- Title
-
The Ciliopathy Protein CC2D2A Associates with NINL and Functions in RAB8-MICAL3-Regulated Vesicle Trafficking
- Authors
- Bachmann-Gagescu, R., Dona, M., Hetterschijt, L., Tonnaer, E., Peters, T., de Vrieze, E., Mans, D.A., van Beersum, S.E., Phelps, I.G., Arts, H.H., Keunen, J.E., Ueffing, M., Roepman, R., Boldt, K., Doherty, D., Moens, C.B., Neuhauss, S.C., Kremer, H., van Wijk, E.
- Source
- Full text @ PLoS Genet.
|
CC2D2A and NINL co-localize at the ciliary base in hTERT-RPE1 cells and in zebrafish retina. (a, a' and inset) When expressed alone, eCFP-tagged CC2D2A (green signal) localizes to the ciliary base (basal body, accessory centriole). The cilium is marked by anti-polyglutamylated tubulin (red signal, a' and inset). eCFP-tagged CC2D2A (green signal; b) also (partly) localizes to the ciliary transition zone, which was visualized using anti-RPGRIP1L as a marker (red signal; b). (c, c' and inset) mRFP-tagged NINL isoform B was localized at the ciliary base (cilium in green, c' and inset). (d and inset) mRFP-tagged NINL isoform B (red signal) localizes adjacent to the ciliary transition zone (anti-RPGRIP1L; green signal). (e-e” and inset) Co-expression of mRFP-tagged NINL isoform B (red signal) and eCFP-tagged CC2D2A showed co-localization of both proteins around the ciliary base (yellow signal). (f) In wild-type larval zebrafish retina (4 dpf), Cc2d2a marked by anti-Cc2d2a antibodies (red signal) is localized apically to the photoreceptor basal body (marked by anti-centrin antibodies, green signal). (g) Ninl, stained with anti-Ninl antibodies, (red signal) is localized at the zebrafish photoreceptor ciliary base, partially overlapping with and apical to the green centrin signal. (h) Cc2d2a localization is unaffected by ninl knockdown and (i) Ninl localization is normal in cc2d2a-/- larvae. (j) Schematic representation of the localization of Ninl and Cc2d2a in zebrafish photoreceptor cells. (f-i) are immunostainings on cryosections from 4 dpf larvae. Nuclei were stained with DAPI (blue signal) in all panels. Scale bars are 10 μm in a-e, and 4 μm in f-i. EXPRESSION / LABELING:
|
|
Zebrafish ninl knockdown causes loss of axonemes and outer segments, opsin mislocalization and vesicle/vacuole accumulation. (a-b) Paraffin sections stained with Hematoxylin/Eosin of control (a) and ninl knockdown larvae (b) demonstrating shortened outer segments and grossly preserved retinal lamination in the morphants. (c-d') Bodipy-stained cryosections highlight the shortened (brackets c'-d') and dysmorphic outer segments of ninl knockdown larvae (d and d') compared to the long cone- or rod-shaped outer segments of controls (c and c'). (e-f) Axonemes and connecting cilia marked with anti-acetylated alpha-tubulin and anti-Ift88 antibodies are severely shortened and reduced in numbers in ninl knockdown larvae (arrowhead in f). (g-h') Immunofluorescence with anti-opsin antibody 4D2 demonstrates mislocalization of opsins within the cell body in ninl knockdown larvae (arrow in h') compared to controls (g) where opsins are restricted to the outer segment. (i) Quantification of the intracellular opsin accumulation in ninl morphant photoreceptors compared to control: each single datapoint in the scatter graph displays the averaged mean grey value from one larva. The mean value and the Standard Error of the Mean (SEM) are displayed as bars. The difference is statistically significant (*** = p<0.0001, Student's t-test). (j-l') Transmission electron microscopy of control (j) and ninl knockdown larvae (k-l') demonstrates absent or shortened and dysmorphic outer segments (OS) and accumulation of large vacuoles (v, arrow in l') and smaller vesicular structures (bracket in k” and white arrowheads in l') in morphants. Black arrowheads point to the connecting cilium in k and k”. k' and k” are the boxed areas in k and l' is the boxed area in l. (m) Quantification of the % of photoreceptors displaying these phenotypes. Absolute numbers of photoreceptors are also indicated. Error bars indicate 95% Confidence Intervals. The differences between morphant (red bars) and controls (blue bars) are statistically significant (*** = p<0.0001, Fisher's exact test). Larvae in all panels are 4 dpf old. Scale bars are 30 μm in a-b, 15 μm in c-d and g-h, 3 μm in c'-d' and g'-h', 4 μm in e-f, 0.5 μm in j-k and l and 150nm in k'-k” and l'. OS outer segment, CC connecting cilium, m mitochondria, n nucleus, v vacuole. |
