- Title
-
Vgll2a is required for neural crest cell survival during zebrafish craniofacial development
- Authors
- Johnson, C.W., Hernandez-Lagunas, L., Feng, W., Melvin, V.S., Williams, T., and Artinger, K.B.
- Source
- Full text @ Dev. Biol.
|
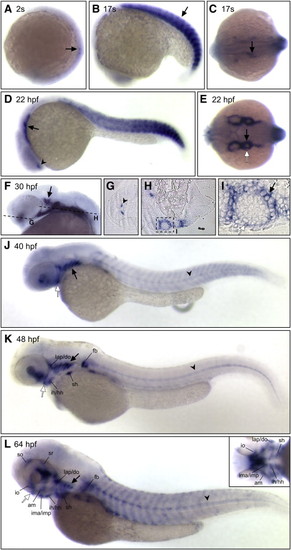
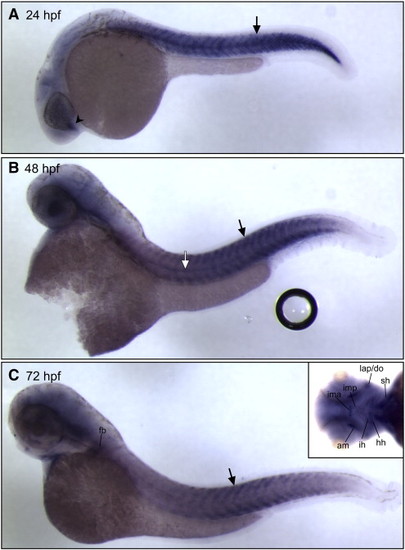
vgll2a expression in the developing zebrafish. In situ hybridization (ISH) using a vgll2a probe at 2 somites (A), 17 somites (B, C), 22 hpf (D, E), 30 hpf (F–I), 40 hpf (J), 48 hpf (K) and 64 hpf (L) stages showing lateral (A, B, D, F, J, K, L), dorsal (C, E) or ventral (inset, L) views. 4 µm plastic sections (G, H, I) of 30 hpf embryos sectioned in planes shown in panel F. vgll2a is first detected in the developing somites (black arrows, A and B) of zebrafish at the 2-somite stage (A). By 17 somites (B,C), vgll2a is also expressed at low levels in a population of endodermal cells (black arrow, C) as they evaginate to form the first pharyngeal pouch. At 22 hpf, lateral (D) and dorsal (E) views reveal that vgll2a is also expressed in the lateral pharyngeal endoderm (black arrows, D, E) as the pouches continue to form, as well as in the pharyngeal ectoderm (white arrow, E) and ventral forebrain (black arrowhead, D). At 30 hpf, expression of vgll2a is also apparent in the region of the developing trigeminal ganglion (black arrow, F) Dashed lines indicate plane of section in G–I. 4 µm plastic sections of 30 hpf embryos (G, H, I) reveal that vgll2a expression, in addition to being expressed within a discrete population of cells within the ventral forebrain (black arrowhead, G) and prominently in the pharyngeal pouch endoderm (H, and black arrow in higher magnification view of boxed region in, I), is also expressed in the overlying pharyngeal ectoderm (white arrow, I). From 30 to 48 hpf (F–K), vgll2a continues to be expressed in the endodermal pouches (black arrow, J,K), somite expression fades, and forebrain expression expands. At 40 hpf and later stages, vgll2a is expressed in the stomodeum, the presumptive mouth (white arrow, J, K, L). By 48 hpf (K), it begins to be expressed in sites of myogenesis other than the somites, including the fin bud (fb) and cephalic musculature and is also apparent in the notochord (black arrowhead). Cranial muscle expression is more apparent by 64 hpf (L), at which time vgll2a becomes reactivated in the somitic muscle. Anterior is to the left in all panels. am, adductor mandibulae; do, dilator operculi; hh, hyohyoideus; ih, interhyoideus; ima, intermandibularis anterior; imp, intermandibularis posterior; io, inferior oblique; lap, levator arcus palatini; sh, sternohyoideus; so, superior oblique; sr, superior rectus. EXPRESSION / LABELING:
