- Title
-
Nkx2.7 and Nkx2.5 function redundantly and are required for cardiac morphogenesis of zebrafish embryos
- Authors
- Tu, C.T., Yang, T.C., and Tsai, H.J.
- Source
- Full text @ PLoS One
|
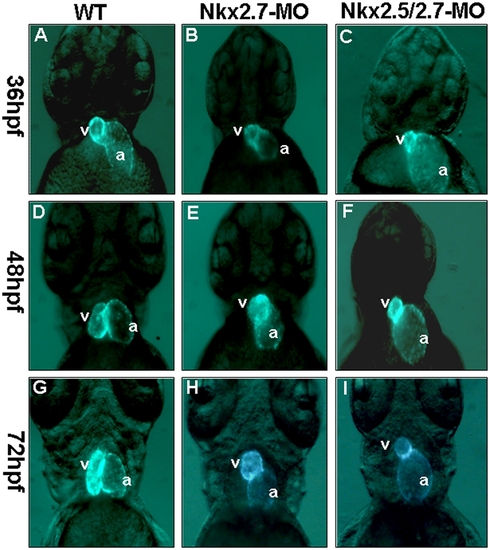
The defective phenotypes of zebrafish embryo heart injected with Nkx2.5-MO, Nkx2.7-MO and Nkx2.5/2.7-MO. Eight nanograms of MO were injected into one-cell stage embryos derived from transgenic line Tg (cmlc2::GFP) to knock down the Nkx protein specifically. The embryos are shown at 36 hpf (A, B, C), 48 hpf (D, E, F), and 72 hpf (G, H, I). The heart phenotype of Nkx2.5-MO embryos was similar to that of control embryos whose ventricle is located at the right side of the atrium when embryos were observed at 36 hpf, 48 hpf and 72 hpf from the ventral view under fluorescence microscope (A, D, G). However, embryos injected with Nkx2.7-MO displayed an unlooping defect from 36 hpf to 72 hpf (B, E, H). Embryos injected with Nkx2.5/2.7-MO displayed a shrunken ventricle and an expanding atrium (C, F, I). v: ventricle; a: atrium. EXPRESSION / LABELING:
PHENOTYPE:
|
|
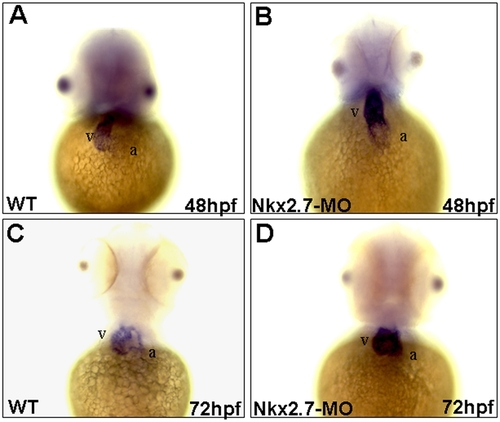
Nkx2.5 was overexpressed in the Nkx2.7-knockdown embryos. Nine nanograms of Nkx2.7-MO were injected into one-cell stage of embryos. Nkx2.5 was expressed predominantly in ventricle (v) and weakly in atrium (a) of wild-type (WT) embryos at 48 hpf (A), but only minimally and weakly in ventricle at 72 hpf (C). However, the Nkx2.7-MO-injected embryos retained robust Nkx2.5 expression in ventricle both at 48 and 72 hpf (B and D). The embryonic stages were as indicated, and embryos were observed ventrally. v: ventricle; a: atrium. EXPRESSION / LABELING:
PHENOTYPE:
|
|
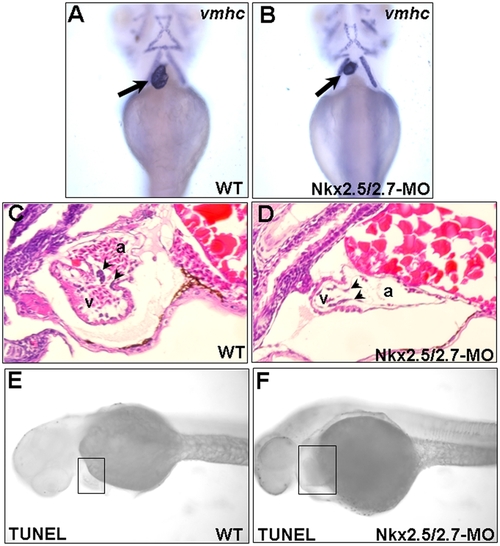
Ventricle becomes smaller and consists of a single layer in the Nkx2.5 and Nkx2.7 double knockdown morphants (Nkx2.5/2.7-MO). Ventricular myosin heavy chain (vmhc) was used as a probe to detect the ventricle morphology in (A) wild-type (WT) and (B) Nkx2.5/2.7-MO embryos at 72 hpf. The ventricle of Nkx2.5/2.7-MO embryos was smaller than that of WT embryos (indicated by arrows). Hematoxylin and eosin staining showed that ventricular myocardium of WT was two or more cell layers in thickness. However, only one cell layer was retained in the ventricular myocardium of the Nkx2.5/2.7-MO embryos. In addition, compared to wild-type embryos, the endocardium of Nkx2.5/2.7-MO-injected embryos did not form endocardial cushion (indicated by arrowheads in C and D). Like WT embryos, TUNEL assay did not display the increase of TUNEL-positive cells in the heart region of Nkx2.5/2.7-MO embryos at 40 hpf (indicated by boxes in E and F). Embryos were observed ventrally (A–B) or laterally (C–F). v: ventricle; a: atrium. EXPRESSION / LABELING:
PHENOTYPE:
|
|
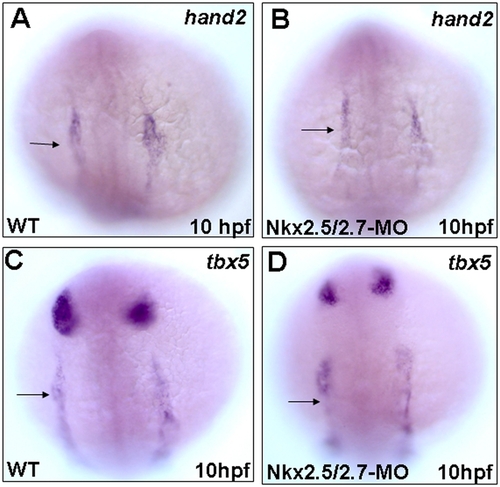
Early cardiac markers express normally in the Nkx2.5 and Nkx2.7 double knockdown morphants (Nkx2.5/2.7-MO). Whole mount in situ hybridization showed that the expression patterns of hand2 and tbx5, lateral plate mesoderm markers, were similar between wild-type (WT) and Nkx2.5/2.7-MO embryos at 10 hpf (A vs. B; C vs. D, respectively). Arrows: heart field. EXPRESSION / LABELING:
|
|
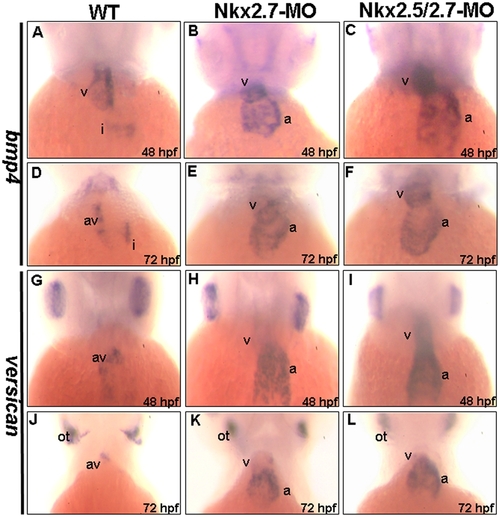
Abnormal cardiac differentiation occurred in the Nkx2.7-knockdown zebrafish embryos. The expressions of bmp4 (A–F) and versican (G–L) in hearts were compared between wild-type (WT) (A, D, G, J), Nkx2.7-MO- (B, E, H, K) and Nkx2.5/2.7-MO- injected embryos (C, F, I, L) at 48 (A–C, G–I) and 72 hpf (D–F, J–L). In WT embryos, bmp4 was expressed in the ventricle and inflow tract at 48 hpf (A), and then bmp4 was restricted in its expression at the AV boundary at 72 hpf (D). However, in the Nkx2.7-MO (B, E) and Nkx2.5/2.7-MO (C, F) embryos, bmp4 was still expressed predominantly in the ventricle and atrium from 48 to 72 hpf. Similarly, in WT embryos, the versican expression was more predominant in ventricle than in atrium, at about 31 to 33 hpf, and then versican was confined in its expression at the AV boundary after 33 hpf (G, J). In contrast, in the Nkx2.7-MO (H, K) and Nkx2.5/2.7-MO (I, L) embryos, the versican was significantly expressed in the atrium and ventricle. In addition, the versican expression pattern in otoliths remained unchanged (J, K, and L). All images are ventral views, anterior to the top. a: atrium; v: ventricle; i: inflow tract; av: atrioventricular boundary; ot: otoliths. EXPRESSION / LABELING:
PHENOTYPE:
|
|
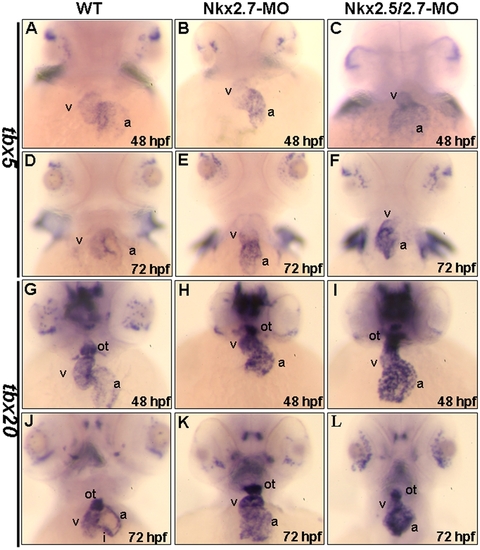
Chamber maturation was affected in the Nkx2.7-knockdown zebrafish embryos. The expressions of tbx5 (A–F) and tbx20 (G–L) in hearts were compared between wild-type (WT) (A, D, G, J), Nkx2.7-MO- (B, E, H, K) and Nkx2.5/2.7-MO- injected embryos (C, F, I, L) at 48 (A–C, G–I) and 72 hpf (D–F, J–L). In WT embryos, tbx5 was expressed strongly in ventricle, but weakly in atrium at 48 hpf and beyond (A, D). However, in the Nkx2.7-MO (B, E) and Nkx2.5/2.7-MO (C, F) embryos, tbx5 retained its strong expression in the heart, although the expression pattern was gradually changed from ventricle-enriched expression to atrium-enriched expression from 48 hpf to 72 hpf (B and E; C and F). In WT