|
Genetic interaction between ninl and cc2d2a. (a-d) Partial ninl knockdown enhances the cystic kidney phenotype of cc2d2a mutants. (a-c) Glomerulus and proximal pronephric tubules highlighted in the transgenic line Tg(wt1b-EGFP). (a) Injection of a low dose of ninl atgMO (0.75 ng/nl) causes no cysts in wild-type larvae. (b) cc2d2a-/- larvae display small dilatations of the proximal tubules (arrow) in ~40% of cases. (c) Injection of this low dose of ninl atgMO in the cc2d2a-/- background leads to large dilatations of the proximal tubules and glomerular space (arrow) in 89% of mutants. g glomerulus, p pancreas. (d) Quantification of the glomerular + proximal tubular area displayed as a scatter plot, demonstrating a significant increase in proximal pronephric area in cc2d2a-/- larvae injected with low-dose ninl atgMO. The bars represent the mean and standard error of the mean (SEM) for each treatment group and each datapoint is an individual fish. (e-g') Immunohistochemistry with anti-opsin antibody (4D2, green) on retinal cryosections of 4dpf cc2d2a-/- uninjected larvae (f-f”) and cc2d2a-/- larvae injected with subphenotypic doses of ninl MO (g'g”'), that cause no mislocalization in wild-type fish (e-e'), demonstrates that partial ninl knockdown increases the mislocalization of opsins (e'-g'). (h) Quantification of the mean intracellular fluorescence displayed as a scatter plot shows significant increase in intracellular fluorescence in cc2d2a-/- larvae injected with low dose of ninl atgMO. The bars represent the mean and standard error of the mean (SEM) for each treatment group and each datapoint represents the mean intracellular fluorescence from 10 photoreceptors in one individual fish. Cell membrane and outer segments are stained with bodipy (red in e-g). Nuclei are counterstained with DAPI. Scale bars are 100 μm in (a-c) and 4 μm in (e-g'). (i) Pedigree of a consanguineous family with one affected boy (UW48-3) and 4 unaffected siblings. UW48-3 carried a homozygous missense CC2D2A mutation as well as a frameshift mutation in NINL leading to premature truncation. (j) Pedigree of a family where the affected individual (UW36-3) carries the same homozygous CC2D2A mutation as in (i) but no additional rare deleterious variants. (k) Pedigree of a family where the affected individual (UW07-3) carries compound heterozygous C5ORF42 frameshift mutations and a nonsense mutation in NINL. (l) Pedigree of a family where the affected individual (UW57-3) carries compound heterozygous TMEM67 mutations and a missense NINL mutation. The phenotype of the affected individuals is detailed in italic on each pedigree under the corresponding mutations. MTS Molar Tooth Sign, DD Developmental Delay, ESRF End-Stage Renal Failure. |
|