|
|
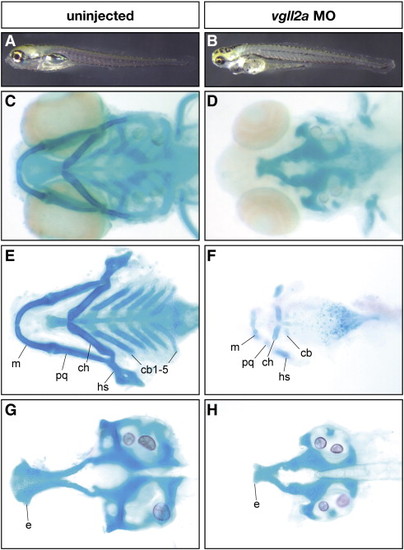
Pharyngeal cartilage development is affected in vgll2a morphants. Uninjected (A,C,E,G) and vgll2a morphant (B,D,F,H) embryos at 5 dpf (A,B) and alcian blue staining of these embryos (C–H) in whole mount (C,D), and after dissection to isolate the viscerocranium (E,F) and neurocranium (G,H). A typical embryo injected with 5 ng vgll2a ATG morpholino (B) exhibits a reduction in jaw structure relative to an uninjected control (A). Staining of the cranial cartilages with alcian blue in whole mount (C–D) and in dissected embryos (E–H) reveals the absence of the ceratobranchials 1–5 and hypoplastic Meckel′s, palatoquadrate, ceratohyal and hyosymplectic cartilages in vgll2a morphants (F) relative to uninjected controls (E). Additionally, the anterior part of the neurocranium, the ethmoid plate, is truncated in vgll2a morphants (H) relative to controls (G). Anterior is to the left in all panels. e, ethmoid plate; cb, ceratobranchial; ch, ceratohyal; hs, hyosymplectic; m, Meckel′s cartilage; pq, palatoquadrate. PHENOTYPE:
|
|
Endodermal pouch morphogenesis is affected in vgll2a morphants. Single confocal, Z-plane images of an uninjected (A) and vgll2a morphant embryo (B), stained with the zn-8 antibody, marking the endodermal pouches in lateral views. Uninjected embryos (A) exhibit elongated endodermal pouches, numbered 1–5, formed by tight folds of epithelium at 38 hpf. In contrast, the endodermal pouches of embryos injected with 5 ng vgll2a ATG MO (B) are shorter and wider, and the epithelial cells appear less organized relative to uninjected controls. Anterior is to the left in both panels. |
|
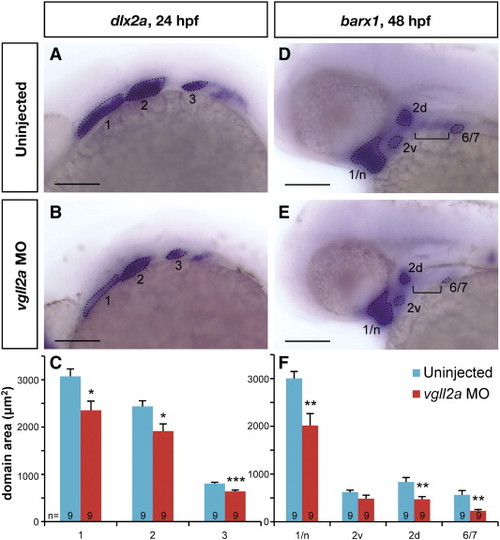
vgll2a knockdown causes a reduction in the size of NCC domains. Later views of representative uninjected (A,D) and vgll2a morphant (B,E) embryos processed by ISH with probes for dlx2a at 24 hpf (A,B) and barx1 at 48 hpf (D,E). Dotted lines outlined in panels A,B,D and E represent the domain areas quantified (C,F). The domains of NCC marker dlx2a (A,B) in the first three arches (numbered 1–3) are smaller in vgll2a morphants (B) relative to uninjected controls (A) at 24 hpf. barx1 is expressed in cranial NCC condensations at 48 hpf (D,E) and expression domains including the neurocranium and arch 1 (1/n), dorsal and ventral domains within arch 2 (2d and 2v) and a domain representing arches 6 and 7 (6/7) are similarly reduced in vgll2a morphants (E) relative to controls (D). barx1 expression in the