- Title
-
Schwann cells reposition a peripheral nerve to isolate it from postembryonic remodeling of its targets
- Authors
- Raphael, A.R., Perlin, J.R., and Talbot, W.S.
- Source
- Full text @ Development
|
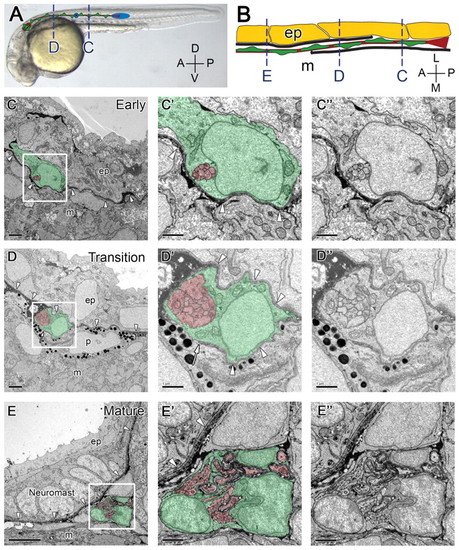
The PLLn invades the epidermal basement membrane. (A) 28 hpf embryo with schematic of the PLLn superimposed. Schwann cells are green, axons and the ganglion are red, and the primordium and a pre-neuromast (asterisk) are blue. Broken lines refer to approximate location of cross-sections shown in C and D. (B) Schematic representation of the transition of the PLLn across the epidermal basement membrane. Basement membrane is shown in black, Schwann cells in green, axons in red, epidermal cells in yellow; only one of two epidermal cell layers is shown. Broken lines correspond to cross-sections in C-E. (C) At 28 hpf, the distal segments of axons and associated Schwann cells of the PLLn are located within the epidermis (n=6). Arrowheads indicate the epidermal basement membrane. (D) At locations more anterior to that shown in C at 28 hpf, the PLLn is found in a transition state, with basement membrane located on both sides of the nerve (arrowheads). Arrow indicates the end of the basement membrane (n=6). (E) By 3 dpf, the PLLn is found at its mature location, medial to the epidermal basement membrane (arrowheads, n=9). A neuromast is present within the epidermis. (C′,C′,D′,D′,E′,E′) Higher magnification of boxed regions in C,D,E, with and without pseudocoloring. Scale bars: 2 μm in C,D; 5 μm in E; 1 μm in C′,D′,E′. Schwann cells are pseudocolored green, axons are red. Abbreviations: D, dorsal; V, ventral; A, anterior; P, posterior; L, lateral; M, medial; ep, epidermis; m, muscle; p, pigment. |
|
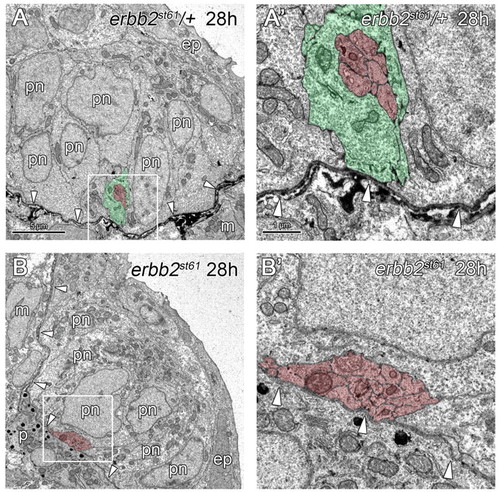
PLLn outgrowth is normal in Schwann cell-deficient mutants. (A,B) At 28 hpf, axons and Schwann cells of heterozygous siblings (A) and axons of erbb2st61 mutants (B) are located within the epidermis (ep) (n=3 in A and n=6 in B), and are associated with pre-neuromast cells (pn). (A′,B′) Higher magnification of the boxed regions in A,B, respectively. Arrowheads indicate the location of the epidermal basement membrane. Scale bars: 5 μm in A,B; 1 μm in A′,B′. Schwann cells are pseudocolored green and axons in red. Abbreviations: ep, epidermis; m, muscle; pn, pre-neuromast cell. |
|
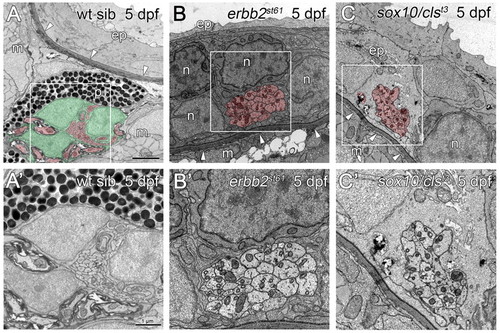
The PLLn is mislocalized in mutants lacking Schwann cells. (A) At 5 dpf, the PLLn of wild-type siblings has migrated from the epidermis (ep), across the basement membrane (arrowheads) into the muscle (m) (n=28). (B,C) By contrast, at 5 dpf in the Schwann cell-deficient mutants erbb2st61 (B) and sox10/clst3 (C), the PLLn remains in the epidermis, outside of the basement membrane (arrowheads, n=7 in B and n=6 in C). Defasciculation of the mutant nerves is also observed (asterisk, C). (A′,B′,C′) Higher magnification of boxed regions in A,B,C, respectively. Scale bars: 2 μm in A,B,C; 1 μm in A′,B′,C′. Schwann cells are pseudocolored green and axons red. Abbreviations: ep, epidermis; m, muscle; p, pigment; n, neuromast cell. |
|