Ninl is required for correct Rab8A localization. (a-a') Expression of a rhodopsin-promoter driven cherry-tagged Rab8a in wild-type photoreceptors is mostly concentrated in one or several puncta (arrows a-a') whereas it is diffuse in the majority of ninl morphant photoreceptors (b-b'). (c) Proportion of Rab8a-cherry expressing photoreceptors with punctate expression versus diffuse expression (bars represent 95% confidence interval; ** P<0.001, Fisher's exact test). (d-d') Endogenous Rab8a localization as seen by immunohistochemistry using an anti-Rab8a antibody (green) displays similar puncta (arrowheads) in wild-type photoreceptors, while the number of puncta is decreased in ninl morphant photoreceptors (e-e'). (f) Quantification of the number of Rab8a puncta displayed in the form of a scatter plot indicating that significantly fewer endogenous Rab8 puncta per μm2 are present in ninl morphants compared to uninjected controls (*P = 0.01, unpaired Student's t-test; bars represent standard error of the mean). Scoring was performed blinded as to injection status for (c) and (f). Outer segments are counterstained with bodipy in (d-e). Nuclei are counterstained with DAPI. All images are cryosections of 4 dpf larvae. Scale bars are 4 μm in all panels. |
|
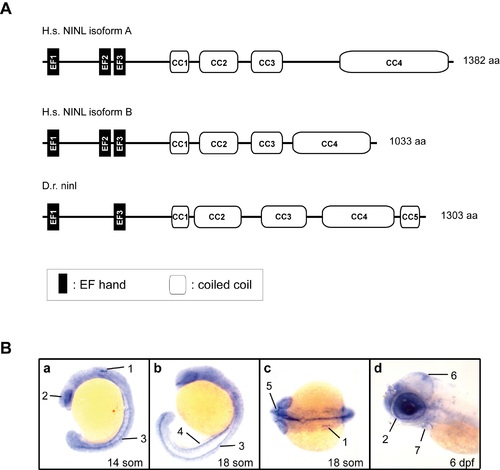
Cloning and characterization of zebrafish ninl. (A) Schematic representation of the protein structure of human NINLisoA and NINLisoB (H.s. NINLisoA and H.s. NINLisoB) and zebrafish ninl (D.r. ninl) as predicted by using the Pfam homepage (http://pfam.xfam.org). (B) Ninl expression during zebrafish development by whole mount RNA in situ hybridization. Specific expression was found in the following structures as indicated by numbers and arrows: (a) 14 somite stage: otic placode (1); developing eye (2); neural tube (spinal cord) (3); (b) 18 somite stage: neural tube (3); pronephros (4); (c) 18 somite stage: inner ear (1); optic nerve (5). (d) At 6 dpf, expression was observed in the tectum (6), the heart (7) and in the eye (2), predominantly in the photoreceptor cell layer. H.s.: homo sapiens; D.r.: danio rerio; CC: coiled-coil; IF: intermediate filament domain; som: somites; dpf: days post-fertilization. EXPRESSION / LABELING:
|
|
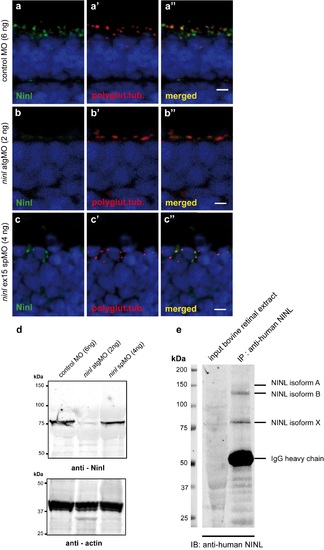
Specificity of the anti-Ninl antibody. (a) Indirect immunohistochemical staining using anti-Ninl antibody on 4 dpf retinal cryosections of control MO-injected larvae (green signal) shows punctate staining, partially overlapping with the ciliary marker anti-polyglutamylated tubulin (a', a”, red signal). (b) Indirect immunohistochemical staining using anti-Ninl antibody on 4 dpf retinal cryosections of ninl atgMO-injected larvae (b, green signal) along with the ciliary marker anti-polyglutamylated tubulin (b', b”, red signal). Specific Ninl-immunofluorescence is largely abolished in ninl morphants, whereas the polyglutamylated tubulin signal is still detected. (c) Indirect immunohistochemical staining of anti-Ninl on 4 dpf retinal cryosections of ninl ex15 spMO-injected larvae (green signal) along with the ciliary marker anti-polyglutamylated tubulin (c', c”, red signal). Specific Ninl-immunofluorescence was still detected but at a diminished level in ninl morphants whereas the polyglutamylated tubulin signal was unaltered. Nuclei are stained with DAPI (blue signal). Scale bars: 4 μm. (d) Western blot analysis using protein extracts obtained from 100 zebrafish larvae injected with either control MO (6ng), ninl atgMO (2ng) or ninl ex15 spMO (4ng). A specific product was detected with a molecular weight of ~80kDa in control MO-injected larvae (left panel). This band was almost completely abolished in the ninl atgMO-treated larvae, but was still detected in ninl spMO-injected larvae although with a slightly diminished intensity. Anti-actin antibodies were used as a loading control (right panel). (e) Immunoprecipitation from bovine retinal extracts with anti-human NINL antibody detects 3 bands, the strongest being of the same size as the band found on Western blot of zebrafish lysates (~80 kDa). |
|
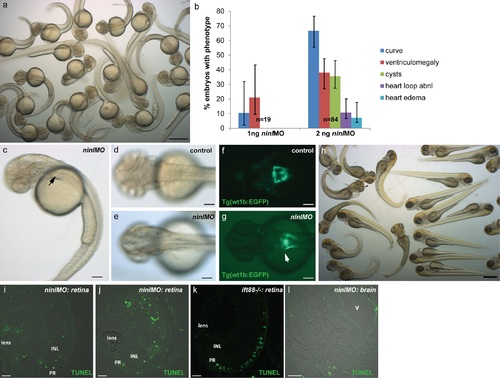