anterior ceratobranchials (bracket, D,E) was not detected in vgll2a morphant embryos (E). Graphs of the average area in these embryos reflect these differences (C, F). Error bars represent standard error of the mean. All domains measured were significantly smaller in vgll2a morphants except 2v (Student′s T Test,*: p < 0.05, **: p < 0.01, ***: p < 0.001). Scare bars = 100 µm. Numbers at the base of each bar represent the number of embryos counted for each condition. |
|
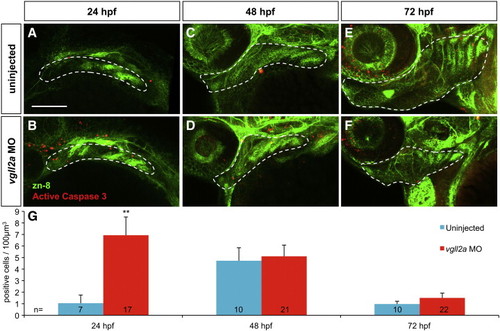
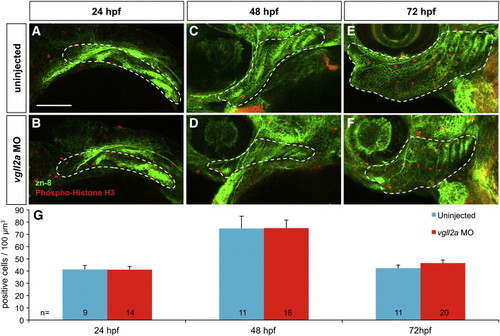
Cell death within the pharyngeal arches is increased in vgll2a morphants. Three-dimensional projections of confocal slices of representative uninjected (A,C,E) and vgll2a morphant embryos (B,D,F) at 24 (A,B) 48 (C,D) and 72 hpf (E,F) stained with zn-8 (anti-Alcama) and anti-activated caspase 3 antibodies in lateral views, and quantification of cell death in uninjected and morphant embryos (G). Embryos injected with 5 ng vgll2a ATG MO (B,D,F) exhibit a greater number of activated caspase 3 positive cells (red) per μm3 within arches volumes modeled within the white dashed lines based on zn-8 staining (green) relative to uninjected controls (A, C, E), at 24, but not 48 or 72 hpf. (G) Quantification of the average number of active caspase 3 positive cells / 100 μm3 of arch volume per embryo (Student′s T Test, **: p < 0.01). Error bars represent standard error of the mean. Numbers at the base of each bar represent the number of embryos counted for each condition. Scale bar = 100 μm. Anterior is to the left in panels A-F. PHENOTYPE:
|
|
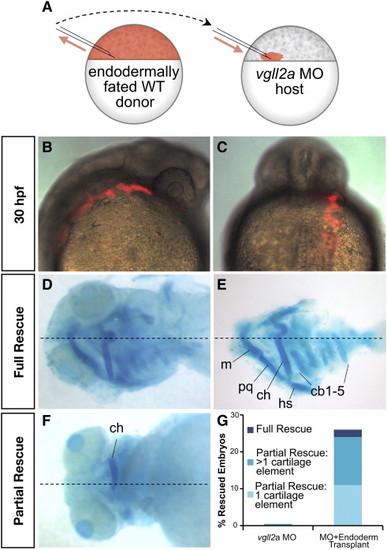
Expression of vgll2a in the pharyngeal endoderm is sufficient for facial cartilage development. Schematic representation of transplantation performed with endodermally fated wildtype vgll2 expressing (WT) cells (A). To create mosaic embryos that express vgll2a within the pharyngeal endoderm but are otherwise Vgll2a knockdown in all other cells, donor embryos were injected at the single-cell stage with cas/sox32 mRNA to fate these embryos to become endoderm along with a rhodamine-dextran tracer. At blastoderm stage, cells were transplanted from the margin of these endodermally fated donor embryos to the margin of vgll2a morphant embryos (A). In lateral (B,) and dorsoposterior (C) views of a representative mosaic embryo at 30 hpf following transplantation, these cells appear red by fluorescent microscopy and often contribute to one side of the pharyngeal endoderm. In ventral views at 5 dpf, alcian blue staining reveals that pharyngeal cartilage development is