitate visualization of individual myocardial nuclei at 26.5 (A,B), 36 (C,D) and 50 hpf (E,F). Heat-induced expression of dnfgfr1 at 18 hpf results in a gradual shift in chamber proportions that leads to an enlarged atrium and a reduced ventricle. In addition, there is a visible increase in the presence of Amhc+ cells within the ventricle over time. (G) Numbers of Amhc+ (gray) and Amhc− (white) cardiomyocytes, as well as the total number of cardiomyocytes (mean+s.d.); asterisks indicate statistically significant differences from nontransgenic (P<0.0001). Cell counts reveal a gradual increase in numbers of Amhc+ cells and a corresponding decrease in numbers of Amhc− cells in hearts of Tg(hsp70:dnfgfr1) embryos [26.5 hpf: n=8 for nontransgenic, n=6 for Tg(hsp70:dnfgfr1); 36 hpf: n=11 for nontransgenic, n=9 for Tg(hsp70:dnfgfr1); 50 hpf: n=8 for nontransgenic, n=12 for Tg(hsp70:dnfgfr1)]. See Table S1 for the number of ectopic Amhc+ cells in a subset of these embryos. Scale bar: 50 μm. |
|
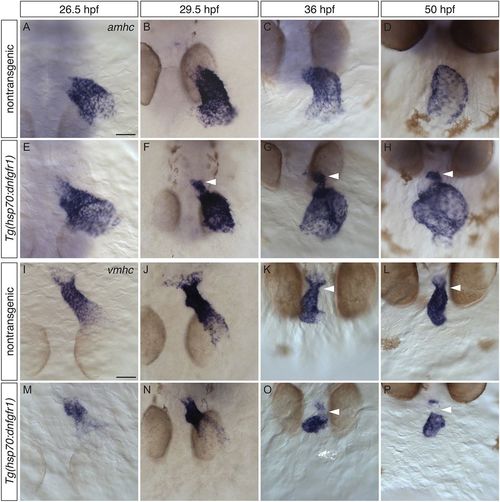
Increased expression of amhc and reduced expression of vmhc in the ventricle of embryos with reduced FGF signaling. (A-P) In situ hybridization showing amhc (A-H) and vmhc (I-P) expression in dorsal (A,B,E,F,I,J,M,N) and frontal (C,D,G,H,K,L,O,P) views, following heat shock at 18 hpf. (A-H) In contrast to nontransgenic embryos (A-D), Tg(hsp70:dnfgfr1) embryos (E-H) fail to restrict amhc to the atrium (n>10 each). (E) At 26.5 hpf, amhc-expressing cells can be seen infringing the boundary between the atrium and the ventricle. (F) By 29.5 hpf, amhc levels in ectopic cells are upregulated (arrowhead). (G,H) A prominent population of ectopic cells is found at the inner curvature and arterial pole of the ventricle (arrowheads). (I-P) Whereas vmhc is robustly expressed in the ventricle of nontransgenic embryos (I-L), this expression pattern is disrupted in Tg(hsp70:dnfgfr1) embryos (M-P) (n>10 each). (M,N) At early stages, Tg(hsp70:dnfgfr1) embryos display lower levels of vmhc expression in regions adjacent to the atrium. (O,P) As development proceeds, vmhc levels at the arterial pole become reduced or absent (arrowheads); moreover, the vmhc expression pattern appears to be reciprocal to that of ectopic amhc in the ventricle. Scale bars: 50 μm. |
|
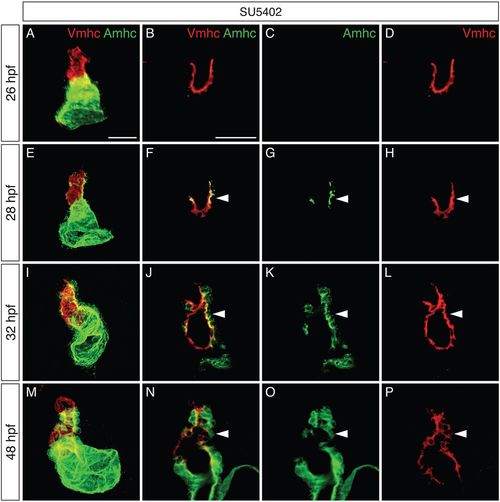
