- Title
-
Fgf8 drives myogenic progression of a novel lateral fast muscle fibre population in zebrafish
- Authors
- Groves, J.A., Hammond, C.L., and Hughes, S.M.
- Source
- Full text @ Development
|
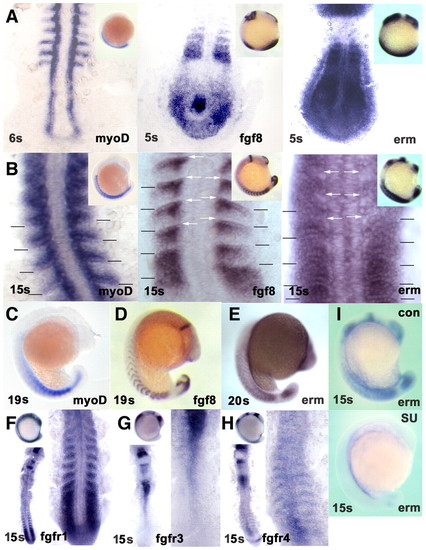
Fgf8 signalling correlates with myogenic marker expression in the lateral somite. In situ mRNA hybridisation for myod (A-C), fgf8 (A,B,D) and erm (A,B,E,I) at 4-6 s, 15 s and 19-20 s, and fgfr1 (F), fgfr3 (G) and fgfr4 (H) at 15 s. (A,B) Discreet expression of fgf8 overlaps or is immediately adjacent to locations of erm expression in somites. Myod expression parallels fgf8 and erm in the somites, but not elsewhere. Flatmount dorsal view, anterior towards the top. (Insets) Lateral view, anterior towards the top, dorsal towards the left. By 15 s, erm expression is becoming excluded from regions of high fgf8 expression in anterior somites (white arrows). myod and erm expression overlap within the posterior somite. Somite boundaries in the newest five somites are indicated. (C-E) myod, fgf8 and erm expression persist as somites mature. (F) fgfr1 is expressed in presomitic mesoderm and nascent somites. (G) fgfr3 shows little expression in nascent somites. (H) fgfr4 is expressed segmentally in maturing somites and in neural tube. (F-H) Dorsal flatmounts of whole embryo, anterior towards the top, showing tailbuds and newest somites at higher magnification (right). (I) Treatment with SU5402 blocks erm expression throughout the embryo. (C-E,I and insets in F-H) Lateral view, anterior towards the top. EXPRESSION / LABELING:
|
|
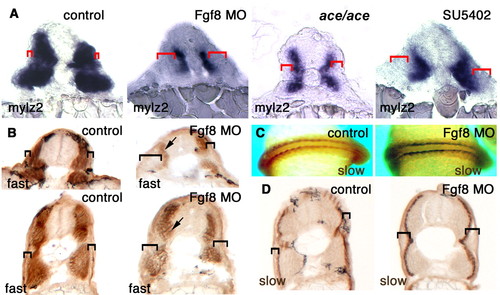
Fgf8 is required for fast muscle differentiation in the lateral somite. (A) Cryostat sections at the gut extension level of whole-mount in situ mRNA hybridisation for myosin light chain 2 (mylz2) at 20-22 s in unmanipulated control, Fgf8 MO-treated, ace mutant and SU5402-treated embryos. Red brackets show expansion of the lateral undifferentiated tissue. (B-D) Immunohistochemical detection of fast MyHC (B) or slow MyHC (C,D) on whole mounts at 15 s (C) or cryosections of Fgf8 MO-injected and control embryos at 33 hpf (B,D). Brackets indicate expansion of lateral non-muscle tissue, arrows show weakened fast MyHC. EXPRESSION / LABELING:
|
|
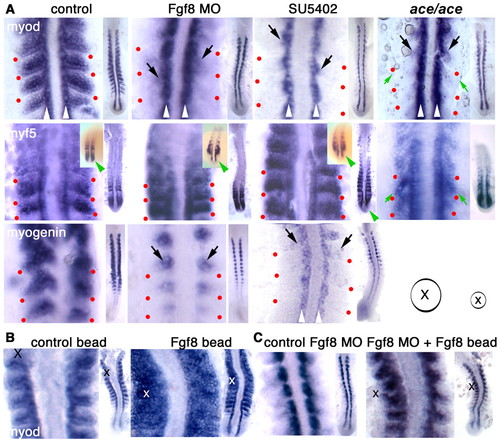
Fgf8 drives progression from myf5 to myod expression in the lateral somite. (A) Flatmounts of whole-mount in situ mRNA hybridisation for myod, myf5 and myogenin at 15 s in unmanipulated control, Fgf8 MO-treated, SU5402-treated and ace mutant embryos. In each panel, the entire posterior embryo is shown on the right in dorsal view, anterior upwards. On the left the youngest somites are magnified. Residual medial MRF expression is indicated (black arrows) adjacent to adaxial slow muscle cells (white arrowheads). The lateral extent of intersomitic border is marked (red dots) revealing the loss of lateral myod, and reduction in myogenin, but retention of myf5. Tailbud myf5 signal