- Title
-
A vertebrate-specific Chp-PAK-PIX pathway maintains E-cadherin at adherens junctions during zebrafish epiboly
- Authors
- Tay, H.G., Ng, Y.W., and Manser, E.
- Source
- Full text @ PLoS One
|
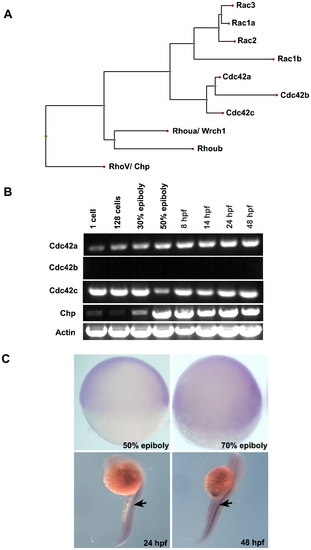
Protein sequence analysis and spatio-temporal expression of Chp. (A) The relationship between Chp, Cdc42 and Rac in zebrafish (Danio rerio). Protein sequences were aligned using ClustalW (DNAStar) and the dendrogram generated by Phylip. Accession numbers for cDNA sequences of Cdc42 and Rac family are: Rac1a, AY865568. Rac1b, XP_001332092.1. Rac2, AY865569. Rac3, AY865570. Cdc42a, AY865566. Cdc42b, XM_678979. Cdc42c, AY865567. Rhoua, AY865564. Rhoub, NM_001017784, Chp/ RhoV, NM_001012250. (B) RT-PCR products showing mRNA transcript profile of zebrafish Chp at different developmental stages as indicated. Both Cdc42a and Cdc42c were detected throughout the stages tested. Chp transcripts appear at epiboly. (C) Whole mount in situ hybridization (WISH) using dioxygenin (DIG) anti-sense Chp RNA probes indicates all cell types express the transcript at 50% and 80% epiboly (lateral views): Chp mRNA is enriched in notochord (indicated by arrows) at 24 hpf and 48 hpf. |
|
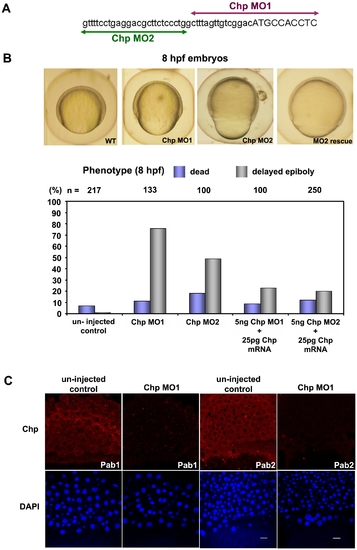
Chp function is required for epibolic movements. (A) Positions and sequence of Chp morpholino-oligoncleotides used in the study. The Chp MO1 and MO2 (corresponding to the reverse strand) are represented in purple and green respectively; lowercase sequence is 5′ UTR region. (B) Phenotypic examination of Chp morphants at 8 hpf revealed delayed closure of the yolk plug when compared with the un-injected controls. Rescue of the phenotype was performed by co-injecting Chp MO 1/2 with WT Chp (Zf) mRNA. Co-injection of 25 pg Chp mRNA with 5 ng Chp MO1/2 efficiently reduced the number of yolk plug defects at 8 hpf. Phenotypic analysis shows significant rescue in the delay of yolk plug closure at 8 hpf. (C) Immuno-histochemical staining of un-injected controls and Chp morphants at 60% epiboly using Chp antibody raised and purified from two animals (Pab1 and Pab2). Confocal images show endogenous Chp is strongly depleted Chp morphants. Decreased protein levels were observed in EVL, DEL and YSL layers with Chp MO. Embryos were counter-stained with DAPI. Scale bars = 20 μm. EXPRESSION / LABELING:
PHENOTYPE:
|
|
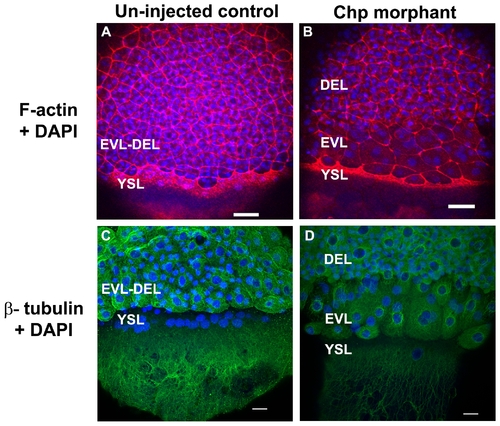