Phenotypes of the ninl atgMO. (a) Representative clutch of zebrafish larvae at 2dpf injected with the phenotypic dose of ninl atgMO (2ng/nl). (b) Titration curve for the ninl atgMO illustrating the distribution of phenotypes in 2dpf larvae at two different concentrations: at 1ng/nl, a minority of injected larvae present a curved body shape (10%) and/or ventriculomegaly (20%) (n = 19). At ~2ng/nl, on average 66% of injected larvae present a curved body shape and ~40% present ventriculomegaly and/or pronephric cysts (n = 84). 95% Confidence Interval bars are shown. (c) Representative 2dpf-old ninl atg-morphant displaying curved body shape, ventriculomegaly and pronephric cyst (arrow). (d-e) Dorsal view of 2dpf larvae showing the normal morphology of the brain folds in wild-type (d) and the enlarged ventricle in morphants (e). (f-g) Transgenic Tg(wt1b:EGFP) zebrafish line used to highlight the larval pronephros, shows the morphology of the fused glomerulus and proximal tubules in wild-type (f) and the dilatation of the region in ninl morphants (“kidney cysts”, white arrow in g). (h) A clutch of ninl morphants at 4dpf. (i-j) TUNEL assay on 4dpf cryosections through retinas from ninl morphants shows the range of cell death detected (curved larvae in (h) were sectioned for the TUNEL assay). Note the absence of TUNEL-positive cells in the photoreceptor (PR) cell layer in the morphants. (k) The ift88-/- retina is used as a positive control, given the known death of photoreceptors in this mutant at 4dpf. (l) Cryosection through a 4dpf brain in a morphant larva displaying a dilated brain ventricle (v) shows no significant neuronal cell death. Scale bars represent 500 μm in (a) and (h), 100 μm in (c-g), 10 μm in (i-k) and 30 μm in (l). PR PhotoReceptors, INL Inner Nuclear Layer. |
|
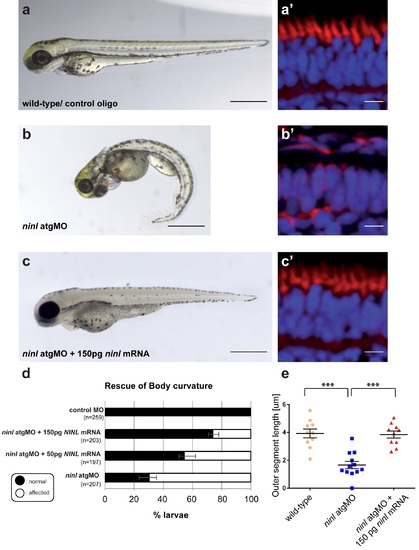
Rescue of the morphant phenotype supports its specificity. (a-d) Co-injection of 2 ng ninl atgMO with 150 pg capped MO-resistant mRNA encoding human NINL isoform B reduced the incidence of body curvature defects from 71% in ninl atgMO injected larvae (n = 207) to 36% in ninl atgMO + NINL mRNA injected larvae (n = 203) (P<0.0001, two-tailed Fisher's exact). A subset of these larvae were sectioned and a perfect correlation was observed in rescue between body curvature defects and defects in photoreceptor outer segment formation (a'-c'). (e) Quantification of the rescue of retinal outer segment length showing that mean OS length was rescued from 1.6 +/- 0.26 μm in ninl morphants to 3.8 +/- 0.25 μm with co-injection of NINL mRNA (P<0.0001, unpaired Student's t-test). PHENOTYPE:
|
|
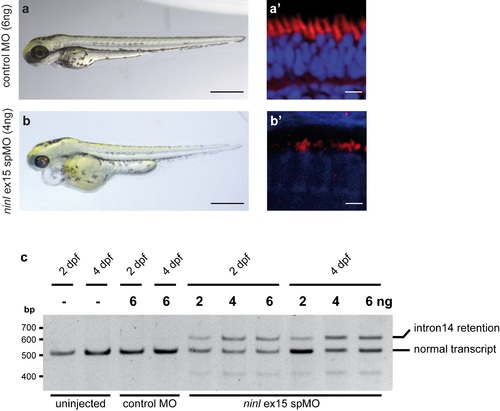
Recapitulation of phenotype by a ninl ex15 spMO (4 dpf). (a-b) Injection of 4ng ninl ex15 spMO (n>100) results in heart edema and small eyes. No defects in body curvature were observed in comparison to control MO-injected larvae. (a', b') Analyses of bodipy-stained retinas of ninl ex15 spMO-injected larvae (n = 10) revealed defects in photoreceptor outer segment formation (10 of 10) similar to those observed in ninl atgMO-treated larvae, whereas stained retinas of control MO-injected larvae (n = 10) appeared normal (10 of 10). Scale bars represent 500μm (a-b) and 5μm (a'-b'). (c) RT-PCR analysis on RNA isolated from 25 larvae that were either uninjected, injected with control MO (6ng) or injected with various amounts of ninl ex15 spMO (2, 4, 6ng), collected at two different time points after injection (2 dpf and 4 dpf). One PCR product of the expected length (~500bp) was obtained from RNA from uninjected and control MO-injected larvae. Sequence analysis revealed that this was the predicted transcript including exons 13–16. RT-PCR analysis on RNA obtained from the morphant larvae resulted in two products: Sequence analysis of both fragments revealed that the shorter product is the predicted wild-type transcript (ex13-16) and that the longer transcript in addition includes the entire intron 14 (85 bp), resulting in premature termination of translation already after two codons in intron 14. This aberrant splicing persists at 4dpf. PHENOTYPE:
|