embryos, tbx20 expression was similar to the expression gradient of tbx5 in the heart at 48 hpf, and the tbx20 expression was stronger than that of tbx5. In addition, tbx20 expression was also detected in outflow tract at 48 hpf (G), and tbx20 expression was restricted to outflow tract and inflow tract by 72 hpf (J). However, in Nkx2.7-MO (H, K) and Nkx2.5/2.7-MO (I, L) embryos, tbx20 was expressed strongly in the heart and outflow tract from 48 to 72 hpf. All images are ventral views, anterior to the top. v: ventricle; a: atrium. ot: outflow tract; i: inflow tract. EXPRESSION / LABELING:
PHENOTYPE:
|
|
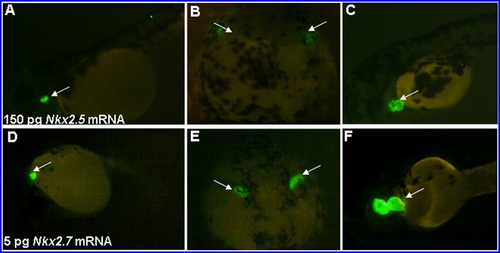
Overexpression of Nkx2.5 and Nkx2.7 in embryos resulted in serious heart defects. Amounts equaling 150 pg Nkx2.5 mRNA (A–C) and 5 pg Nkx2.7 mRNA (D–F) were injected individually into one-celled stage embryos derived from transgenic line Tg(cmlc2::GFP), whose hearts were specifically tagged with green fluorescent protein. We observed that there were many phenotypes of heart defects at 48 hpf resulting from the overexpression of either Nkx2.5 mRNA or Nkx2.7 mRNA. These defects included malposition of the reducing heart (A, D), bilateral heart (B, E), small heart (C) and large heart (F). Interestingly, overexpression by injection of Nkx2.7 mRNA as low as 5 pg caused an effect similar to that induced by injection 30 greater times than that of Nkx2.5 mRNA. (A, C, D, F), lateral views; (B, E), dorsal views; Arrows: green fluorescent heart. |
|
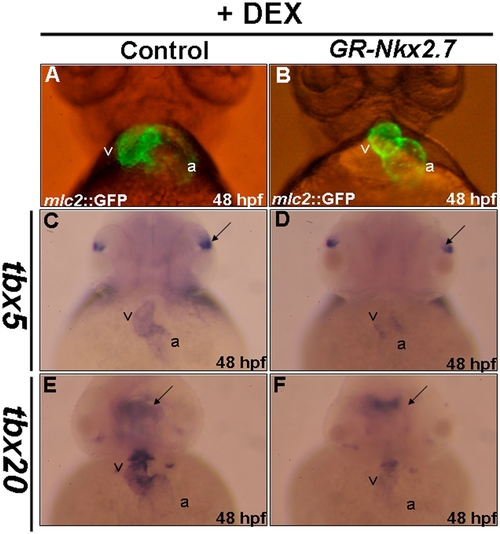
The expressions of tbx5 and tbx20 were modulated by Nkx2.7. Each embryo derived from transgenic line Tg(cmlc2::GFP), whose heart was specifically tagged with green fluorescent protein, was injected with 0.15 ng of plasmid, in which Nkx2.7 mRNA was transcribed conditionally by adding dexamethasone. All embryos were treated with dexamethasone at 10 hpf and took a ventral view at 48 hpf to observe the cardiac development under the fluorescence microscope. The cardiac morphologies of the control embryos and the GR-Nkx2.7-overexpression embryos were shown. Compared to the control (A), various cardiac defects were found in around 35% of embryos from the GR-Nkx2.7-overexpression group, including unlooping and shrunken heart (B). In addition, the expressions of tbx5 (C, D) and tbx20 (E, F) were down-regulated in the GR-Nkx2.7-overexpresion embryos. However, the expression levels of tbx5 in eyes (indicated by arrows in C and D) and tbx20 in brain (indicated by arrows in E and F) remained unchanged. v: ventricle; a: atrium. |

Unillustrated author statements PHENOTYPE:
|