The actin cytoskeleton and microtubule networks in Chp morphants. Low resolution confocal images (10x objective) comparing (A) un-injected control and (B) Chp morphant with respect to F-actin organization at the vegetal margins of EVL and DEL, and at the external yolk syncytial layer (YSL). The deep cells marked by DAPI stained nuclei, fail to properly migrate in Chp morphants (B and D). As a result the cell margin is thinner and the F-actin ring of the EVL and YSL appears more compact. Confocal images at higher magnification (40x) comparing (C) un-injected control and (D) Chp morphant stained with β-tubulin. Microtubule organization in Chp morphant was largely normal. Scale bars represent 40 μm in panels A-B and 20 μm in panels C-D. PHENOTYPE:
|
|
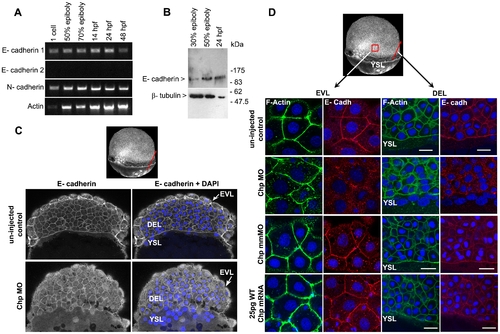
E-cadh is not maintained at the AJs in the absence of Chp. (A) RT-PCR products of zebrafish E-cadh1, E-cadh2 and N-cadherin (N-cadh) at the stages shown. E-cadh1 transcript was present throughout all stages however we did not detect expression of E-cadh2 (although it is represented 3 times in the EST database). N-cadh transcripts accumulate more significantly at 50% epiboly. Primers encoding the cytoplasmic domain of E-cadh1 NM_131820, E-cadh2 XM_690906 and N-cadh AF 418565 were used. (B) Immunoblot analysis of endogenous E-cadh at the stages indicated. The E-cadh antibody (BD Biosciences) detects a single E-cadh band (approx. 120kDa). (C) Low resolution image of phalloidin stained embryo of 60% epiboly, marked with red arrow representing the ‘y’ component in xy cross-section of the embryo (EVL→ DEL→ YSL) where the confocal image is taken. A middle sagittal plane of the embryo at 60% epiboly is derived from the Z stack. E-cadh was no longer maintained at the AJs in the EVL and was found predominately in cytoplasm. (D) Schematic diagram (3D view) of 60% epiboly. Red box indicates the lateral area of EVL and red arrow indicates the ‘y vector’ of the xy confocal slice. Confocal images (40x mag.) showing E-cadh co-localized with F-actin at the AJs between adjacent cells in the EVL and DEL. These observations were similar with the controls that were injected with 25 pg WT Chp mRNA alone and Chp mmMO. The images represent a stack of 10 images (each 0.5 μm); E-cadh staining was rarely detected at AJs in Chp morphant but intracellular signal was not diminished, suggesting E-cadh is mis-localized without Chp signaling. Identical loss of E-cadh was observed (data not shown) with a Mab that recognized the extracellular domain of E-cadh (ECM Biosciences; CM1681). Scale bars represent 20 μm. |
|
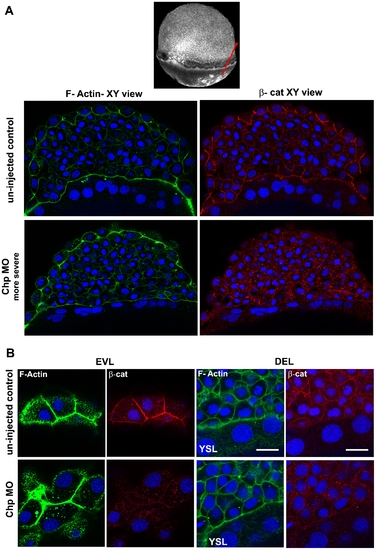
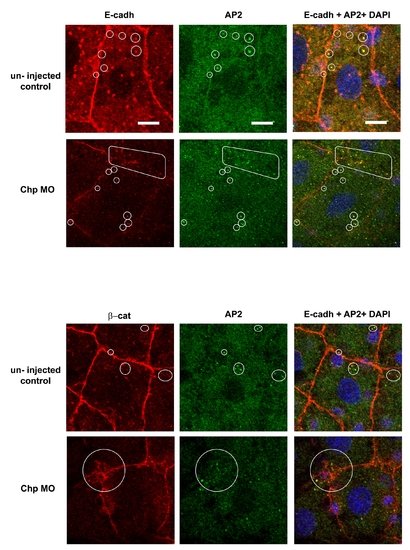