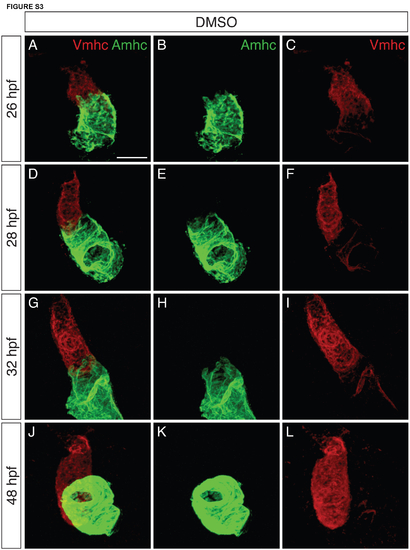
Increased colocalization of Vmhc and Amhc in the ventricle when FGF signaling is inhibited. (A-P) Immunofluorescence showing Vmhc (red) and Amhc (green) localization in embryos treated with SU5402 from 18 hpf. Lateral views are three-dimensional reconstructions (A,E,I,M) or magnified single optical sections, showing both channels (B,F,J,N), green only (C,G,K,O) or red only (D,H,L,P). See Fig. S3 for DMSO-treated control embryos. (A-D) At 26 hpf, the SU5402-treated ventricle contains only Vmhc (n=10). (E-H) By 28 hpf, Amhc is evident within some Vmhc+ cells, particularly along the inner curvature (arrowheads; n=9). We presume that these cells are early-differentiating cardiomyocytes, as late-differentiating cardiomyocytes normally contribute to the heart after 30 hpf (Lazic and Scott, 2011). (I-L) By 32 hpf, increasing numbers of Amhc+Vmhc+ cells are visible in the ventricle (arrowheads; n=11). (M-P) By 48 hpf, higher levels of Amhc accumulate in ectopic cells (arrowheads), in concert with diminishing levels of Vmhc (n=9). Scale bars: 50 μm. |
|
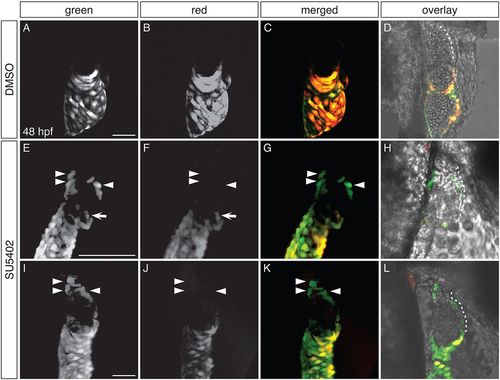
Ectopic amhc-expressing cells are not derived from the atrium or AVC. (A-L) Dendra fluorescence in live DMSO-treated (A-D) and SU5402-treated (E-L) Tg(amhc:dendra) embryos at 48 hpf, following photoconversion at 26 hpf; two representative SU5402-treated embryos are shown. Images are three-dimensional reconstructions (A-C,E-G,I-K) or single optical sections (D,H,L); lateral views, anterior to the right. (D,H,L) Overlay illustrates location of Dendra+ cells within the heart; white dashes outline the ventricle. See Fig. S4 for additional information regarding experimental design. (A-D) In DMSO-treated controls, Dendra is found only in the atrium and AVC. Both green and red forms of Dendra are visible, as illustrated in single channel and merged views (n=10). (E-L) SU5402-treated embryos exhibit Dendra+ cells in the ventricle, in distinct clusters at the arterial pole and in the inner curvature (arrowheads). Cells at the arterial pole and inner curvature fluoresce green, but not red (arrowheads; n=15). Dendra+ cells are also found in and near the AVC (arrows), as seen in controls (A-D), reflecting the wild-type expression of amhc in this area (Foglia et al., 2016). Scale bars: 50 μm (A,I); 100 μm (E). |