completely (wholemount, D; and dissected, E) or partially (wholemount,F) rescued in some of these embryos. Quantification of number vgll2a morphant embryos that exhibited rescue upon introduction of endoderm from embryos expressing vgll2a (G). Rescue was scored as rescue of one cartilage element, rescue of more than one cartilage element, or rescue of all cartilage elements (full rescue) relative to morphants and expressed as a percent of the total number of mosaic embryos analyzed (G, n = 46). 26% of endoderm transplants showed partial or full rescue. Dotted lines mark the approximate midline. cb, ceratobranchial; ch, ceratohyal; hs, hyosymplectic; m, Meckel′s cartilage; pq, palatoquadrate. Anterior is to the right in B and to the left in D-F. |
|
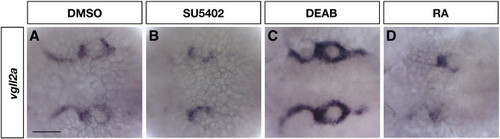
Expression of vgll2a is regulated by FGF and RA signaling. Dorsal views of embryos processed by ISH using a vgll2a probe following treatment with DMSO (control, A), SU5402 (B), DEAB (C), and RA (E). Embryos treated with 20 μM SU5402 to inhibit FGF signaling for one hour from 23 to 24 hpf and processed for by ISH for vgll2a expression show a reduction in vgll2a mRNA in the endodermal and ectodermal pharyngeal epithelia surrounding the arches (B, n = 22/23) compared to DMSO-treated control embryos (A, n = 32/34). Embryos treated from 23 to 24 hpf with 100 μM DEAB to inhibit RA synthesis (C, n = 25/27) exhibit increased vgll2a expression in the pharyngeal epithelia while treatment with 100 nM all-trans RA (D, n = 25/25) results in reduced expression of vgll2a in this domain. Other domains of vgll2a expression were not affected in these treatments (data not shown). Anterior is to the left in all panels. EXPRESSION / LABELING:
|
|
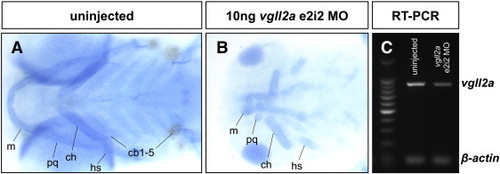
Injection with vgll2a e2i2 MO results in reduced vgll2a message and defects of the pharyngeal skeleton. A representative example of an uninjected embryo (A) and an embryo injected with 10 ng vgll2a e2i2 MO stained with alcian blue at 5 dpf in a lateral view (B) and RT-PCR of the vgll2a message in these embryos (C). Injection with 10 ng vgll2a e2i2 MO resulted in hypoplasia or absence of the pharyngeal cartilages as stained with alcian blue (B, n = 6/6), similar to the phenotype observed upon injection with the vgll2a ATG MO (Fig. 2D). RT-PCR of the entire vgll2a coding sequence (C) shows 10 ng vgll2a e2i2 MO results in reduced vgll2a message (44% reduction as measured by densitometry). Anterior is to the left in A and B. PHENOTYPE:
|
|
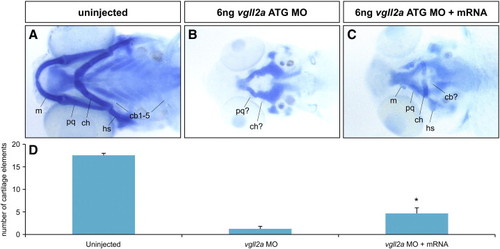