Localization of β-cat at the AJs requires Chp signaling. (A) A middle sagittal plane of the embryo at 60% epiboly immunostained with β-cat is also derived from the Z stack. β-cat was no longer maintained at the AJs in the EVL and was found predominately in cytoplasm. Scale bars represent 20 μm. (B) Immunostaining of β-cat comparing the EVL and DEL of un–injected control and Chp morphant. Levels of anti-β-cat staining at AJs were severely reduced in Chp morphants. The delocalized β-cat protein was detected within intracellular structures, but no nuclear enrichment was observed relative to controls. |
|
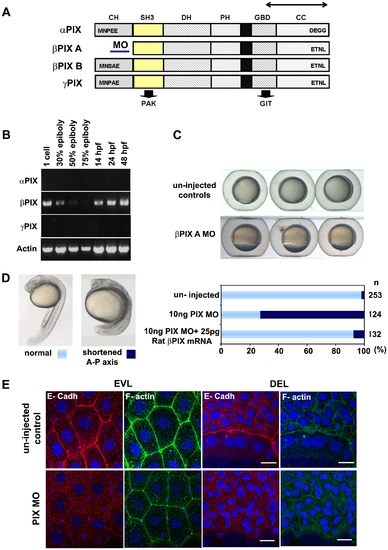
PIX is required to localize E-cadh at the AJs. (A) Schematic of zebrafish PIX isoforms designated αPIX, βPIX-A, βPIX-B [7] and the newly described γPIX. Arrows indicate the positions of the oligonucleotide primers used for RT-PCR. The position of the PIX-MO at the 5′ UTR of βPIX-A is indicated: this transcript encodes the smaller PIX isoform which is equivalent to the ubiquitous mammalian βPIX. SH3 domain in yellow and GBD binds to PAK and GIT respectively. (B) Transcript profile showing RT-PCR products for PIX at the developmental stages indicated. Primers cover essentially the same region of the PIX ORFs and therefore do not discriminate between the alternate spliced forms at the 5′ terminus. (C) PIX morphant embryos exhibit epibolic delay compared to un-injected controls at 8 hpf. (D) The typical phenotype of embryos depleted of βPIX-A at 24 hpf, exhibit shortened AP axes suggesting gastrulation defects. Phenotypic analysis showing significant rescue at 24 hpf after co-injection with 25 pg of rat βPIX mRNA. (E) Reduced cell junctional E-cadh signals in the EVL and DEL after PIX knock-down. The level of cortical F-actin (phalloidin) is similar to controls but the junctional network is more irregular. Intracellular E-cadh puncta suggest PIX functions downstream of Chp to maintain E-cadh at cell adhesions. Scale bars = 20 μm. |
|
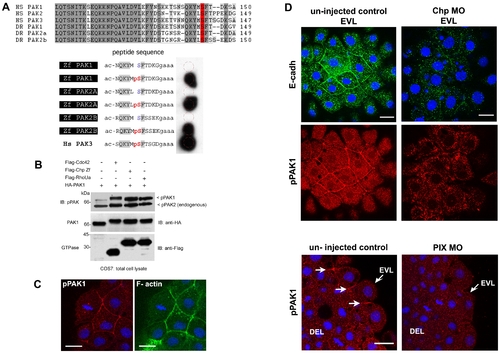
PAK lies downstream of Chp signaling and active kinase co-localizes with E-cadh. (A) Characterization of the phospho-PAK (pPAK) antibody. Alignment of auto-inhibitory domain (AID) of human (Hs) and zebrafish (Zf) PAKs: HsPak1, NP_001122092.1; HsPak2, NP_002568.2; HsPak3, NP_002569.1; ZfPak1, NP_958485.1; ZfPak2a, NP_001002717; and ZfPak2b, NP_001020627.1. Dark and light grey shaded sequences represent identical and conserved amino acids; the serine residue highlighted in red is the phosphorylated site recognized by the rabbit anti-pPAK144 antibody. Further characterization of this new antibody will be presented elsewhere; it is ∼10 times more sensitive than the anti-PAK1 pSer199 described previously in zebrafish [85]. Anti-pS144PAK1 recognizes all zebrafish PAK isoforms as assessed by synthetic phospho-peptides representing zebrafish PAK sequences. The peptides were synthesized in situ on cellulose (Jerinini) with a 