|
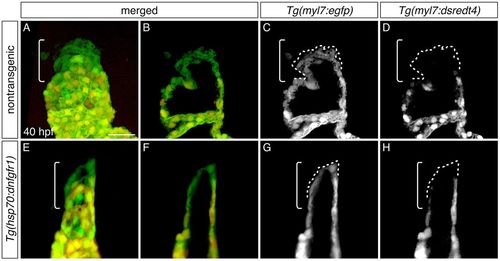
Late-differentiating cardiomyocytes contribute to the heart after inhibition of FGF signaling. (A-H) Three-dimensional reconstructions (A,E) and single optical sections (B-D,F-H) of live embryos carrying Tg(myl7:egfp) and Tg(myl7:dsredt4); lateral views at 40 hpf, after heat shock at 18 hpf. Owing to the differential protein-folding kinetics of eGFP and dsRed (Lepilina et al., 2006), reporter transgene expression distinguishes early-differentiating (eGFP+dsRed+) and late-differentiating cardiomyocytes (eGFP+dsRed−) (de Pater et al., 2009). As in nontransgenic embryos (A-D, n=6), late-differentiating cardiomyocytes (eGFP+dsRed−, dashed outlines) contribute to the arterial pole (brackets) in Tg(hsp70:dnfgfr1) embryos (E-H; n=8). Scale bar: 30 μm. |
|
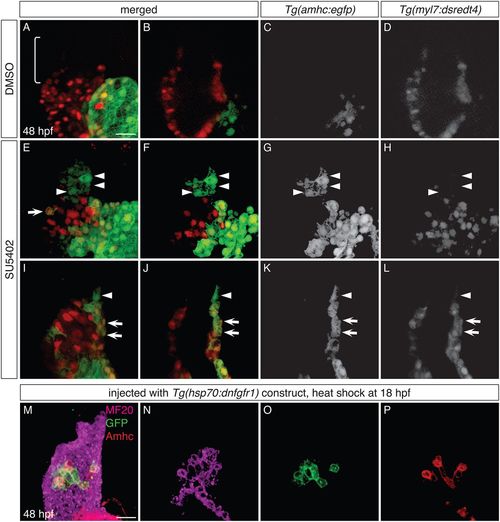
FGF signaling influences the identity of both early-differentiating and late-differentiating ventricular cardiomyocytes. (A-L) Three-dimensional reconstructions (A,E,I) and single optical sections (B-D,F-H,J-L) of live Tg(amhc:egfp);Tg(myl7:dsredt4) embryos; lateral views at 48 hpf, after exposure to DMSO (A-D) or SU5402 (E-L) from 18 hpf. (A-D) As expected, control embryos display eGFP in the atrium and not in the ventricle, and the arterial pole (bracket) is composed of late-differentiating cardiomyocytes (DsRed−) (n=5). (E-L) In SU5402-treated embryos, eGFP is routinely found in the ventricle (n=10). Two representative embryos (E-H and I-L) illustrate detection of eGFP both in early-differentiating cardiomyocytes (arrows, eGFP+DsRed+; n=10/10) and in late-differentiating cardiomyocytes (arrowheads, eGFP+DsRed−; n=7/10). (M-P) Immunofluorescence for MF20 (magenta), GFP (green) and Amhc (red) at 48 hpf indicates mosaic expression of dnfgfr1-egfp in the ventricle of an embryo that was injected with the Tg(hsp70:dnfgfr1) construct and then heat shocked at 18 hpf; three-dimensional reconstruction (M) and single optical sections (N-P). The dnfgfr1-egfp-expressing cells in the proximal ventricle exhibit variegated levels of Amhc (P; n=3). Scale bars: 30 μm. |
|
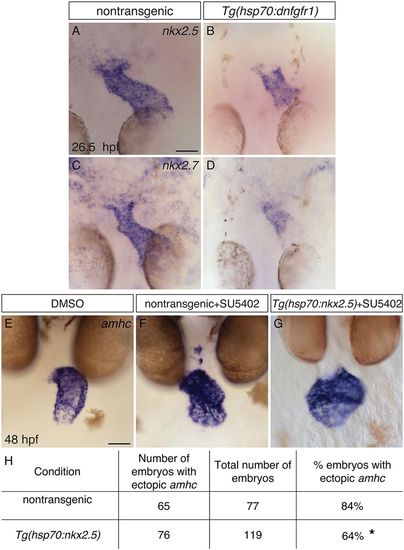