The pharyngeal cartilage phenotype of vgll2a morphants can be partially rescued by mRNA encoding vgll2a. Examples of 5 dpf larvae stained with alcian blue in ventral views (A–C) and quantification of the number of pharyngeal cartilage elements visible for each condition examined (D). In wildtype (A) the total number of pharyngeal cartilage elements at 5 dpf is normally 18 (n = 9). Embryos injected at the 1 cell stage with 100 pg vgll2aΔMO mRNA in addition to 6 ng vgll2a ATG MO injected at the 2 to 4 cell stage (n = 9) averaged 4.7 alcian-positive pharyngeal cartilage elements, significantly more (Student′s T test, *: p < 0.05) than those injected withvgll2a ATG MO alone (n = 8), which averaged 1.3. Error bars represent standard error of the mean. Anterior is to the left in A–C. |
|
vgll2b expression in the developing zebrafish. Embryos processed by ISH using a probe to detect vgll2b expression at 24 (A), 48 (B), and 72 hpf (C) in lateral views. vgll2b is expressed in the somites (black arrow, A) and forebrain (arrowhead, A) at 24 hpf (A). At 48 hpf (B), vgll2b is expression is apparent in the somites (black arrow, B) and the notochord (white arrow, B). By 72 hpf (C), vgll2b is also expressed in other sites of myogenesis, including the fin buds (fb) and the cephalic muscles, which are more clearly observed in the ventral view (C, inset). am, adductor mandibulae; do, dilator operculi; hh, hyohyoideus; ih, interhyoideus; ima, intermandibularis anterior; imp, intermandibularis posterior; lap, levator arcus palatini; sh, sternohyoideus. EXPRESSION / LABELING:
|
|
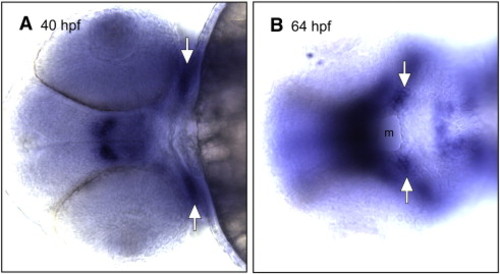
vgll2a expression in the developing zebrafish mouth. ISH using a probe against vgll2a on zebrafish embryos at 40 (A) and 64 hpf (B) in ventral views. At 40 hpf (A), vgll2a is expressed at the lateral edges of the stomodeum (white arrows, A), the presumptive mouth, prior to its opening. vgll2a continues to be expressed in the commissure (white arrows, B) of the now open mouth (m) at 64 hpf (B). |
|
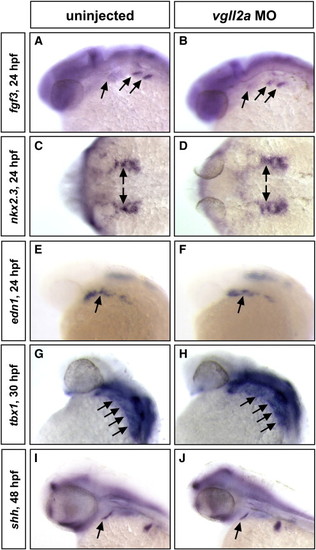
Gene expression in the pharyngeal endoderm is unaffected in vgll2a morphants. ISHs of fgf3 at 24 hpf (A,B), nkx2.3 at 24 hpf (C,D), edn1 at 24 hpf (E,F), tbx1 at 30 hpf (G,H), and shh at 48 hpf (I,J) in uninjected (A,C,E,G,I) and vgll2a morphant (B,D,F,H,J) embryos. Black arrows in all panels point to domains of endodermal expression within the pharyngeal arches. Anterior is to the left in all panels. |
|
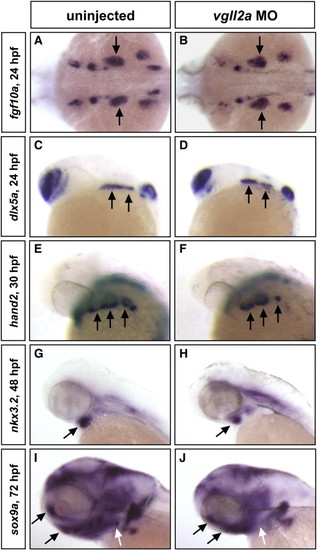