3 amino-acid linker at the C-terminal end, and N-terminally amidated. The filter was blocked with BSA, and probed with anti-pPAK and HRP-anti-rabbit IgG antibodies both at 1:2000 (30 min each, with 3x10 min washes). Human PAK3 (pS139) is shown as a positive control. (B) Constitutively active Chp (G38V) can activate PAK1. Active Cdc42(G12V), Chp(G38V) and RhoUa(Q104L) were cloned in the mammalian expression vector pXJ-Flag (with CMV promoter) and co-expressed with HA-tagged PAK1 in COS-7 cells. The activation of PAK1 is indicated by an upshift in the PAK1 band and by PAK1 phosphorylation on Ser144. (C) Activated pPAK1 was detected at the centrosome of mitotic cell (as previously reported in mammalian cell culture) and can be found at the cell junctions of envelope cells (EVL), but not on the junctions of deeper cells. (D) E-cadh colocalizes with pPAK1 at the AJs of the EVL but not in the cytoplasmic puncta. Both were reduced at the AJs of the EVL in Chp morphants. Junctional pPAK signal is reduced in the envelope cells in PIX morphants. Typical stainings of control and PIX morphant embryos. Both images represent a stack of 3 confocal images, collected under the same laser and gain settings, and at equivalent positions in the embryo. White arrows represent junctional pPAK1. Scale bars represent 20 μm. |
|
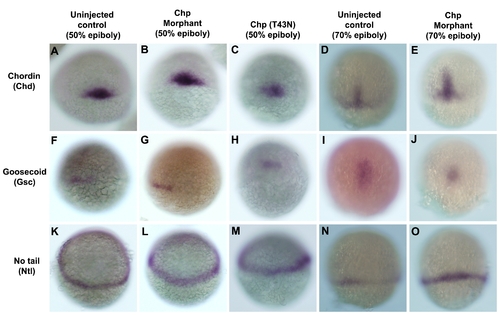
Early patterning and organizer specification of un-injected control and Chp morphants. WISH for mesodermal markers; (A-E) chordin (Chd), (F-J) goosecoid (Gsc) and (K-O) no tail (Ntl) in un-injected controls, Chp morphants or embryos injected with Chp(T43N). Reduced activity of Chp did not affect the expressions of Ntl, Gsc and Chd at 50% and 70% epiboly although the overall morphology of the mutants is affected. Panels A-I are embryos at the shield stage. Panels J-O are embryos at 70% epiboly. Panels A-J provides dorsal midline views, and panels K-O lateral views. |
|
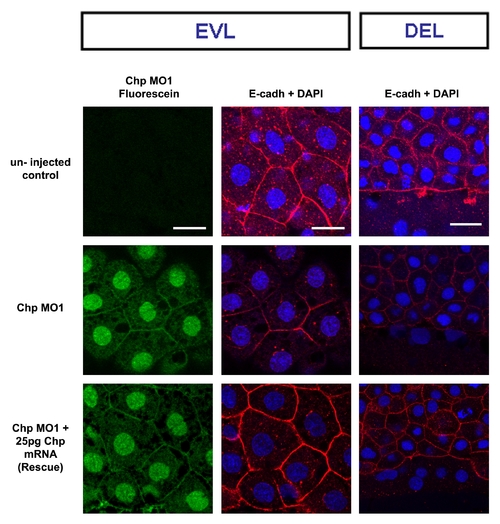
Chp MO1 rescue by synthetic mRNA co-injection. Chp MO1 conjugated with fluorescein blocks E-cadh localization to AJs in EVL at 60% epiboly. The panels show typical phenotype for rescued of E-Cadh localization by the co-injection with 25pg Chp mRNA. Scale bars represent 20 μm. |
|
A portion of E-cadh co-localizes with intracellular AP-2 vesicles. Confocal images (zoomed) of E-cadh with AP-2 in EVL cells. The un-injected controls and Chp MO2 injected embryos are compared. The image is a single confocal slice of 0.5 μm step size. The loss of the Chp signal leads to E-cadh depletion from AJs and becoming associated primarily with intracellular AP-2 vesicles clustered near the AJs. Scale bars represent 20 μm. |