FGF signaling functions upstream of Nkx genes to repress amhc expression in the ventricle. (A-D) In situ hybridization showing nkx2.5 (A,B) and nkx2.7 (C,D) expression in dorsal views at 26.5 hpf, following heat shock at 18 hpf. Nontransgenic embryos (A,C) exhibit robust expression of nkx2.5 (n=24) and nkx2.7 (n=12), and Tg(hsp70:dnfgfr1) embryos (B,D) exhibit reduced expression of nkx2.5 (n=24) and nkx2.7 (n=12). This reflects a reduced amount of ventricular tissue in Tg(hsp70:dnfgfr1) embryos (compare with Fig. 4M), as well as lower expression levels within this tissue. (E-G) In situ hybridization showing amhc expression at 48 hpf in nontransgenic (F) and Tg(hsp70:nkx2.5) (E,G) embryos treated with DMSO (E) or SU5402 (F,G); frontal views. In some cases (G), overexpression of nkx2.5 represses ectopic amhc expression in Tg(hsp70:nkx2.5) embryos that were treated with SU5402 at 18 hpf and subsequently heat shocked at 24 hpf. (H) Table reports the results of experiments gauging whether amhc expression in the ventricle of SU5402-treated embryos can be repressed by overexpression of nkx2.5. Asterisk indicates a statistically significant difference in the frequency of detecting ectopic amhc expression, compared to nontransgenic siblings (P=0.0019, Fisher’s exact test), representing partial rescue of the SU5402-treated phenotype. The degree of rescue was not enhanced when we used embryos carrying two copies of Tg(hsp70:nkx2.5) or when we administered two rounds of heat shock (data not shown). Scale bars: 50 μm. |
|
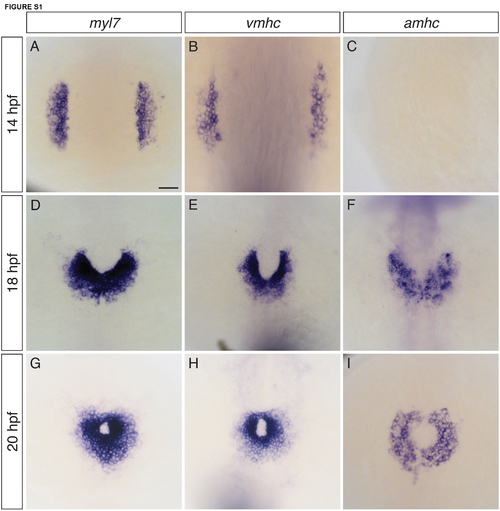
Expression of both vmhc and amhc is initiated by 18 hpf. (A-I) In situ hybridization depicts expression of myl7 (A,D,G), vmhc (B,E,H), and amhc (C,F,I) in dorsal views of wild-type embryos from 14-20 hpf (n=10 for each condition). (A,D,G) myl7 is expressed in all differentiated cardiomyocytes (Yelon et al., 1999). At 14 hpf, a subset of myl7-expressing cells express vmhc (B) and amhc is not yet expressed (C) (Yelon et al., 1999). By 18 hpf, cardiac fusion is underway, and vmhc (E) and amhc (F) are expressed in relatively medial and lateral populations of cardiomyocytes, corresponding to the locations of ventricular and atrial precursors (Schoenebeck et al., 2007). By 20 hpf, cardiomyocytes fuse at the midline to form a cardiac cone (Yelon et al., 1999), and vmhc (H) and amhc (I) expression are expressed in relatively central and peripheral populations of ventricular and atrial cardiomyocytes (Berdougo et al., 2003). We do not consider either