Gene expression in the NCCs of the arches is unaffected in vgll2a morphants. ISHs of fgf10a at 24 hpf (A,B), dlx5a at 24 hpf (C,D), hand2 at 24 hpf (E,F), nkx3.2 at 48 hpf (G,H), and sox9a at 72 hpf (I,J) in uninjected (A,C,E,G,I) and vgll2a morphant (B,D,F,H,J) embryos. Black arrows in all panels point to domains of arch NCC expression. White arrows denote the absence of sox9a expression in the posterior arches or morphants relative to uninjected embryos. Anterior is to the left in all panels. |
|
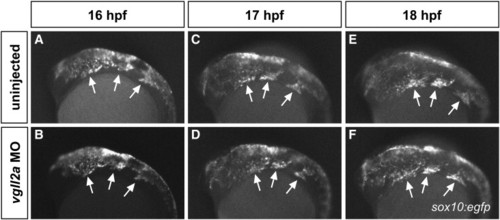
Cranial NCC migration is unaffected in vgll2a morphants. Fluorescent images of uninjected (A,C,E) and vgll2a ATG morpholino injected (B,D,F), sox10:egfp embryos at 16 (A,B), 17 (C,D) and 18 hpf (E,F). Arrows in all panels point to the three streams of migratory cranial NCCs. Anterior is to the left in all panels. |
|
Proliferation within the arches is not affected in vgll2a morphants. Three-dimensional projections of confocal slices of representative uninjected (A,C,E) and vgll2a morphant embryos (B,D,F) at 24 (A,B) 48 (C,D) and 72 hpf (E,F) stained with zn-8 (anti-Alcama) and anti-phospho-histone H3 antibodies in lateral views, and quantification of cell death in uninjected and morphant embryos. (A, C, E) Uninjected and (B, D, F) morphant embryos injected with 5 ng vgll2a ATG MO exhibit a similar number of proliferating cells (red) per 100 μm3 within arches volumes modeled within the white dashed lines based on zn-8 staining (green) relative to uninjected controls, at 24, 48 and 72 hpf. (G) Quantification of the average number of phospho-histone H3 positive cells / 100 μm3 of arch volume per embryo. Error bars represent standard error of the mean. Numbers at the base of each bar represent the number of embryos counted for each condition. Scale bar = 100 μm. |
|
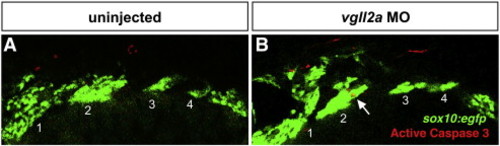
Apoptotic NCCs appear in the arches of vgll2a morphants but not in uninjected embryos at 24 hpf. Single-plane, confocal images showing the pharyngeal arches of a representative uninjected sox10:egfp embryo (A) and a sox10:egfp embryo injected with 5 ng vgll2a MO and 2 ng p53 MO (B) and stained with an anti-active caspase 3 antibody in lateral views. Apoptotic, anti-active caspase 3-postitive NCCs (arrow), were only observed in the arches of vgll2a morphants at 24 hpf. Arches are numbered 1–4. Anterior is to the left in both panels. |

Unillustrated author statements EXPRESSION / LABELING:
|
Reprinted from Developmental Biology, 357(1), Johnson, C.W., Hernandez-Lagunas, L., Feng, W., Melvin, V.S., Williams, T., and Artinger, K.B., Vgll2a is required for neural crest cell survival during zebrafish craniofacial development, 269-81, Copyright (2011) with permission from Elsevier. Full text @ Dev. Biol.