- Title
-
Second harmonic generating (SHG) nanoprobes for in vivo imaging
- Authors
- Pantazis, P., Maloney, J., Wu, D., and Fraser, S.E.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
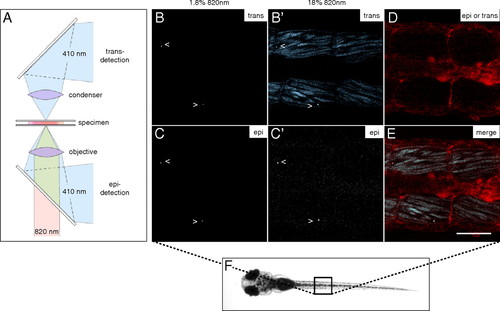
SHG nanoprobes provide superior signal-to-noise ratio after in vivo injections BaTiO3 nanocrystals were injected into one-cell stage zebrafish embryos. Several days after cytoplasmic injection (around 72 hpf) excitation of nanocrystals with femtosecond pulsed 820-nm light results in strong SHG signal detectable in epidirection as well as in transdirection (A) throughout the whole zebrafish body [here, nanocrystals (arrowheads) present in the trunk of a zebrafish (B–C′)]. Endogenous SHG from trunk muscles can only be detected in transdirection with the sarcomere repeat patterns clearly observable (B′). (D) BODIPY TR methyl ester dye labeling the extracellular matrix and cell membranes. (E) Merge of pictures B′, C′, and D. Note that the power levels required to detect endogenous SHG are 10 times higher than those to visualize BaTiO3. The SHG signal intensity of BaTiO3 is comparable in epi- as well as in transdetection mode. Anterior to the left. Bar = 75 μm. |
|
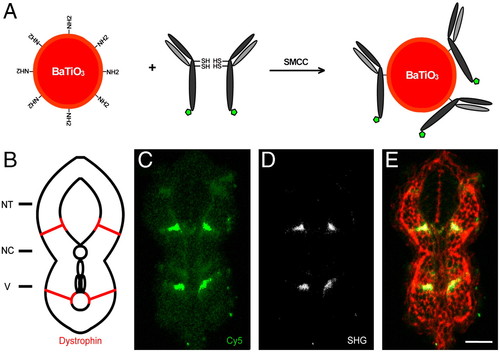
SHG nanoprobe targeting specificity. (A) Scheme of SHG nanoprobe conjugation to Cy5-coupled (green pentagon) antibody fragments via disulphide reduction and sulfhydryl-amine coupling. (B) Schematic representation of a transversal section of trunk of a 24 hpf zebrafish embryo showing Dystrophin protein localization to fiber ends (red). NT, neural tube; NC, notochord; V, vessels. (C–E) Immunostaining showing Dystrophin protein localization in a transversal tissue section using secondary antibody coupled to Cy5 (green) and BaTiO3 (white). Both readouts—Cy5 immunofluorescence (C), and BaTiO3 SHG signal (D)—label specifically Dystrophin. (E) Phalloidin labeling (red) is superimposed to show cell profiles in the transversal tissue section. Note that SHG immunostaining provides superior SNR of Dystrophin detection using a narrow emission filter. Bar = 30 μm. |
|
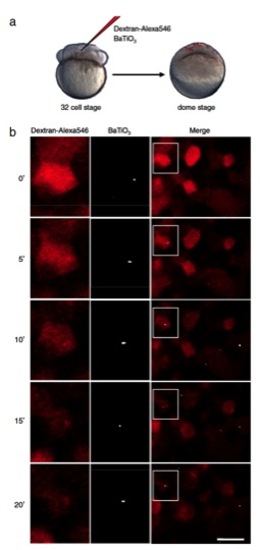
SHG nanoprobes provide superior signal-to-noise ratio and do not bleach after in vivo injections. (A) Single-cell coinjections of BaTiO3 nanoparticles and 10,000 MW Dextran-Alexa546 were performed at the 32-cell stage and imaged at the dome stage of zebrafish embryos. (B) Time-lapse images of individual cells at the dome stage of a zebrafish embryo marked with BaTiO3 nanocrystals (Center column) and Dextran-Alexa546 (Left column). Right column is the merged picture with white boxes magnified in the center and left columns. Times (0′, 5′, 10′, 15′, and 20′) indicate minutes at and after the start of the experiment. Note that continuous scanning with intense excitation power levels bleaches quickly the organic dye, whereas the SHG signal of BaTiO3 stays unchanged, allowing long-term nanoparticle tracking. Anterior to the left. Bar corresponds to 20 μm. |
|
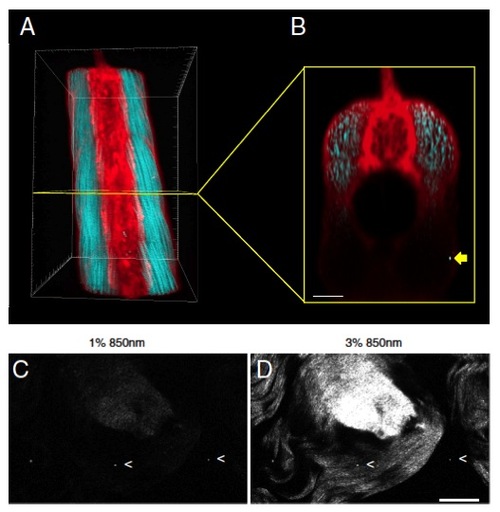
SHG nanoprobes can be readily detected with increased imaging depth and provide superior signal-to-noise ratio when present in mammalian tail tendon. (A and B) BaTiO3 nanocrystals were injected into one-cell stage colorless zebrafish embryos. Several days after cytoplasmic injection (around 72 hpf) part of the zebrafish trunk was imaged with femtosecond pulsed 820-nm light. (A) Dorsal 3D reconstruction of the zebrafish trunk showing endogenous SHG signal from trunk-muscles detected in transdirection (blue), SHG of BaTiO3 nanocrystals detected in epi- as well as in transdetection (white), and BODIPY TR methyl ester dye labeling the extracellular matrix and cell membranes (red). Rostral front, caudal back. (B) Axial view of a slice of the trunk 3D reconstruction (yellow box). Whereas endogenous SHG and the vital dye are only readily detectable in the first 100 μm, the cross-sectional view through the trunk muscle shows that intense SHG signal of BaTiO3 nanocrystals is still detectable with increased imaging depth [here, nanocrystals (yellow arrow) present in the trunk of a zebrafish at an imaging depth of around 200 μm]. Note that the power levels required to detect endogenous SHG are 10 times higher than those to visualize BaTiO3. Bar corresponds to 50 μm. (C and D) BaTiO3 nanocrystals present in mouse tail slices of approximately 2–3-mm thickness. (C) One percent excitation with femtosecond pulsed 850-nm light results in strong SHG signal from diffraction-limited BaTiO3 nanocrystals (two of three spots marked with arrowheads) together with weak endogenous SHG signal originating from collagen in connective tissue of mouse tail tendon. (D) Increasing the excitation power level to 3% results in robust endogenous SHG signal with regular and highly organized collagen fiber clearly observable. Note that the power levels required to detect a strong signal from SHG nanocrystal signal spots is lower than for endogenous SHG of very highly aligned collagen fibers in mouse tail tendon. Bar corresponds to 50 μm. |