vmhc or amhc to be expressed in a strictly chamberspecific fashion: for example, low levels of vmhc are present in atrial cells at early stages (Berdougo et al., 2003; Yelon et al., 1999), and a subset of cells at the apex of the ventricle have been shown to express amhc (Foglia et al., 2016). Even so, the differences between the vmhc and amhc expression patterns point to the initiation of distinct differentiation programs in ventricular and atrial precursor populations, beginning as early as 18 hpf. Scale bar: 50 μm. |
|
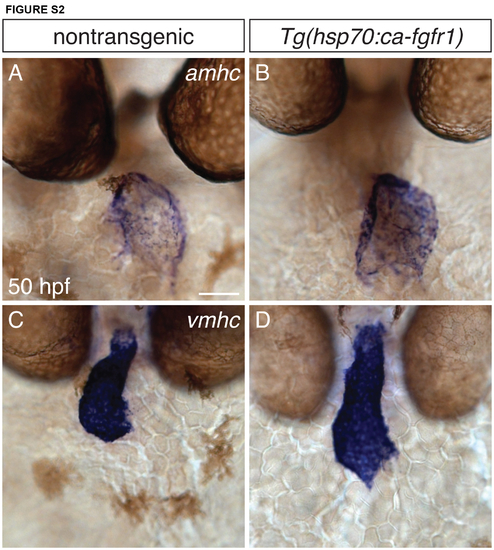
Distribution of amhc and vmhc expression is unaffected by increased FGF signaling. (A-D) In situ hybridization depicts frontal views of amhc (A,B) and vmhc (C,D) expression at 50 hpf, following heat shock at 18 hpf. Nontransgenic embryos (A,C) show stereotypical expression patterns of amhc and vmhc (n=10). Increased FGF signaling in Tg(hsp70l:ca-fgfr1) embryos (B,D) results in slight morphological changes, consistent with prior observations (Marques et al., 2008). However, increased FGF signaling does not repress amhc in the atrium or result in ectopic vmhc expression (n=10). Scale bar: 50 μm. |
|
Vmhc antibody marks expected locations of vmhc expression within the heart. (A-L) Immunofluorescence depicts Vmhc (red) and Amhc (green) localization in wildtype embryos treated with DMSO at 18 hpf. Images are three-dimensional reconstructions of lateral views at 26-48 hpf, showing both channels (A,D,G,J), green only (B,E,H,K), or red only (C,F,I,L). The Vmhc antibody marks the expected locations of vmhc expression within the heart (n=10 for 26.5 hpf; n=8 for 28 hpf; n=8 for 32 hpf; n=8 for 48 hpf). Vmhc immunostaining recapitulates expression of the reporter transgene Tg(vmhc:mCherry-NTR) (Zhang et al., 2013), as well as expression of vmhc itself (Fig. 4I-L). Vmhc is robustly detected in the ventricle, with low levels in the atrium that diminish over time, as has been previously shown for expression of vmhc (Fig. 4I-L) (Yelon et al., 1999). Scale bar: 50 μm. |
|
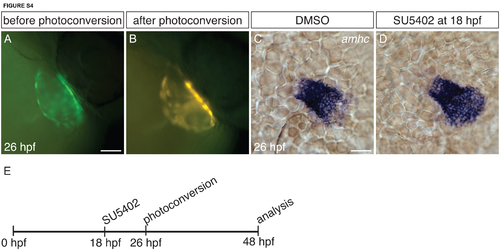