is lost in some Fgf8 MO and all SU5402-treated embryos (green arrowheads, whole mount insets, mildly affected individuals) but not in ace, and the narrowing of the tail of ace mutant on Kwt background with the intersomitic borders angled more sharply to the posterior (green arrows). (B,C) Myod expression 3 hours after implantation of Fgf8 or control beads at 10 s in unmanipulated (B) or Fgf8 MO-injected (C) embryos. Bead centres (X) and sizes (circle) relative to high- and low-magnification panels are indicated. EXPRESSION / LABELING:
|
|
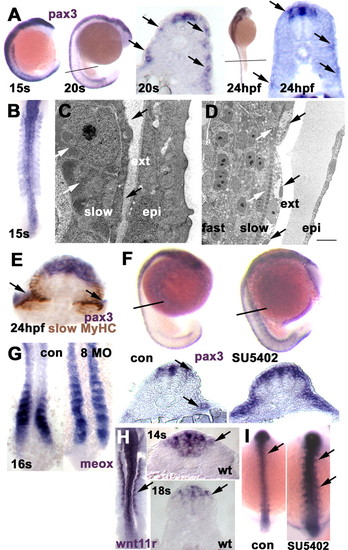
Pax3 marks the somitic external cell layer and is expanded when Fgf8 signalling is blocked. (A) Pax3 in situ mRNA hybridisation (blue/purple) with whole mounts showing expression in somites throughout the axis at 15 s, but more highly expressed in nascent somites by 20 s. Cryosections at the levels indicated reveal that somitic expression is in a discontinuous layer in the lateral region at 20 s (arrows), weakening further by 24 hpf and most prominent at the dorsal and ventral somitic extremes and the horizontal myoseptum level. There is persistent expression in dorsal neural tube. (B) Dorsal flat mount, anterior towards the top showing pax3 expression in all somites at 15 s. (C,D) Electron micrographs in transverse (C) and longitudinal (D) section of 24 hpf lateral somite at midbody level showing external cells (black arrows, ext) superficial to mononucleate slow muscle cells with pronounced medially located myofibrils (white arrows), which themselves overlie multinucleate fast fibres. The epidermis (epi) is separated from the external cells by amorphous material. Scale bar: 1.43 µm in C; 10 µm in D. (E) Oblique section through a heavily developed 24 hpf embryo showing pax3 expression in the region of the external cells (arrows) clearly superficial to slow MyHC (brown). (F) Compared with control 23 s siblings (con), SU5402-treated embryos show upregulation and maintenance of somitic pax3 both in whole mount and in cryosections. (G) Fgf8 MO reduces the downregulation of meox expression as somites mature (22/23 injected embryos). (H) wnt11r marks the dorsomedial somite edge (arrows) and neural tube. (Left) Dorsal flat mount at 15 s. (Right) Cryosections. (I) SU5402 upregulates wnt11r expression in both spinal cord and dorsomedial somite (56/56 treated embryos at 15 s). EXPRESSION / LABELING:
|
|
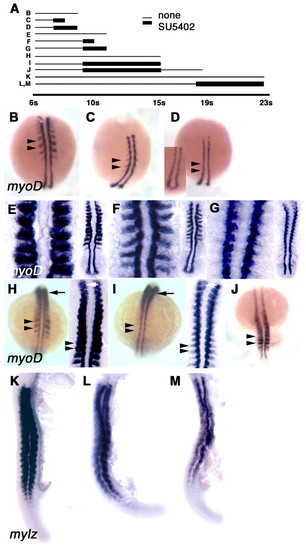
Persistent Fgf signalling is required for maintenance of myod expression and normal fast muscle differentiation. (A) Schematic showing timecourse of SU5402 treatment regimes (C,D,F,G,I,J,L,M) and control vehicle treatment (B,E,H,K). (B) Around 9 s, myod expression is clearly visible in each nascent somite (arrowheads). (C) Treatment with SU5402 for 30 minutes substantially reduces lateral myod expression (arrowheads). (D) Pre-treatment for 1 hour prevents all lateral expression (arrowheads). Inset in D shows adaxial myod expression in