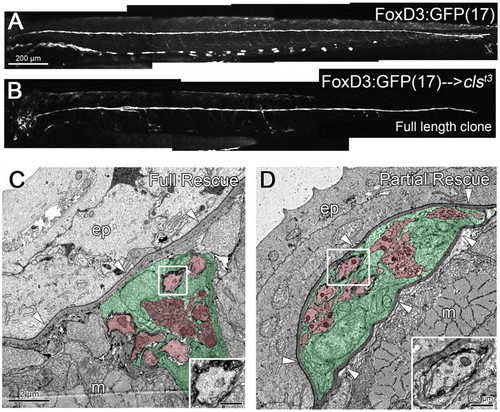
Full-length clones of transplanted wild-type Schwann cells can rescue nerve position in sox10/clst3 mutants. (A) Wild-type 3 dpf embryo carrying the FoxD3:GFP(17) transgene, labeling the PLLn, motor nerves and some pigment cells. (B) FoxD3:GFP(17) cells have been transplanted into a sox10/clst3 mutant host, creating a clone of wild-type Schwann cells that traverse the entire length of the PLLn (3 dpf). (C) Transplanted wild-type Schwann cells can reposition the mutant nerve in the subepidermal space, below the basement membrane (arrowheads). (D) Transplanted wild-type Schwann cells partially rescue the mutant nerve, positioning it within the epidermal basement membrane (arrowheads). Insets show the presence of myelinated axons in C,D. Scale bars: 200 μm for A,B; 2 μm for C,D. Schwann cells are pseudocolored green and axons red in C,D. Abbreviations: ep, epidermis; m, muscle. |
|
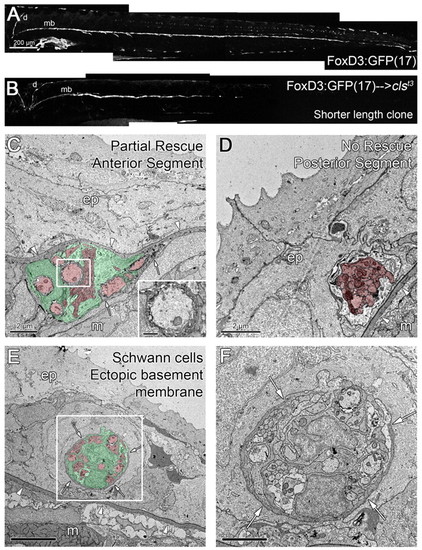
Shorter length clones of transplanted wild-type Schwann cells can partially rescue the position of sox10/clst3 mutant nerves. (A) Wild-type 5 dpf embryo carrying the FoxD3:GFP(17) transgene, labeling the PLLn [dorsal (d) and midbody (mb, tracts], motor nerves and some pigment cells. (B) A clone of wild-type, FoxD3:GFP(17) cells that are present on the dorsal and anterior regions of the midbody tract of the PLLn. (C,D) Micrographs of the anterior (C) and posterior (D) regions of a partial length clone of rescued Schwann cells, as shown in B. (C) Wild-type Schwann cells rescue the position of the mutant nerve, but a part of the old basement membrane remains (arrows). Inset in C shows the presence of myelinated axons. (D) The posterior region of the nerve lacks Schwann cells and is not rescued, remaining within the epidermis. (E,F) In this chimera, wild-type Schwann cells failed to rescue the position of the PLLn, but induced the formation of an ectopic basement membrane surrounding the nerve in the epidermis (arrows). (F) Higher magnification of boxed area in E. Arrowheads indicate the epidermal basement membrane; arrows indicate ectopic basement membrane. Scale bars: 200 μm for A,B; 2 μm for C,D,F; 5 μm for E. Schwann cells are pseudocolored green and axons red in C-E. Abbreviations: d, dorsal tract; mb, midbody tract; ep, epidermis; m, muscle. |
|
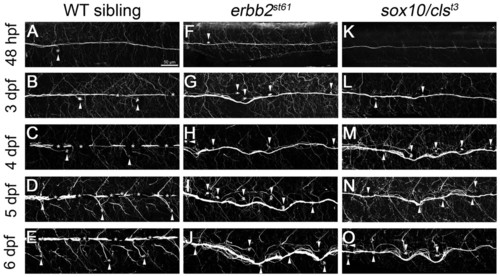
Nerves become disorganized during development in mutants lacking Schwann cells. (A-O) Antibody staining for acetylated tubulin shows the position of the PLLn. (A-E) Wild-type nerves remain anchored at the horizontal myoseptum and run straight along the length of the embryo at all times examined (48 hpf, 3-6 dpf), but the neuromasts migrate ventrally over time (arrowheads). Some axons branch from the main bundle to innervate individual neuromasts. (F-O) Nerves in erbb2st61 (F-J) and sox10/clst3 (K-O) mutants appear normal at 48 hpf but become progressively disorganized (3-6 dpf) and often track with migrating neuromasts (arrowheads). Excess neuromasts are present in nerves lacking Schwann cells (F-O). Asterisks indicate the presence of pigment cells. Scale bar: 50 μm. |