Tg(amhc:dendra) facilitates tracking of amhc-expressing cells. (A-B) Tg(amhc:dendra) hearts before (A) and after (B) photoconversion at 26 hpf, lateral views. Tg(amhc:dendra) expression recapitulates the expression of the reporter transgene Tg(amhc:eGFP) (Zhang et al., 2013) and the expression of amhc itself (Fig. 4A-D): it is expressed throughout the atrium. Upon photoconversion, Dendra fluorescence is converted from green to red. Photoconversion at 26 hpf permits labeling of amhc-expressing cells throughout the atrium, and these photoconverted cells can then be detected at later stages (Fig. 6). (C-D) In situ hybridization depicts amhc expression in dorsal views of DMSO-treated and SU5402-treated embryos at 26 hpf. At 26 hpf, amhc expression in SU5402-treated embryos (D) is limited to the atrium; ectopic amhc-expressing cells are not yet visible at this stage.(E) Timeline of experimental design. Since amhc expression is not found in the ventricle of SU5402-treated embryos at 26 hpf, photoconversion at this stage permits examination of whether the ectopic amhc-expressing cells that appear later are derived from within the atrium or the AVC. Scale bars: 50 μm. |
|
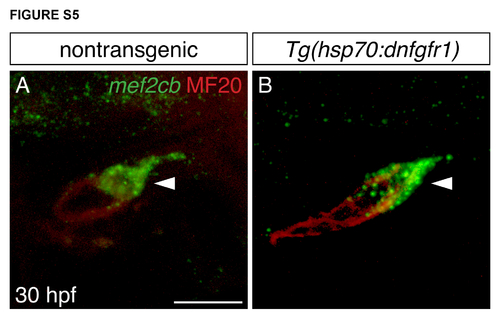
SHF progenitor cells are present after inhibition of FGF signaling. (A-B) MF20 immunofluorescence and mef2cb in situ hybridization (green) in lateral views at 30 hpf, after heat shock at 18 hpf. mef2cb (green, arrowheads) is expressed in SHF progenitor cells residing next to the differentiated myocardium (red) in both nontransgenic (A, n=8) and Tg(hsp70:dnfgfr1) (B, n=8) embryos. Scale bar: 50 μm. |
|
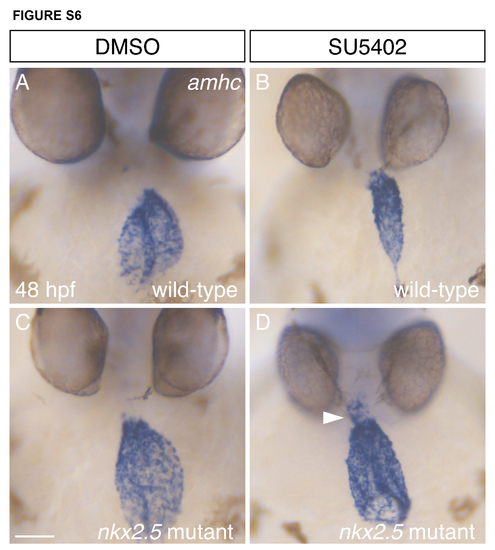
Subtle enhancement of the nkx2.5 mutant phenotype by treatment with a low dose of SU5402. (A-D) In situ hybridization depicts frontal views of amhc expression in wild-type (A,B) and nkx2.5 homozygous mutant (C,D) embryos at 48 hpf. Embryos were treated with DMSO (A,C) or 4 μm SU5402 (B,D) from 18 hpf until 30 hpf. In wild-type embryos, this relatively low dose of SU5402 altered cardiac morphology but did not induce ectopic amhc-expressing cells (B). However, in a few nkx2.5 mutants, this dose of SU5402 resulted in the increased presence of ectopic amhc in the ventricle (arrowhead, D); this example is representative of 9 similar embryos observed in a total of 135 progeny from 4 independent intercrosses of nkx2.5 heterozygotes. This subtle enhancement of the nkx2.5 mutant phenotype (C) suggests that FGF signaling may act in the same pathway as the nkx genes to repress the inappropriate expression of amhc. Scale bar: 50 μm. |