tailbud. (E) At 10-12 s, myod expression is well established in lateral cells of all somites. (F) Exposure to SU5402 for 30 minutes reduces lateral myod expression without noticeable effect on adaxial expression. (E-G) Dorsal flatmounts of posterior embryo (right) with nascent somites shown at high magnification (left). (G) Pre-treatment for 1 hour essentially ablates lateral myod expression. (H,I) SU5402 treatment for 2.5 hours from 10 s (I) abolishes new myod stripes (I, arrowheads; compare with control in H), but has less effect in anterior somites (arrows). Dorsal views of whole mount (left) and flat mount (right). There is similar expression in rostralmost somites (white arrows). (J) SU5402 treatment for 2.5 hours, followed by a 2-hour wash-out permits recovery of myod-expressing stripes in more than the four newly-formed somites (arrowheads). (K) At 23 s, mylz2 expression reveals abundant differentiated fast muscle. Lateral view, dorsal towards the left. (L,M) Moderate (L; 27/36 treated embryos) and severe (M; 5/36) examples of effect of late SU5402 application at 18 s to otherwise wild-type embryos. Anterior is towards top in all panels. EXPRESSION / LABELING:
|
|
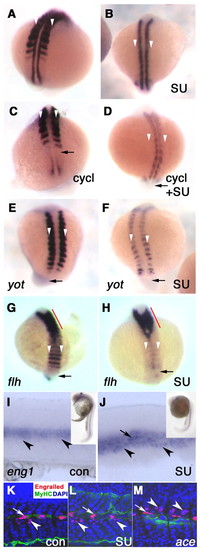
Midline signalling distinct from Hedgehog is required for medial fast myoblast myod expression. In situ hybridisation for myod mRNA in embryos without (A,C,E,G) or with SU5402 (B,D,F,H) from yot (A,B,E,F) or flh (G,H) heterozygote crosses or wild type treated with cyclopamine (C,D). (A,B) Control 15 s embryos show reduction of lateral myod expression after SU5402 treatment (B, white arrowheads). (C,D) Cyclopamine prevents maintenance of myod mRNA in adaxial cells (C, arrow), without affecting lateral expression. Addition of both cyclopamine and SU5402 ablates myod mRNA in adaxial (arrow, D) and lateral (white arrowheads, D) cells, leaving medial myod expression. (E,F) In 15 s yot/yot mutant embryos, adaxial myod mRNA is lacking (arrows). Lateral myod expression is severely reduced by SU5402 treatment (F, white arrowheads). (G,H) In flh/flh embryos, myod mRNA is lost from adaxial cells (arrows) but expressed in stripes across the bilaterally fused somites (G, white arrowheads). Additional SU5402 treatment ablates essentially all myod expression in nascent somites (H, white arrowheads). Strong anterior myod expression originating at the midline (red line) may be in recovering slow muscle driven by floor-plate signals (Blagden et al., 1997). (I-M) At 26 s, control (I,K), SU5402-treated (J,L) and ace mutant (M) embryos show maintenance of eng1 mRNA (I,J) and engrailed protein (K-M; single confocal slices with 4D9 anti-engrailed red, nuclei revealed with DAPI in blue) in both MP (arrow, labelled strongly with A4.1025 anti-MyHC green) and MFF (arrowheads). ace mutants were identified by lack of her5PAC:egfp in mid-hindbrain (Tallafuss and Bally-Cuif, 2003). EXPRESSION / LABELING:
|

Unillustrated author statements EXPRESSION / LABELING:
|